ABSTRACT
The use of plant biostimulants jointly or without macro or micro fertilization may be an alternative to change plant metabolism and consequently, improve bean crop yield. Under this circumstance, we aim to evaluate the influence of biostimulants and its interaction with macro and micronutrient fertilizers on common bean var. Pérola yield in Bahia State (BA). The research started on 12 December, 2013 in the experimental farm of the State University of Southwestern Bahia (UESB), campus in Vitória da Conquista – BA, Brazil. The experimental design used was the total randomized block with three replications, arranged in a factorial scheme (2x2x4) under two macronutrient fertilization levels (present or absent) and four biostimulants (Control, Stimulate, Booster and Biozyme), in which doses followed manufacturer recommendation for common bean crop. The following traits were assessed: plant height, stem diameter, first pod height, pod number, pod length, grain per plant, 100-grain dry mass and yield. The data was submitted to variance analysis, and means were compared by “F” and Scott Knott tests at 5% probability. We found that biostimulants enhanced common bean yield with or without macro and/ or micronutrient supply.
Key words: Plant regulators, nutrients, fertilization, yield and Phaseolus vulgaris L.
Common bean (Phaesolus vulgaris L.) is one of the most important domestic crops because of its high nutritional value and use as basic food in Brazil. The average consumption per capita, according to MAPA (2014), reaches 19 kg per year and seven out of ten Brazilians consume it daily. Common bean average yield in Bahia State (Brazil) is 509 Kg ha-1, below the Brazilian State yield, which is 1,032 Kg ha-1 (IBGE, 2014). However, when it is used in a higher technological level, bean yield may exceed 3,000 Kg ha-1 (Vieira et al., 2006). In Bahia, there are three main periods to cultivate beans: “rainy season” that is between November to December, “dry season” being in February and March and “winter beans” sown from July to September (Ferreira et al., 2002).
This State produces the eighth worse yield from Brazil. Producing 509 Kg ha-1, the State is only above seven others from Northeastern region. This value is 50.67% under national yield, which is 1,032 Kg ha-1 (IBGE, 2014).
In Vitória da Conquista – BA, Northeastern Brazil, most of the common bean producers are located at Mata de Cipó. The greatest part is small farmers that grow beans intercropped with other plants such as corn, cassava and coffee (Ganem, 2013). Crop yield in this region for 2011 was 398 Kg ha-1, which is 21.80% under state average yield and 61.43% under national yield (IBGE, 2014). Decreased yield of crops is mainly related to low rainfall during the year and low technology level applied by most of the farmers.
There are some factors that may promote crop yield and performance losses in the field, e.g. minimal use of certified seed, resistance to technical innovations, improper irrigation management etc. After all, in recent years, great farmers have been increasing interest for common bean cropping, due to worth prices of the last harvests, mainly the carioca beans that is the most consumed in the country, which corresponds to 62.8 % of national bean production (CONAB, 2014).
Production and yield of several crops are related to genetic performance and selection of new varieties with traits of interest. Nonetheless, despite advances in plant breeding programs, the long term to obtain new cultivars increases the use of alternative tools to enhance crop yield and efficiency in available resource use. In such context, plant regulators are considered as alternative to change plant metabolism and, consequently, crop yield and product quality (Almeida, 2011). As reported by Avila et al. (2010), yield might be improved by means of effective use of new and consolidated technologies for beans, especially fertilizer and irrigation management and biostimulant use. New biotechniques’ use such as bioregulators add to bean qualitative and quantitative traits (Alleoni et al., 2000), thereby availing the stress of overcoming diseases related to genetic (Bertolin et al., 2009) and environmental (Avila et al., 2010) factors.
Biostimulant narrow knowledge basis for beans combined with varied fertilization types support studies on this. As mentioned by Castro and Vieira (2011), plant biostimulants are defined as mixture of bioregulators with same or different chemical character (amino acids, vitamins, minerals etc.). Biostimulant use in agriculture to enhance commercial crop yield have been increasing within the last decades (Bourscheidt, 2011).
Researches on growth regulator usage associated or not with fertilizations have been increasingly common (Lana et al., 2009). Positive outcomes on biostimulant use were verified by several researches on Soybean (Bertolin et al., 2010), Grapes (Leão et al., 2005), Sugarcane (Miguel et al., 2009), Watermelon (Costa et al., 2008) and Caupi beans (Oliveira et al., 2013).
On the other hand, some surveys found no significant differences on Cotton (Baldo et al., 2006; Lima et al., 2006), Passion fruit (Ataíde et al., 2006) and Corn (Ferreira et al., 2008; Silva et al., 2008). Even though no significant difference was found for Beans, Alleoni et al. (2000) stated that biostimulants contribute to improve crop yield and other agronomical traits. Abrantes et al. (2011) observed that growth regulator application at appearance of the first flower bud increased grains per plant and grain yield for Carioca Precoce and IAC Apuã cultivars. Similarly, Cobucci et al. (2005) noticed Stimulate application at appearance of the first flower bud and appearance of the first pod promoted meaningful increment on bean yield. Just as in many other crops, results involving biostimulant usage have not always been significant for agronomical traits as reported by Bernardes et al. (2010) and Avila et al. (2010).
Accordingly, we aim to evaluate the influence of biostimulant use and its interaction with macro and micronutrient fertilizations on var. Pérola beans in Vitória da Conquista – BA, Brazil.
The experiment was carried out in the Experimental Farm of the State University of Southwestern Bahia (UESB), in Vitória da Conquista County, located at 14° 51' 58” S latitude and 40° 50' 22” W longitude. The city lies at an average altitude of 940 m and according to Köppen classification, the climate is High Altitude Tropical (Cwb). Mean annual temperature is 21°C and mean annual rainfall of 730 mm concentrated between November to March.
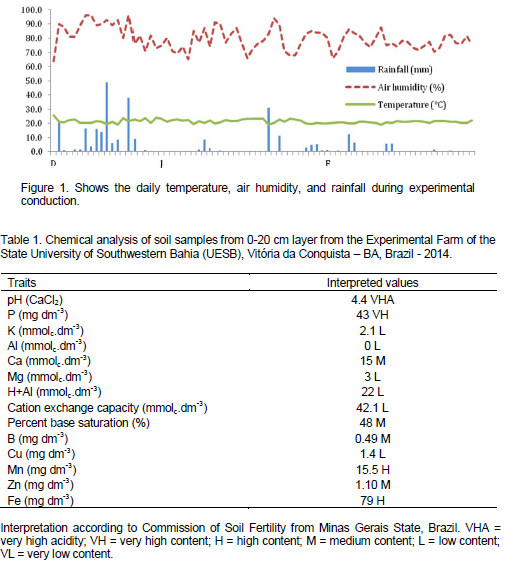
The temperature, relative humidity and rainfall that occurred daily during the test driving period, are shown in Figure 1 Accumulated rainfall along experiment was 292-mm. Complementary irrigation was performed due to water requirement at each stage through spray irrigation. Local soil is classified as a Dystrophic Tb Haplic Cambisol (Inceptisol) (Vieira et al., 1998) medium-textured and flat topography. For soil analyses, single samples were collected from 0 to 20 cm layer and together sent to the Soil Analyses Laboratory of the Campinas Agricultural Institute (IAC). Table 1 shows the chemical analysis of soil from the experimental area.
The experimental design was randomized block with three replications arranged in a factorial scheme of 2 x 2 x 4. Wherein there were two macronutrient fertilization levels (level 1 – absence of fertilization and level 2 – fertilization with 400 Kg ha-1 04-14-08 NPK at sowing and top dressing at opening of the first trifoliate leaf with 80 Kg ha-1 urea), two micronutrient fertilization levels (level 1 – absence of fertilization and level 2 – fertilization with 30 Kg ha-1 FTE - BR12 at sowing). In addition, four levels of biostimulant (Stimulate, Booster, Byozime TF, Control), totaling 48 plots was used.
The stimulate is manufactured by Stoller do Brasil, it is composed by 0.09 g L-1 cytokinin (kinetin) + 0.05 g L-1 indole-butyric acid + 0.05 g L-1 gibberellic acid (GA 3) and 4% molybdenum, and it is classified as slightly toxic and soluble concentrate solution (Stoller do Brasil, 1998). The Booster® - ZnMo is a liquid product with 2.3% molybdenum (Mo) and 3.5% zinc (Zn), 3.0% copper (Cu), auxin and cytokinin, manufactured by Agrichem. Arysta LifeScience (2013) manufactures the Biozyme TF, it is a liquid fertilizer for foliar application, containing in the formulation, macro and micronutrients associated with hydrolyzed vegetal extracts (2.43% Zn; 1.73 % N; 5% K2O; 0.08% B; 0.49% Fe; 1% Mn; and 2.1% S). This later product is obtained from natural extracts and it has similar effects to the main plant growth-promoting hormones (cytokinins, auxins and gibberellins), micronutrients and other biologically active molecules.
Dosages, number and phenological stages of applications were followed according manufacturers:
1. Absence
2. Two applications of 200 ml ha-1 each, at opening of the third trifoliate leaf and appearance of the first flower bud using Stimulate.
3. Two applications of 100 mL ha-1 each, at opening of the third trifoliate leaf and appearance of the first flower bud using Booster
4. Three applications of 200 mL ha1 each, at opening of the third trifoliate leaf, appearance of the first flower bud and appearance of the first pod using Byozime TF.
The experiment commenced on December 12, 2013. Soil preparation was plowing and level harrowing, followed by furrow plough spaced by 50 cm using a 10 cm depth chisel plow. Seeds were manually sown into furrows, at 5 cm depth with 13 seeds per meter. Then, ten days after sow, thinning was performed leaving a density of eight plants per meter. Thus, a 160,000 plant/ hectare stand was obtained as suggested by Barbosa and Gonzaga (2012).
Crop handlings, such as pest control and irrigation were performed according to crop needs. Top dressing nitrogen fertilization for NPK treated plots was carried out at opening of the first trifoliate leaf stage. Stimulate and Booster were applied at 21 and 33 days after emergence (DAE) during opening of the third trifoliate leaf and appearance of the first flower bud stages, respectively. Moreover, Byozime TF was applied at 21, 33 and 60 DAE, at opening of the third trifoliate leaf, appearance of the first flower bud and appearance of the first pod respectively. Biostimulant application was made through spraying with a precision knapsack sprayer at CO2 constant pressure of 2.0 kgf cm-2, equipped with sprayer boom and flat fan jet nozzles (110 01) at 30 cm above plant canopy, spray outflow of 300 L ha-1 (Abrantes et al., 2011). Portable plastic curtains with 1.7-m height were set surrounding the plots at application time.
At 76 DAE, one day before harvesting, ten plants were collected by plot (third line within useful area), identified and taken to the laboratory. Then, evaluations were performed on production traits as number of pods per plant (NPP), number of grains per pod (NGP) and 100-grain weight (100GW). Plant height (PH), stem diameter (SD), first pod height (FPH) and pod length (PL) were also measured.
AT 77 DAE, we harvested the two planting lines (first and second line within useful area) of each plot; the plants were manually removed, sun-dried and weighed to obtain dry mass (kg.ha1); then weight was corrected by assuming 13% moisture, to determine yield (Y). Data were submitted to normality test (Lilliefors) and variance homogeneity (Barlett). For that, the software SAEG 9.1 was used (Ribeiro Junior, 2001). Subsequently, variance analyses were carried out to determine macro, micronutrients and biostimulant effects. Means comparison was performed by “F” and Scott Knott tests at 5% probability through Sisvar 5.3 software (Ferreira, 2010).
There were no effect of the factors at the level of the 5% of probability for the test F, for the variables: (H), (FPH), (NPP), (PL) and (NG). It was observed a significant effect for macronutrient on SD and means are shown in Table 2.
NPK use increased in 5.39% SD, which is an important trait for beans since the smaller the diameter, the greater are crop-lodging chances (Bezerra et al, 2012). In agreement with Leal and Prado (2008), NPK promotes SD thickening; these authors studied bean nutritional disorders by deficiency of macronutrients, bore, and zinc, they observed that lack of each nutrient (N, P, K) resulted in SD decreasing. Rodolfo Junior et al. (2008) found similar result for passion fruit, who observed an increase in SD when NPK was applied.
Rodrigues (2010) combined NPK doses with Trichorderma spp. in common beans and observed a quadratic effect of NPK rates, obtaining the larger diameter at 25% of the recommended dose. This author reported that dose from 25 to 75% decreased SD, and from 75 to 125% diameter growth was retaken.
We observed a significant effect of NPK application on 100GW. Table 3 presents means related to that variable. NPK fertilization promoted 6.06% increment. As stated by Ramalho and Abreu (2006), consumer market preferences are involving medium-size ‘Carioca-like’ grain, whose size correspond to 23 to 25 g per 100 grains, and both treatments had values close to those indicated by the authors. Despite being a qualitative heritage trait, 100-grain weight is little influenced by environment and controlled by some few genes (Zilio et al., 2011); our data confirm that NPK fertilizations may interfere positively with this parameter. Some researches under macronutrient availability, mainly nitrogen, interfere in 100GW, since N has great influence during grain filling stage, when large amount of nutrients are translocated into grains. By the time N availability is low, during that stage, old leaves might rapidly drop and photosynthesis rate also decreases, which interfere negatively in grain filling (Soratto et al., 2011; Teixeira et al., 2005).

Pereira Jr (2009), who studied Caupi beans, observed a significant effect on 100GW at 1% probability for N and P2O5 applied doses. The same authors checked a variation from 24.2 g (control) to 29.7 g (75 kg ha-1 N + 25 kg ha-1 P2O5).
Carvalho et al. (2010),studying beans intercropped with coffee, established that NPK use with irrigation enhanced linearly 100GW values of common beans; however, the NPK dose increase in non-irrigated plants promoted decrease of this variable.
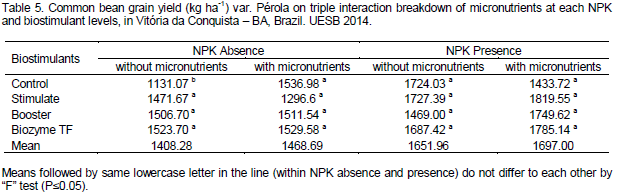
Regarding seed densities, Souza et al. (2008) tested NPK levels and liming effects on beans under conventional cropping; they found no differences on 100-grain weight among NPK levels. Significant effect of NPK factor and triple interaction of NPK* Micronutrients *Biostimulants was noted. Table 4 present statistical breakdown means of biostimulants at each NPK and micronutrient levels. Biostimulant treatments in NPK and micronutrient absence produced more than Control, highlighting the Biozyme TF that promoted a yield of 1,523.7 Kg ha-1, that is, 34.71 % superior to control yield. In treatments without NPK but with micronutrients, biostimulants did not promote significant yield gain in relation to control. On the contrary, with NPK and without micronutrients, biostimulants demonstrated means higher than Control, highlighting Stimulate that reached a yield of 1,819.55 Kg ha-1, that is, 26.91% higher than Control.
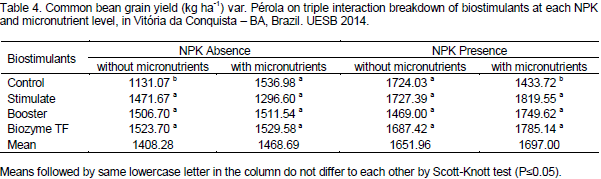
In general, biostimulants had greater yields with NPK and micronutrients, even if it was not statistically compared, and Biozyme TF detached when micronutrients were used. Plant hormones and growth regulators play an important role in several vegetal metabolism processes, including cell division, morphogenesis, elongation, compound biosynthesis and senescence (Taiz and Zeiger, 2009; Albrecht et al., 2011). Plant growth and development are controlled by a set of plant hormones, whose biosynthesis and degradation are responses of a complex interaction among physiological, metabolic and environmental factors (Dario et al., 2005). Releasing of analog growth-promoting hormones influence, condition, stimulate and maximize the performance of many crops such as common beans (Albrecht et al., 2011).
Therefore, beans under application of macro (NPK) and micronutrients with biostimulants demonstrated superior yield at 26.91% (Stimulate), 22.03 % (Booster) and 24.51% (Biozyme TF). Abrantes et al. (2011) found similar result testing Stimulate doses (0, 0.5, 1.0, 1.5 and 2.0 L ha-1) in common beans. The authors verified that the crop had an increment of 40.1% yield at 2.0 L ha-1 with application during appearance of the first flower bud. Furthermore, these authors used NPK doses near those used in our study.
Lana et al. (2009) studied two biostimulants applied at various doses and manners and concluded that their use increase yield index compared to control (no plant regulators). In contrast, Almeida et al. (2014) did not observe increases in bean yield. Avila et al. (2010) also found no yield increment when testing two biostimulant and foliar fertilizer on beans with and without irrigation. Triple interaction breakdown means of micronutrients at each NPK and biostimulant levels are shown in Table 5.
It was verified that without NPK and with micronutrient application, control had a 36.06% yield increment. This result corroborates with Martins et al. (2013), who studied N doses with and without micronutrient application in Caupi beans. These authors noted that the highest yield (784.0 kg ha-1) was obtained in micronutrient presence without N application, which was compared without micronutrient application (687.0 kg ha-1), therefore, having a 12.4% yield increase.
It is important to note that micronutrient level within the area where the experiment was conducted are medium to high (Table 1), except for Cu, whose level was considered low.
Even with or without NPK, biostimulant treated plants did not have yield increase with FTE-BR12 application, probably due to adequate amount of micronutrient found in the soil. Venegas et al. (2010), assessing single and combined application of biostimulant with micronutrient and Trichorderma spp. in the initial growth of cotton, found no differences between both types for the evaluated traits.
Lana et al. (2008), searching micronutrients in no-tillage bean cultivation, evaluated the effects of them applying isolated (zinc) or associated (cobalt and molybdenum) and the worst means were found in control and the treatment with all micronutrients (micronutrient cocktail); thus, there was no significant difference between them. With this, we may point out that it is important to balance micronutrients, once unbalanced application could act against plant development.
Table 6 shows triple interaction breakdown means of macronutrients (NPK) at each micronutrient and biostimulant levels for yield variable.
Studying the breakdown, we verified that micronutrient absence with NPK application in control increased yield in 52.42%. This outgrowth can be partly explained by soil P amount. It is possible to affirm that NPK supply, especially N and K, raised nutrient availability and absorption, allowing grain growth and mass increment, because K content attained in the soil analysis was considered low.
Surprisingly, Control had yield upper to national average, which is 1,032 kg ha-1 (IBGE, 2014), and greater than National Supply Company (CONAB) estimates for 2013/ 2014 that was 1,045. Probably, it was due to the certified seed use and proper crop handling during experiment (CONAB, 2014).
Souza et al. (2008), who studied NPK levels and liming on beans, observed linear and positive effects, and the highest NPK dose (50% above recommended dose) got 88% yield increase.
By contrast, studying NPK effect on Caupi beans, Rodrigues et al. (2004) found that control treatment generated losses, NPK application at 250 kg.ha-1 and 500 kg.ha-1 rates promoted 35 and 32% profit on invested capital. Moreover, when micronutrients were used, NPK application increased plant yield at 40.32% together with Stimulate; what may infer that for this biostimulant best performance, it is necessary on adequate plant nutrition. Nevertheless, NPK presence did not raise significantly yield of plants treated with Booster and Biozyme TF in micronutrient presence or absence.
NPK promoted wider stem diameters and heavier grains. Biostimulants enhanced common bean yield with or without macro and micronutrients.
The authors have not declared any conflict of interest.
REFERENCES
|
Albrecht LP, Braccini AL, Scapim CA, Ávila MR, Albrecht AJP, Ricci TT (2011). Manejo de biorregulador nos componentes de produção e desempenho das plantas de soja. Biosci. J. 27(6):865-876. |
|
|
Abrantes FL, Sá ME, Souza LCD, Silva MP, Simidu HM, Andreotti M, Buzetti S, Valério Filho WV, Arruda N (2011). Uso de regulador de crescimento em cultivares de feijão de inverno. Pesq. Agropec. Trop. 4(2):148-154.
Crossref |
|
|
|
Alleoni F, Bosqueiro M, Rossi M (2000). Efeito dos reguladores vegetais de Stimulate no desenvolvimento e produtividade do feijoeiro (Phaseolus vulgaris). Ciências Exatas e da Terra, Ciências Agrárias e Engenharias. 6(1):23-35. |
|
|
|
Almeida AQ (2011). Aplicação de bioestimulante e sua relação com a adubação nitrogenada nas culturas do feijão e trigo. Botucatu: São Paulo: UNESP. Tese de Doutorado em Agronomia. P. 207. |
|
|
Almeida AQ, Soratto RP, Broetto F, Cataneo AC (2014). Nodulação, aspectos bioquímicos, crescimento e produtividade do feijoeiro em função da aplicação de bioestimulante. Semina: Ciências Agrárias. 35(1):77-88.
Crossref |
|
|
Ataíde EM, Ruggiero C, Oliveira, JC, Rodrigues JD, Barbosa JC (2006). Efeito de giberelina (GA3) e do bioestimulante 'Stimulate' na indução floral e produtividade do maracujazeiro amarelo em condições de safra normal. Revista Brasileira de Fruticultura. 28(3):343-346.
Crossref |
|
|
Avila MR, Barizão DAO, Gomes EP, Fedri G, Albrecht LP (2010). Cultivo de feijoeiro no outono/inverno associado à aplicação de bioestimulante e adubo foliar na presença e ausência de irrigação. Scientia Agrarian. 11:221-230.
Crossref |
|
|
Baldo R, Scalon SPQ, Rosa YBCJ, Mussury RM, Betoni R, Barreto WS (2009). Comportamento do algodoeiro cultivar delta opal sob estresse hídrico com e sem aplicação de bioestimulante. Ciênc. Agrotecnol. 33: 1804-1812.
Crossref |
|
|
|
Barbosa FR, Gonzaga ACO (2012). Informações técnicas para o cultivo do feijoeiro-comum na Região Central-Brasileira: 2012-2014. Embrapa Arroz e Feijão. P. 247. |
|
|
Bertolin DC, Sá ME, Arf O, Furlani Junior E, Colombo AS, Carvalho FLBM (2010). Aumento da produtividade de soja com a aplicação de bioestimulantes. Bragantia. 69:339-347.
Crossref |
|
|
|
Bernardes TG, Silveira PM, Mesquita MAM (2010). Regulador de crescimento e Trichoderma harzianum aplicados em sementes de feijoeiro cultivado em sucessão a culturas de cobertura. Pesq. Agropec. Trop. 40(4):439-446. |
|
|
Bezerra AAC, Neto FA, Neves AC, Maggioni K (2012). Comportamento morfoagronômico de feijão-caupi, cv. BRS Guariba, sob diferentes densidades de plantas. Revista Ciências Agrárias. 55 (3):184-189.
Crossref |
|
|
|
Bourscheidt CE (2011). Bioestimulantes e seus efeitos na cultura da soja (Glycine max L.) Rio Grande do Sul. UNIJUI. Dissertação Mestrado em Fitotecnia. P. 61. |
|
|
Carvalho AJ, Andrade MJB, Guimarães RJ, Morais AR (2010). Sistemas de produção de feijão intercalado com cafeeiro adensado em período de formação ou após recepa. Revista Ceres. 57(3):383-392.
Crossref |
|
|
|
Conab. Companhia Nacional de Abastecimento (2014). Indicadores da Agropecuária. |
|
|
|
Costa CLL, Costa ZVB, Costa Junior CO, Andrade R, Santos JGR (2008). Utilização de bioestimulante na produção de mudas de melancia. Revista Verde. 3(3):110-115. |
|
|
|
Dario GJA, Martin TN, Dourado Neto D. Manfron PA, Bonnecarrere RAG, Crespo PEN (2005). Influência do uso de fitorreguladores no crescimento da soja. Rev. Faculdade Zootec. Vet. Agron. 12(1):63-70. |
|
|
|
Ferreira CM, Del Peloso MJ, Faria LC (2002). Feijão na economia nacional. Embrapa-CNPAF (Documentos, 135). P. 47. |
|
|
Ferreira LA, Oliveira JA, Pinho EVRV, Queiroz DL (2007). Bioestimulante e fertilizante associados ao tratamento de sementes de milho. Revista Brasileira de Sementes. 29(2):80-89.
Crossref |
|
|
|
Ferreira DF (2010). Sisvar: Sistema de análise de variância. Versão 5.3. Lavras:MG. |
|
|
|
Ganem ELO (2013). Caracterização dos sistemas de produção de milho no município de Vitória da Conquista-BA estudo de caso. Vitória da Conquista:Bahia. UESB. Dissertação Mestrado em Fitotecnia. P. 104. |
|
|
|
Ibge. Instituto Brasileiro Geografia e Estatística (2014). Indicadores de Produção Agropecuária.
View
|
|
|
|
Lana AMQ, Lana RMQ, Gozuen CF, Bonnoto I, Trevisan LR (2009). Aplicação de reguladores de crescimento na cultura do feijoeiro. Biosc. Journal, 25(1):13-20. |
|
|
|
Lana RMQ, Pereira RP, Lana AMQ, Faria MV (2008). Utilização de micronutrientes na cultura do feijoeiro cultivado no sistema plantio direto. Biosci. J. 24(4):58-63. |
|
|
|
Leal RM, Prado RM (2008). Desordens nutricionais no feijoeiro por deficiência de macronutrientes, boro e zinco. Revista Brasileira de Ciências Agrárias. 3(4):301-306. |
|
|
Leão PCS, Silva DJ, Silva EEG (2005). Efeito do ácido giberélico, do bioestimulante crop set e do anelamento na produção e na qualidade da uva 'Thompson Seedless' no Vale do São Francisco. Rev. Bras. Frutic. 27(3):379-382.
Crossref |
|
|
|
Lima MM, Azevedo CA, Beltrão NE, Lima VLA, Nascimento MBH, Figueredo ICM (2006). Níveis de adubação nitrogenada e bioestimulante na produção e qualidade do algodão BRS verde. Rev. Bras. Engenharia Agríc. Ambiental. 10(03): 619–623. |
|
|
|
Mapa. Ministério da Agricultura, Pecuária e Abastecimento (2014). |
|
|
|
Martins RNL, Nóbrega RSA, Silva AFT, Nóbrega JCA, Amaral FHC, Costa EM, Lustosa Filho JF, Martins LV (2013). Nitrogênio e micronutrientes na produção de grãos de feijão-caupi inoculado. Semina: Ciênc. Agrárias 34(4):1577-1586. |
|
|
|
Miguel FB, Silva JAA, Barbaro IM, Esperancini MST, Ticelli M, Costa AGF (2009). Viabilidade econômica na utilização de um regulador vegetal em cana-planta. Informações Econômicas. 39(1):53-59. |
|
|
|
Oliveira FA, Medeiros JF, Oliveira MKT, Souza AAT, Ferreira JÁ, Souza MS (2013). Interação entre salinidade e bioestimulante na cultura do feijão caupi. Revista Brasileira de Engenharia Agrícola e Ambiental, 17(5):465–471. |
|
|
|
Pereira Junior EB (2012). Adubação nitrogenada e fosfatada na cultura do feijão caupi no município de Souza-PB. Mossoro: Paraiba: UFERSA. Tese Doutorado em Fitotecnia. P. 69. |
|
|
|
Ramalho MAP, Abreu AFB. (2006). Cultivares. In: Vieira C, Paula Júnior TJ, Borém A Feijão. (2.ed). Viçosa, Minas Gerais, Brasil. pp. 415-436. |
|
|
|
Rodolfo Junior F, Cavalcante LF, Buriti ES (2008). Crescimento e produção do maracujazeiro-amarelo em solo com biofertilizantes e adubação mineral com NPK. Caatinga. 21(5):134-145. |
|
|
|
Rodrigues J (2010). Trichoderma spp. associado a níveis de adubação de NPK no patossistema Sclerotinia sclerotium feijoeiro. Santa Maria: Rio Grande do Sul. Dissertação Mestrado em Agronomia. P. 84. |
|
|
|
Rodrigues JEF, Alves RNB, Teixeira RNG, Rosa ES (2004). Adubação NPK na cultura do feijão-caupi em agricultura familiar, no município de Ponta de Pedras – PA. Embrapa Amazônia Oriental (Comunicado Técnico 95) P. 4. |
|
|
|
Ribeiro Junior (2001). Análises estatísticas no SAEG (Sistema para análises estatísticas). Universidade Federal de Viçosa. P. 301. |
|
|
Soratto RP, Fernandes AM, Souza EFC, Souza-Schlick GD (2011). Produtividade e qualidade dos grãos de feijão em função da aplicação de nitrogênio em cobertura e via foliar. Revista Brasileira de Ciências do Solo. 35: 2019-2028.
Crossref |
|
|
|
Souza AB, Andrade MJB, Vieira NMB, Albuquerque A (2008). Densidades de semeadura e níveis de NPK e calagem na produção do feijoeiro sob plantio convencional. Pesquisa Agropecuária Tropical 38(2):39-43. |
|
|
|
Stoller do Brasil (1998). Stimulate Mo em hortaliças. Informativo Técnico, Stoller do Brasil, Divisão Arbore. |
|
|
Silva TTA, Pinho EVRV, Cardoso DL, Ferreira CA, Alvim PO, Costa AAF (2008). Qualidade fisiológica de sementes de milho na presença de bioestimulantes. Ciência e agrotecnologia. 32(3):840-846.
Crossref |
|
|
|
Taiz L, Zeiger, E (2013). Fisiologia vegetal. (5 th ed) P. 954. |
|
|
Teixeira CM, Carvalho GJ, Andrade MJB, Furtini Neto AE, Marques ELS (2005). Palhadas e doses de nitrogênio no plantio direto do feijoeiro. Acta Scientia Agronomica. 27:499-505.
Crossref |
|
|
|
Venegas F, Tomasele R, Farias LN (2010). Efeito de diferentes produtos para tratamento de sementes no desenvolvimento inicial do algodoeiro (Gossypium hirsutum). Ensaio e Ciênc.: Biologicas, Agrarias e da Saude. 14(1):41-50. |
|
|
|
Vieira C, Paula Júnior TJ, Borém A (2006). Feijão:Aspectos gerais e cultura no Estado de Minas. Universidade Federal de Viçosa. Vicosa, Minas Gerais, Brasil. (2 th ed.) P. 600. |
|
|
|
Vieira EI, Nascimento EJ, Paz JG (1998). Levantamento ultra detalhado de solos do campus da UESB em Vitória da Conquista – BA. Boletim técnico do Departamento de Engenharia Agrícola e Solos. P. 37. |
|
|
Zilio M, Cileide Coelho CMM, Souza CA, Santos JCP, Miquelluti DJ (2011). Contribuição dos componentes de rendimento na produtividadede genótipos crioulos de feijão (Phaseolus vulgaris L.). Revista CiênciaAgronômica. 42(2):429-438.
Crossref |
|
|
|
|