ABSTRACT
Sunflower is an important oil crop in India. The present study aims to find out the response of biofertilizers and homo-brassinolide on growth, yield and oil content of sunflower (Helianthus annuus L.). The results showed that inoculation of biofertilizers significantly affected plant height, total chlorophyll content and also significantly increased yield attributes viz., thalamus diameter, weight of thalamus, filled seeds capitulum-1 and 1000 seed weight as well as seed yield, biological yield and oil content. The combined inoculation of Phosphate solubilizing bacteria (PSB) + Vesicular-arbuscular mycorrhizal (VAM) + Azotobacter recorded higher seed yield (3189 and 3263 kg ha-1) over Azotobacter (1866 and 2073 kg ha-1), PSB + Azotobacter (2269 and 2421 kg ha-1) and VAM + Azotobacter inoculation (2545 and 2752 kg ha-1) respectively, during the both years. Twice sprayings of homo-brassinolide at budding + flowering stages significantly recorded higher value of plant height, total chlorophyll content and yield parameters as compared to brassinolide spraying at budding stage alone. The maximum seed yield (2760 and 2917 kg ha-1) was obtained from crop receiving both spraying of homo-brassinolide at budding + flowering stages over only one spraying at budding stage (2171 and 2337 kg ha-1) respectively, during 2010-2011 and 2011-2012.
Key words: Azotobacter, brassinolide, phosphate solubilizing bacteria (PSB), Sunflower, vesicular-arbuscular mycorrhizal (VAM).
Sunflower (Helianthus annuus L.) is one of the most important oilseed crops containing high quality edible oil. Sunflower kernel has 50% oil content, of which 30% is the essential fatty linoleic acid (Oraki et al., 2011). It is easily cultivated and grown in different conditions and soils (Kaya and Kolsarici, 2011; Lopez-Valdez et al., 2011). Sunflower oil has excellent nutritional properties (Seiler, 2007). Sunflower is one of the fastest growing oilseed crops in India popularly known as "Surajmukhi." Majority of the present day varieties grown all over the world is originated from former USSR. In India, sunflower as an oilseed crop was introduced in 1969. Prior to which it was used mainly as an ornamental plant. Sunflower oil is popular as healthy cooking oil due to its health benefits, while the meal is used in animal feed industry and also being used in the manufacture of soap and vanishes (Lawal et al., 2011). Among sunflower products, meal is the most traded in world market.
In view of the environmental and health problems arising from chemical fertilizers usage, many attentions have been drawn to the application of biological fertilizers in agriculture. Use of biofertilizers is one of the important components of integrated nutrient management, as they are cost effective and renewable source of plant nutrients to supplement the chemical fertilizers for sustainable agriculture. Several microorganisms and their association with crop plants are being exploited in the production of biofertilizers. Azotobacter has been used as biofertilizer because of their positive effects on growth and productivity of plants via several mechanisms including plant hormones production, nitrogen (N2) fixation, antagonism against phytopathogenic microorganisms and solubilization of nutrients (Rokhzadi and Toashih, 2011).
Phosphorus (P) is one of the most essential plant nutrients for plant growth after nitrogen and required for higher and sustained productivity of oil from sunflower. However, the availability of this nutrient for plants is limited by different chemical reactions especially in tropical and subtropical soils (Mehrvarz et al., 2008). Phosphorus plays a significant role in several physiological and biochemical plant activities like photosynthesis, transformation of sugar to starch and transporting of the genetic characters because phosphorus is a vital component of the substances that are building blocks of genes and chromosomes. Sharma (2002) reported that one of the advantages of feeding the plants with phosphorus is to produce deeper and more profuse roots. Malakooti (2000) reported that phosphorus had an important role in early ripening, decreasing grain moisture, improving crop quality. Arpana et al. (2002) reported that a large proportion of phosphorus in chemical fertilizer becomes unavailable to the crop plants after its application in the soil due to its rapid fixation in soil. Its influence on seed yield, oil yield and oil quality has been well established by Loubser and Human (1993), Bahl and Toor (1999), Chandrashekara et al. (1995) and Zubillaga et al. (2002); also, application of phosphorus has become an essential part of sunflower fertilizer program.
In general, phosphorus is added to soil as inorganic phosphates, because the free inorganic P in soil solution plays a central role in P-cycling and plant nutrition (Peix et al., 2001). However, a large portion of soluble inorganic phosphate applied to soil as chemical fertilizer is immobilized rapidly after application due to phosphate fixation by aluminum, calcium, iron, magnesium and soil colloids (Rodriguez and Fraga, 1999) and becomes unavailable to plants (Singh and Kapoor, 1994). Therefore, P is often a limiting nutrient in agricultural soils. Micro-organisms are also involved in a range of process that affect the transformation of soil P and thus an integral part of the soil P cycle (Chen et al., 2006).
In particular, P-solubilizing micro-organisms (bacteria or fungi) are able to solubilize unavailable soil P and increase the yield of crops (Adesemoye and Kloepper, 2009). Plant growth-promoting rhizobacteria (PGPR) and rhizosphere bacteria are free-living soil organisms that can benefit plant growth by different mechanisms (Glick, 1995). P-solubilization ability of micro-organisms is considered to be one of the most important traits associated with plant P nutrition (Chen et al., 2006). Several bacterial species, in association with plant rhizosphere are capable of increasing availability of Phosphorus to plants either by mineralization of organic phosphate or by solubilization of inorganic phosphate by production of acids (Rodriguez and Fraga, 1999).
Vesicular-arbuscular mycorrhizal (VAM) fungi are normally known to benefit plant health, with the net benefit to plant increasing as stress increases due to lack of nutrient and soil moisture (Bethlenfalvay and Svejcar, 1991; Sieverding, 1991). VAM fungi contribute greatly to phosphorus uptake (George et al., 1992; Smith et al., 1992) and to nitrogen uptake directly (Azcon-Aguilar et al., 1993). Regardless of the actual mechanism involved, VAM fungi can increase the efficiency of phosphorus and nitrogen removal from the soil solution over that of roots alone, which has obvious implications for reducing fertilizer inputs and leaching. VAM fungi have also been shown to be important in the uptake of other ions, including K, S, Mg, Fe, Z, Cu (Cooper, 1984; Sieverding, 1991). Brassinosteroids (BRs) are naturally occurring steroidal plant hormones and their various forms, widely distributed in the plant kingdom, have a distinct role in stem elongation, pollen tube growth, leaf bending, ethylene biosynthesis, and xylem differentiation (Khripach et al., 2002). The standing plants supplemented with BRs improved the productivity potential of economically important crops by activating cell elongation, vascular differentiation, and/or proton pump (Rao et al., 2002). Plants supplemented with BRs exhibited an increase in the activities of carbonic anhydrase and nitrate reductase (Hayat et al., 2001; Hayat et al., 2001a), phosphoenolpyruvate carboxylase, ribulose-1,5-bisphosphate carboxylase/oxygenase (Rubisco), and the contents of soluble proteins (Braun and Wild, 1984; Braun and Wild, 1984a; Yang et al., 1992). BRs treated plants also exhibit higher resistance to stress and produce more seeds in crop plants (Rao et al., 2002). Brassinosteroids (BRs) are regarded essential for the normal growth and development of plants (Li and Chory, 1999). Hence, an experiment was conducted to study the response of biofertilizers and homo-brassinolide phytohormone on growth, yield and oil content of sunflower (H. annuus. L).
A field experiment was conducted during the winter season (October to March) of 2010-2011 and 2011-2012 at farmers field adjacent to the farm of the Institute of Agriculture (Palli Siksha Bhavana), Visva-Bharati, Sriniketan, West Bengal. The place is situated at 23°39’ N latitude, 87°42’ E longitude and an elevation of 58.9 m above mean sea level. The experiment was established in sandy loam soil with pH 5.7, low in available nitrogen (130 kg ha-1), phosphate (12.50 kg ha-1) and medium in potassium (163.5 kg ha-1). Seed bed preparation included ploughing, disk harrowing and cultivation. Sowing was performed manually.
The experiment was laid out in factorial randomized block design with four types of biofertilizers inoculation (Azotobacter, Phosphate solubilizing bacteria (PSB) + Azotobacter, Vesicular arbuscular mycorrhizae (VAM) + Azotobacter and Phosphate solubilizing bacteria (PSB) + Vesicular arbuscular mycorrhizae (VAM)) + Azotobacter and two spraying of homo-brassinolide (HBR) @ 0.5 ml L-1 of water at 50% budding stage and 50% budding + 50% flowering stages. In all, eight treatments were replicated three times. Each plot consisted of ten rows, and each row was 5 m long. The first and last rows in each plot were considered as marginal effects. The seed was inoculated with Azotobacter and PSB by slurry method whereas the soil was inoculated with VAM inoculums (Manufactured by Symbiotic Sciences, New Delhi). The VAM inoculums were placed at the seeding depth of the soil and then pre-inoculated seeds were sown according to the treatment. Homo-brassinolide (DOUBLE) was used according to the treatment. Plots were fertilized with the same amount of fertilizer as 80 kg ha-1N, 100 kg ha-1 P2O5 and100 kg ha-1 K2O but ½th nitrogen fertilizer was added before sowing as basal dose and rest amount was added before bud formation.
In the study, “PAC 36” commercial hybrid of oilseed sunflower, which had early to medium maturation, high yield potential, responsive to higher inputs, more tolerant to diseases and pests, higher drought tolerance, more self fertile, superior in their seed filling ability and higher adaptation ability, was used as plant material. Seeds were sown in rows 45 cm apart and plant to plant distance 20 cm, using 5 kg seed ha-1.
The sunflower plants were harvested by hand, from middle seven rows excluding side rows and 1 m from each end of plots at the stage of physiological maturation when the back of the head had turned from green to yellow and the bracts were turning brown (Anonymous, 2005) or fruit dry weight (FDW) has reached its maximum value with a water content about 38% (Rondanini et al., 2007). Plant height and head diameter were determined at physiological maturity, by harvesting 10 plants of sunflower per experimental unit. Aerial dry matter production was determined at 45, 60, 75 and 90 DAS, by cutting 5 plants at ground level from each plot kept in a hot air oven at 65°C for 48 h till constant weight was obtained. The dry weight of plants were recorded and used for determination of aerial dry matter production. Heads were separated from vegetative parts. Head diameter was estimated for distance in cm across the apical head at its widest point. The head samples for yield were also dried at 60°C for 48 h in hot air oven to get constant weight and threshed mechanically. Seed yield was adjusted to a 10% moisture basis. Filled grain and empty hulls were separated by hand. Hereafter, grain number per head refers to filled grains only. Harvest index was computed by dividing the seed yield from the total biological yield and was expressed as percentage. The harvest index (HI) was calculated by the formula given by Donald (1962).
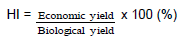
Seed oil contents were determined using the Soxhlet and Kjeldahl method in seed kernel (dehulled). Fully open fresh new leaves were clipped from plants of each plot at morning hours and immediately brought to the laboratory in a plastic bag for estimation chlorophyll content at 30, 45, 60 and 75 DAS. Total chlorophyll content was measured adopting the method of Hiscox and Israelstam (1979), using Dimethyl sulfoxide (DMSO). The chlorophyll content was determined using the formula given by Arnon (1949) and expressed as mg g-1 of fresh leaf. Arnon’s formula estimate total chlorophyll as follows.
Total Chlorophyll = [20.2(D645) + 8.02 (D663)] ×V/[1000×W],
Where, D = Absorbance, V = Final volume of DMSO (ml); W = Weight of fresh leaf (g)
Statistical Analysis
Data collected were subjected to statistical analysis of variance according to Gomez and Gomez (1984) using MSTAT computer program.
Plant height
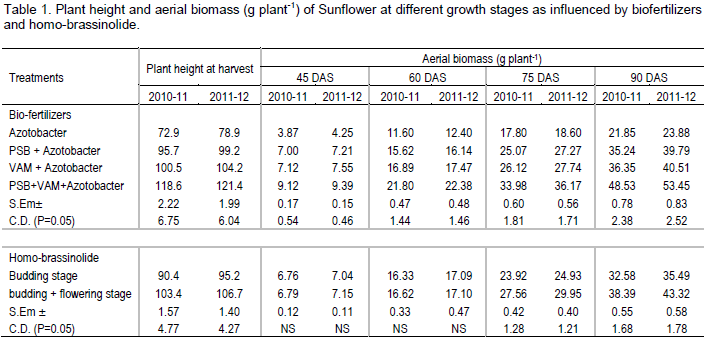
Effect of biofertilizers and homo-brassinolide on plant height at harvest stage of sunflower is presented in Table 1. The tallest plant height (118.6 and 121.4 cm) was recorded with inoculation of PSB + VAM + Azotobacter over other biofertilizers treatment during 2010-11 and 2011-12 seasons, respectively. There was no significant difference between the treatments of PSB + Azotobacter and VAM + Azotobacter in respect of plant height during both years. The dwarf plant height (72.9 and 78.9 cm) was obtained from crop receiving Azotobacter alone during both years and it was significantly lowest than other biofertilizers treatments. The tallest height might be due to the strong synergistic effect of PSB + VAM + Azotobacter. Inoculations of PSB which are known to produce growth hormones like IAA and GA (Sattar and Gaur, 1987) are likely to favor increased plant height. The results are in conformity with those of Mukherjee and Rai (2000). Twice sprayings of homo brassinolide at budding + flowering stages significantly improved plant height as compared to one spraying of homo brassinolide at budding stage. Increased plant height might be due to positive effect of homo-brassinolide on meristamatic tissues of plant as well as in increasing number and size of cell (Prakash et al., 2008). Similar result was reported by Mitchell and Gregory (1972).
Aerial biomass production
Data on crop aerial biomass production at different growth stages (45, 60, 75 and 90 DAS) are presented in Table 1. Scrutiny of records during all the observations revealed that the maximum aerial biomass production (9.39 to 53.45 g plant-1) of crop was obtained using PSB + VAM + Azotobacter as compared to other treatment or treatment combinations irrespective of the growth stages during both years. The minimum aerial biomass production (4.25 to 23.88 g plant-1) of crop was obtained from the crop receiving Azotobacter and it was significantly lower than other biofertilizers treatment. The high response of plant to the PSB + VAM + Azotobacter inoculation might be due to solubilization and mobilization of available P by the native soil microflora, or attributed due to increased PSB and VAM activity in the rhizosphere and consequently enhanced P solubilization and mobilization. Therefore, increased aerial biomass production of crop by PSB + VAM + Azotobacter might be due to better development of root systems resulting in tapping larger volume of bound soil water and nutrients especially phosphorus. This result is in conformity with the findings of Mukherjee and Rai (2000). Homo-brassinolide level exerted significant positive effect on aerial biomass production. The maximum aerial biomass production was recorded in crop receiving spraying of homo-brassinolide at budding + flowering stages at 75 and 90 DAS over one spraying of homo-brassinolide at budding stage. There was no effect of homo-brassinolide because it was applied after 60 DAS. Increased aerial biomass production of crop with homo-brassinolide might be due to higher plant growth as well as more positive effect on meristamatic tissues of plant and in increasing number and size of cell (Prakash et al., 2008). Homobrassinolide (HBR) is one of the bioactive brassinosteriods considered as plant hormone with pleiotropic effects as they influence on developmental processes such as growth (Sasse, 1999).
Total chlorophyll content
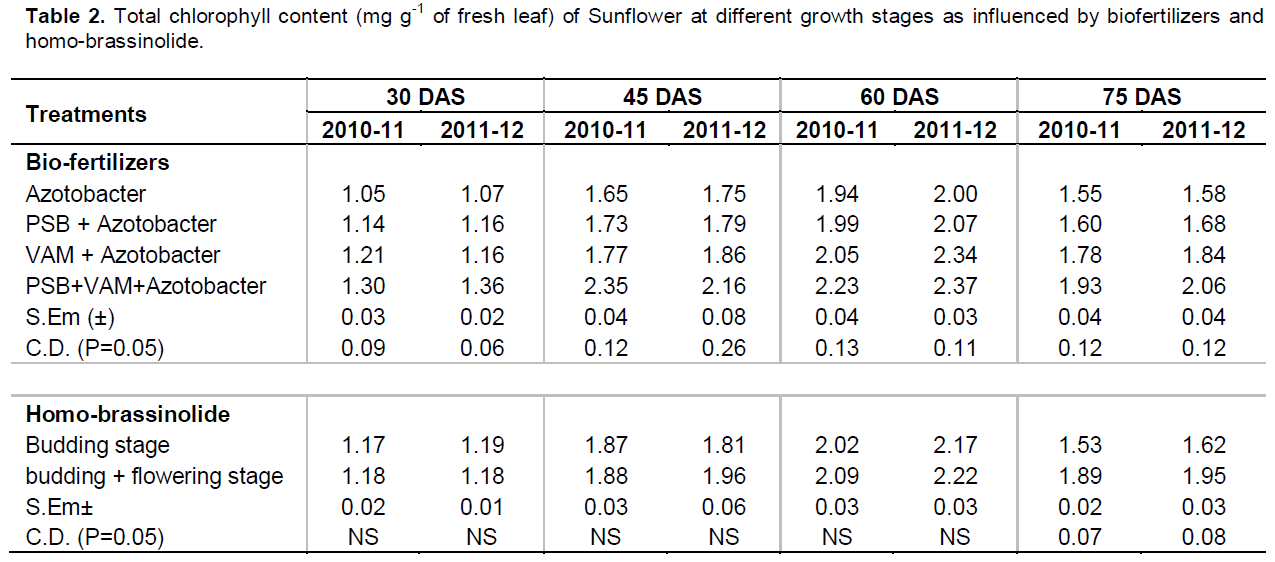
Data on total chlorophyll content in leaf as affected by application of biofertilizers and homo-brassinolide are presented in Table 2. Data also indicated that total chlorophyll content increased with advancement of the crop age up to 60 DAS and thereafter marginally decreased at 75 DAS. The decrease in the amount of chlorophyll contents suggested that the leaves were at their senescence stage. During the process of senescence, the composition of cell components of the leaves are progressively degraded (Erickson, 1968; Sinclair et al., 1971; Craig and Shih, 1998). Inoculation of crop with PSB + VAM + Azotobacter recorded significantly higher total chlorophyll content at 75 DAS. This might be due to higher content of nitrogen and magnesium which are the core component of chlorophyll (Ruiz-Lozano and Azicon, 1995). This finding is generally in agreement with the previous finding of Demir et al. (2011). Spraying of homo-brassinolide significantly influenced total chlorophyll content at 75 DAS. Twice spraying of homo-brassinolide at budding + flowering stage recorded significantly higher total chlorophyll content (1.89 and 1.95 mg g-1 of fresh leaf) as compared to one spray of budding stage (1.53 and 1.62 mg g-1 of fresh leaf). This might be due to better vigorousness of root system with consequent supply of nitrogen from soil. It has been evidenced that there is a positive correlation between nitrogen and total chlorophyll content. These results are in agreement with the findings of Ramraj et al. (1997), Vardhini and Rao (1998) and Nakashita et al. (2003).
100 seed weight
The highest 100 seed weight was obtained with inoculation of PSB + VAM+ Azotobacter (Table 3) over Azotobacter, PSB + Azotobacter and VAM + Azotobacter inoculation. The lowest test weight of 100 seed weight recorded was obtained with Azotobacter inoculation alone. These results are in agreement with the previous findings of Barea et al. (1975). Twice spraying of homo-brassinolide at budding + flowering stage recorded a significant highest 100 seed weight as compared to one spraying of homo-brassinolide at budding stage. The results are in conformity with those of Mitchell and Gregory (1972).
Diameter of thalamus
Diameter of thalamus recorded was significantly influenced by the combined inoculation of PSB + VAM + Azotobacter (Table 3) as compared to Azotobacter, PSB + Azotobacter and VAM + Azotobacter inoculation. The high response of plant to the PSB + VAM+ Azotobacter inoculation might be due to mobilization of available P by the native soil microflora, or attributed to increased phosphate solubilizing bacteria activity in the rhizosphere following PSB + VAM + Azotobacter application and consequently by enhanced P solubilization. For these reasons, it enhanced P uptake by the crops and an increased thalamus diameter ultimately leading to higher seed yields. Similar result was reported by Barea et al. (1975). Spraying of homo-brassinolide at budding + flowering stage was significantly highest in diameter of thalamus as compared to one spray at budding stage. The results are in conformity with those of Mitchell and Gregory (1972).
Seed yield
Inoculation of PSB + VAM + Azotobacter showed significant effect on seed yield (Table 3) as compared to Azotobacter, PSB + Azotobacter and VAM + Azotobacter inoculation. The result of pooled analysis showed that combined inoculation of PSB + VAM resulted in 63.78, 37.70 and 21.79% higher seed yield over Azotobacter, PSB + Azotobacter and VAM + Azotobacter inoculation, respectively. This increase in yield parameters by PSB + VAM + Azotobacter inoculation might be due to increase in phosphorus and nitrogen availability, roots, relative water content, root biomass, nodule number and dry weight that could be ascribed to a better translocation of photosynthates towards yield attributes and yield. Photosynthesis generates the high energy sugars. Increased availability of phosphorus provides the mechanism for energy storage in the form of ATP and the transfer of that energy source to fuel vital plant functions such as N fixation. Rose (1957) recorded similar findings. The phosphate solubilizing bacteria is known to produce vitamins (Baya et al., 1981) and IAA- and GA-like growth substance (Satter and Gaur, 1987). The result is in conformity with those of Jones and Sreenivas (1993). Spraying of homo-brassinolide at budding + flowering stage was significant as compared to one spraying at budding stage. The result of pooled analysis showed that two spraying of homo-brassinolide increased 25.90% higher seed yield as compared to one spraying. The increase in yield might be due to the higher photochemical efficiency in terms of hill reaction, and CO2 intake in twice spraying of homo-brassinolide may be attributed to increased accumulation of photosynthetic pigments particularly chlorophyll and higher soluble protein content. The results are in conformity with those of Nayak and Murthy (1980) and Chowdhary et al. (1994).
Stalk yield
Inoculation of PSB + VAM + Azotobacter recorded significantly higher stalk yield as compared to Azotobacter, PSB + Azotobacter and VAM + Azotobacter inoculation (Table 3). This increase in stalk yield by PSB + VAM + Azotobacter inoculation might be due to better growth of the plants. Shinde (1990) and Yadav and Shrivastava (1997) recorded similar findings. Twice spraying of homo-brassinolide at budding + flowering stage produced significantly higher stalk yield over one spraying of homo-brassinolide at budding stage. The result is in conformity with those of Meudt et al. (1983).
Harvest index
The maximum harvest index was recorded with the combined inoculation of PSB + VAM + Azotobacter (Table 3) followed by Azotobacter + VAM, PSB + VAM and Azotobacter. The minimum harvest index was obtained from the crop receiving only Azotobacter, PSB + Azotobacter and VAM + Azotobacter inoculation. The higher harvest index might be due to better yield with PSB + VAM + Azotobacter. Similar type of result was previously reported by Barea et al. (1975). Spraying of homo-brassinolide at budding + flowering stage showed significant highest harvest index as compared to one spray at budding stage. The results are in conformity with those of Mitchell and Gregory (1972).
Oil content (%)
Combined inoculation of PSB + VAM + Azotobacter recorded significantly higher oil content over Azotobacter + VAM, PSB + VAM and Azotobacter. Our results were similar to the findings of Patra et al. (2013). The spraying of homo-brassinolide at budding + flowering stage recorded maximum oil content whereas Azotobacter recorded minimum oil content in seed The increase in oil content might be due to spraying of homo-brassinolide which was in consonance with the findings of Mai et al. (1989) and Prakash et al. (2008) in rice and sesame respectively. The increase in oil content might be due to increase in morphological, physiological and biochemical parameters (Prakash et al., 2008).
Correlation studies
Looking at the correlation coefficients in Table 4, a significant positive correlation was noted between seed yield and test weight during both years of experiment. There was also a positive relationship between seed yield and chlorophyll content. Sharafai et al. (2006) reported positive and significant relation between grain yield and 100-grain weight in maize. Thus, correlation studies showed that seed yield was positively and significantly correlated with test weight and total chlorophyll content at different stages of crop growth during both years of experiment.
Relationship between yield parameter and seed yield
Head diameter, head weight and number of filled seed head-1 showed significant positive association with seed yield during both years (Figures 1 and 2). The increase in seed yield with increasing head diameter, head weight and number of filled seed head-1 was linear in fashion. Head diameter accounted for 75% variability in seed yield during 2010-11 and 86% variability in seed yield during 2011-12. Similarly, head weight accounted for 76% variability in seed yield during 2010-11 and 85% variability in seed yield during 2011-12. On the other hand, number of filled seed head-1 accounted for 85% variability in seed yield during 2010-11 and 84% variability in seed yield during 2011-12. Hence, increasing head diameter, head weight and number of filled seed head-1 has direct effect on increasing the seed yield of sunflower.
The maximum plant height, aerial biomass production, chlorophyll content, 100 seed weight, diameter of thalamus, seed yield, stalk yield, harvest index and oil content were obtained with inoculation of PSB + VAM + Azotobacter. On the other hand, twice spraying of homo-brassinolide at 50% budding initiation + 50% flowering stages also recorded maximum aerial biomass production, chlorophyll content, 100 seed weight, diameter of thalamus, seed yield, stalk yield, harvest index and oil. The result indicated the importance of adopting suitable combination of biofertilizers and spraying of homo-brassinolide in this experiment lateritic soil. Thus, the appropriate combination of biofertilizers and homo-brassinolide can increase seed yield and improve oil content in sunflower.
The author(s) have not declared any conflict of interest.
REFERENCES
Adesemoye AO, Kloepper JW (2009). Plant-microbes interactions in enhanced fertilizer-use efficiency. Appl. Microbiol. Biotechnol. 85:1-12.
Crossref |
|
|
|
Anonymous (2005). Sunflower Magazine: Harvest Fundamentals.
View
|
|
|
Arnon DI (1949). Copper enzyme in isolated chloroplasts polyphenol oxidase in Beta vulgaris. Plant Physiol. 24:1-15.
Crossref |
|
|
|
Arpana N, Kumar SD, Prasad TN (2002). Effect of seed inoculation, fertility and irrigation on uptake of major nutrients and soil fertility status after harvest of late sown lentil. J. Appl. Biol. 12:23–26. |
|
|
|
Azcon-Aguilar C, Alba C, Montilla M, Barea JM (1993). Isotopic (15N) evidence of the use of less available N forms by VA mycorrhizas. Symbiosis 15:39-48. |
|
|
Bahl GS, Toor GS (1999). Efficiency of P utilization by sunflower grown on residual P fertility. Bioresour. Technol. 67:97-100.
Crossref |
|
|
|
|
|
Barea JM, Azcon R, Hayman DS (1975). Possible synergistic interaction between Endogene and phosphate solubilizing bacteria in low phosphorus soils. In Endomycorrhizas Eds. F.E. Sanders, B. Mosse and P.B. Tinker. pp. 409-418. Academic press. London. New York. |
|
|
Baya AM, Boethling RS, Ramos CA (1981). Vitamin production in relation to phosphate solubilization by soil bacteria. Soil Biol. Biochem. 13:527-531.
Crossref |
|
|
|
Bethlenfalvay GJ, Svejcar TE (1991). Mycorrhizae in plant productivity and soil conservation. In: Proceedings of the IV th International Rangelend Congress, Montpellier, France. |
|
|
Braun P, Wild A (1984). The influence of brassinosteroid on growth and parameters of photosynthesis of wheat and mustard plants. J. Plant Physiol. 116:189-196.
Crossref |
|
|
|
|
|
Braun P, Wild A (1984a). The influence of brassinosteroid on growth promoting steroidal lactone, on development and CO2-fixation capacity of intact wheat and mustard seedlings In: Sybesma C (ed) Advances in photosynthesis research Vol III, Martinus Nijhoff/ Dr. W Junk Publishers. Hague- Boston-Lancaster pp. 461-464. |
|
|
Chandrashekara CP, Patil VC, Sreenivasa MN (1995). VA-mycorrhiza mediated P effect on growth and yield of sunflower (Helianthus annuus L.) at different P levels. Plant Soil 176:325-328.
Crossref |
|
|
Chen YP, Rekha PD, Arun AB, Shen FT, Lai WA, Young CC (2006). Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl. Soil Ecol. 34:33-41.
Crossref |
|
|
|
|
Chowdhary PK, Thangaraj M, Jayapragasam M (1994). Biochemical changes in low-irradiance tolerant and susceptible rice cultivars. Biol.Plantarum 36(2):237-242.
Crossref |
|
|
|
|
|
Cooper KM (1984). Physiology of VA mycorrhizal associations. In:VA Mycorrhiza, Powell, C.L. and Bagyaraj, D.J., Eds. CRC Press Boca Raton, FL. pp. 155-188. |
|
|
|
Craig JC, Shih SF (1998). The spectral response of stress conditions in citrus trees: development of methodology. Soil and Crop Science Society of Florida. 57:16-20. |
|
|
|
Demir K, Başak H, Okay FY, Kasım R (2011). The effect of endo-mycorrhiza (VAM) treatment on growth of tomato seedling grown under saline conditions. Afr. J. Agric. Res. 6(14):3326-3332. |
|
|
|
Donald CM (1962). In search of high yields. J. Aust. Inst. Agric. Sci. 28:171-178. |
|
|
|
Erickson LC (1968). The general physiology of citrus. The Citrus Industry, Anatomy, Physiology, Genetics, and Reproduction, (Eds.): W. Reuhter, L.D. Batchelor and H.J. Webber. 2:86-122. |
|
|
George E, Haussler K, Vetterlein D, Gorgus E, Marschner H (1992). Waternand nutrient translocation by hyphae of Glomus mosseae. Can. J. Bot. 70:2130-2137.
Crossref |
|
|
Glick BR (1995). The enhancement of plant growth by free-living bacteria. Can. J. Microbiol. 41:109-117.
Crossref |
|
|
|
|
|
Gomez KA, Gomez AA (1984). Statistical procedure for Agricultural Research 2nd Edition, John Wiley and Sons. |
|
|
Hayat S, Ahmad A, Hussain A (2001). Growth of wheat seedlings raised from the grains treated with 28-homobrassinolide. Act Physiol. Plant 23:27-30.
Crossref |
|
|
Hayat S, Ahmad A, Mobin M (2001a). Carbonic anhydrase, photosynthesis, and seed yield in mustard plants treated with phytohormones. Photosynthetica 39:111-114.
Crossref |
|
|
|
|
Hiscox JA, Israelstam GE (1979). A method for the extraction of chlorophyll leaf tissue without mecreation. Can. J. Bot. 57(2):1332-1334.
Crossref |
|
|
|
Jones NP, Sreenivas MN (1993). Effect of inoculation of VA mycorrhiza and phosphate solubilizing bacteria on rhizosphere microflora of sunflower. II. AzotobacterAzotobacter and phosphate solubilizing bacteria. J. Ecotoxicol. Environ. Monit. 3(1):55-58. |
|
|
|
Kaya MD, Kolsarici O (2011). Seed yield and oil content of some sunflower (Helianthus annuus L.) hybrids irrigated at different growth stages. Afr. J. Biotechnol. 10(22):4591-4595. |
|
|
Khripach V, Zhabhinskii V, de Groot A (2002). Twenty years of brassinosteroids: steroidal plant hormones warrant better crops for the XX1 century. Ann. Bot. 86:441-447.
Crossref |
|
|
|
|
|
Lawal BA, Obigbesan GO, Akanbi WB, Kolawole GO (2011). Effect of planting time on sunflower (Helianthus annuus L.) productivity in Ibadan, Nigeria. Afr. J. Agric. Res. 6(13):3049-3054. |
|
|
Li JM, Chory J (1999). Brassinosteroids action in plants. J. Exp. Bot. 50:275-282.
Crossref |
|
|
Lopez-Valdez F, Fernandez-Luqueno F, Ceballos-Ramirez JM, Marsch R, Olalde- Portugal V, Dendooven L (2011). A strain of Bacillus subtilis stimulates sunflower (Helianthus annuus L.) growth temporarily. Sci. Horticult. 128:499-505.
Crossref |
|
|
|
|
Loubser HL, Human JJ (1993). The effect of nitrogen and phosphorus fertilization on the phosphorus absorption by sunflowers. J. Agron. Crop Sci. 171(3):206-215.
Crossref |
|
|
|
Mai YY, Lin JM, Zeng XL, Pan RZ (1989). Effects of brassinolide on the activity of nitrate reductase in rice seedlings. Plant Physiol. Commun. 2:50-52. |
|
|
|
Malakooti MJ (2000). Sustainable agriculture and yield increment by optimum fertilizer utilization in Iran",2nd edition. Agricultural Extension Publications, Iran. |
|
|
|
Mehrvarz S, Chaichi MR, Alikhani HA (2008). Effects of phosphate solubilizing microorganisms and phosphorus chemical fertilizer on yield and yield components of barley (Hordeum vulgare L.). Am. Eur. J. Agric. Environ. Sci. 3(6):822–828. |
|
|
|
Meudt WJ, Thompson MJ, Bennett HW (1983). Investigations on the mechanism of brassinosteroid response. III. Techniques for potential enhancement of crop production. In: Proceedings of the 10th Annual Meeting of the Plant Growth Regulators Society of America. Madison, USA. pp. 312-318. |
|
|
|
Mitchell JW, Gregory LE (1972). Enhancement of overall growth, a new response to brassins. Nature 239:253-254. |
|
|
|
Mukherjee PK, Rai RK (2000). Effect of Vesicular arbuscular mycorrhizae and phosphate solubilizing bacteria on growth, yield and phosphorus uptake by wheat (Triticum aestivum) and chick pea (Cicer arietinum). Indian J. Agron. 45(3):602-607. |
|
|
Nakashita H, Yasuda M, Nitta T, Asami T (2003). Brassinosteroid functions in a broad range of disease resistance in tobacco and rice. Plant J. 33(5):887-898.
Crossref |
|
|
|
Nayak SK, Murthy KS (1980). Effect of varying light intensities on growth parameters in rice. Indian J. Plant Physiol. 23:309-316. |
|
|
|
Oraki H, Alahdadi I, Khajani FP (2011). Sunflower (Helianthus annuus L.) hybrids seeds distribution modelling: Normal, lognormal and weibull models. Afr. J. Agric. Res. 6(2):618-623 |
|
|
|
Patra P, Pat BK, Ghosh GK, Mura SS, Saha A (2013). Effect of Biofertilizers and Sulphur on Growth, Yield and Oil content of Hybrid Sunflower (Helianthus annuus L.) in a Typical Lateritic Soil. 2:603. |
|
|
Peix A, Mateos PF, Barrueco CR, Molina EM Velazquez E (2001). Growth promotion of common bean (Phaseolus vulgaris L.) by a strain of Burkholderia cepacia under growth chamber conditions. Soil Biol. Biochem. 33:1927-1935.
Crossref |
|
|
|
Prakash M, Suganthi S, Gokulakrishnan J, Sabesan T (2008). Effect of Homobrassinolide on Growth, Physiology and Biochemical Aspects of Sesame. Karnataka J. Agric. Sci. 20(1):110-112. |
|
|
Ramraj VM, Vyas BN, Godrej NB, Mistry KB, Swami BN, Singh N (1997). Effects of 28-homobrassinolide on yields of wheat, rice, groundnut, mustard, potato and cotton. J. Agric. Sci. 128:405–413.
Crossref |
|
|
|
|
|
Rao SSR, Vardhini BV, Sujatha E (2002). Brassinosteroids- a new class of plant phytohormones. Curr Sci. 82:1239-1244. |
|
|
Rodriguez H, Fraga R (1999). Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol. Adv. 17:319-339.
Crossref |
|
|
|
|
|
Rokhzadi A, Toashih V (2011). Nutrient uptake and yield of chickpea (Cicer arietinum L.) inoculated with plant growth-promoting rhizobacteria. Aust. J. Crop Sci. 5(1):44-48. |
|
|
Rondanini DP, Savin R, Hall AJ (2007). Estimation of physiological maturity in sunflower as a function of achene water concentration. Eur. J. Agron. 26:295–309.
Crossref |
|
|
|
Rose RE (1957). Techniques of determining the effect of micro-organisms on insoluble inorganic phosphates. New Zealand Journal of Science and Technology. 38:773-780. |
|
|
Ruiz-Lozano JM, Azcon R (1995). Hyphal contribution to water uptake in mycorrhizal plants as affected by the fungal species and water status. Plant Physiol. 95:472-478.
Crossref |
|
|
|
Sasse JM (1999). Physiological actions of brassinosteroids. In: Brassinosteroids – Steroidal Plant Hormones, eds. Sakurai, A. Yokota, T. and Clouse, S. D., Springer, Tokyo, pp. 137-161. |
|
|
|
Sattar MA, Gaur AC (1987). Production of auxins and gibbereline by phosphate dissolving microorganisms. Zentralbl. Mikrobiol. 142:393-396. |
|
|
Seiler GJ (2007). Wild annual Helianthus anomalus and H. deserticola for improving oil content and quality in sunflower. Ind. Crops Prod. 25:95–100.
Crossref |
|
|
|
Sharafai AI, Mahmud M, Lawal AB, Abubakar IU, Mohammad SG (2006). Correlation and path coefficient analysis for growth, yield and yield components of early maturing maize (Zea mays L). Crop Res. 21:255-259. |
|
|
|
Sharma AK (2002). Bio-fertilizers for sustainable agriculture". Agrobios Indian Publications. |
|
|
|
Shinde VS (1990). Response of chickpea (Cicer arietinum L.) to phosphorus with and without PSB (Microphos) as influenced by applied sulphur. Ph.D. Thesis, IARI. Division of Agronomy, New Delhi. |
|
|
|
Sieverding E (1991). Vesicular- Arbuscular Mycorrhiza management in Tropical Agrosystem. Deutsche Gesellschaft fur Technische Zusammenarbeit, Eschborn. |
|
|
Sinclair TR, Hoffer RM Schreiber MM (1971). Reflectance and internal structure of leaves from several crops during a growing season. Agron. J. 63:864-868.
Crossref |
|
|
|
|
|
Singh S, Kapoor KK (1994). Solubilization of insoluble phosphates by bacteria isolated from different sources. Environ. Ecol. 12:51-55. |
|
|
Smith SE, Robson AD, Abbott LK (1992). The involvement of mycorrhizas in assessment of genetically dependent efficicncy of nutrient uptake and use. Plant Soil 146:169-179.
Crossref |
|
|
|
|
Vardhini BV, Rao SSR (1998). Effect of brassinosteroids on growth, metabolite content and yield of Arachis hypogaea. Photochemistry 48:927-930.
Crossref |
|
|
|
Yadav SP, Shrivastava UK (1997). Response of chickpea (Cicer arietinum) to phosphorus and biofertilizer. Legume Res. 20(2):137-140. |
|
|
|
Yang ZS, Shi GA, Jin JH (1992). Effects of epibrassinolide, a growth promoting steroidal lactone I. Activity in selected bioassays. Phsiol. Plant 53:445-452. |
|
|
Zubillaga MM, Aristi JP, Lavado RS (2002). Effect of phosphorus and nitrogen fertilization on sunflower (Helianthus annuus L.) nitrogen uptake and yield. J. Argon. Crop Sci. 188:267-274.
Crossref |
|
|
|
|