ABSTRACT
Notwithstanding the nutritional, economic and fisheries importance of Chrysichthys nigrodigitatus, and its relatively high abundance and propagation in Lake Ahozon (South Benin), nothing is known about the well-being of the species in this water body. Thus, this study investigated length and weight frequency distributions, length-weight relationships and condition factors of C. nigrodigitatus. A total of 1020 individuals of C. nigrodigitatus were sampled with seines, cast nets, gill nets and hooks during a 15 month period, from August 2014 to October 2015. Total length (TL) and body weight (W) frequency curves showed a unimodal distribution. The longest TL and highest W recorded were 324 mm and 456.1 g, respectively. In Lake Ahozon, C. nigrodigitatus population exhibited a significant (P≤0.01) isometric growth pattern, indicated by the power function (W = 0.011TL2.99; r=0.96) and the log-transformed linear regression equation (LogW = 2.99 LogTL - 1.94; r=0.96). Growth models varied spatially, sexually, ontogenetically, and seasonally, while corresponding subpopulations showed allometric or isometric growth trends, with slopes b ranging between 2.47 and 3.21. The population showed conditions factors (K) ranging between 0.8 and 3.08 which significantly varied with habitats (F1,1018 = 7.613, P = 0.006), seasons (F2,1017 = 29, P = 0.0001) and life stage categories (F2,1017 =5.583, P = 0.004). The active breeding that provided an important stock of offspring for recruitment, the isometric growth model and the relatively high condition factors indicated the perfect establishment of C. nigrodigitatus in the artificial lake of Ahozon. The sustainable exploitation of fisheries in this non-conventional medium-environment requires an integrated approach of lake management.
Key words: Claroteid, condition factor, establishment, fisheries, isometric growth, length-weight model, man-made lake.
The silver catfish Chrysichthys nigrodigitatus (Lacepède, 1803) is widely distributed in the Western African inland waters, but also occurs to a lesser extent in Central, Northern and Southern Africa (Azeroual et al., 2010). This catfish is a highly valuable resource for its numerous fisheries and commercial importance (Leveque, 1997; Andem et al., 2013), its intensive use in aquaculture (Kareem et al., 2015) and its nutritional role. In human health, C. nigrodigitatus provides highly nutritive proteins and nutrients, such as the omega-3 fatty acid, required by human body to maintain its cardiovascular health and to reduce tissue inflammation (Abowei and Ezekiel, 2013). Furthermore, C. nigrodigitatus is known for its ecological role in determining the dynamics and the structure of aquatic ecosystems (Abu and Agarin, 2016).
In the Benin freshwater and brackish water systems, C. nigrodigitatus is widely distributed (Van Thielen et al., 1987) and is among the most exploited fish species (Leveque et al., 1992; Laleye et al., 1995). Moreover, in Southern Benin, the species was introduced in a sand-dragged artificial lake of Ahozon where it constituted about 29.40% of the lake’s fish biomass (Gbaguidi et al., 2016a, b). In Lake Ahozon, C. nigrodigitatus showed a sex-ratio of 1:2.1 and fecundities ranged between 350 and 26 040 eggs (Adité et al., 2017). In this special habitat, the species reproduced all seasons and exhibited a multiple spawner behavior, with sizes at first sexual maturity of 20 and 17 cm-TL for males and females, respectively. C. nigrodigitatus exhibited an opportunistic benthic food habit and fed mainly on aquatic insects, sand particles, detritus, seeds and algae (Lawal et al., 2010; Atobatele and Ugwumba, 2011; Gbaguidi et al., 2016). In the artificial lake of Ahozon, the species is sporadically exploited by a couple of migrant fishermen for domestic uses and sales.
Despite the nutritional, economic and fisheries importance of C. nigrodigitatus, and its relatively high abundance and propagation in Lake Ahozon, nothing is known about length-weight relationships and condition factors of the species in this water body. Length-weight models of fishes depict growth patterns that could be isometric when length and weight increase at identical growth rates, or rather allometric when these two morphometric traits increase at different growth rates (Adeyemi, 2010; Abowei, 2010b; Adeboyejo et al., 2016). Likewise, condition factor indicates the well-being and robustness of fishes and reflects interactions between biotic and abiotic parameters (Iyabo, 2015). Body condition of fishes is mainly affected by food availability (Ayoade and Ikulala, 2007), climate (Atobatele, 2013), gender, maturity stage, environment degradation and the overall ecological health of habitats (Bolarinwa, 2016). Indeed, domestic waste dumping, intrusion of agricultural pesticides and effluent discharges from various industries could alter water quality and modify the biological diversity with in particular, changes in the fish community structure and growth patterns. As reported by Seiyaboh et al. (2016), fish are able to bioaccumulate and biomagnify contaminants from aquatic ecosystems and both water quality alterations, and direct intrusions of contaminant in the body will negatively affect the growth and condition factors of fishes.
This investigation seeks to examine length-weight relationships and condition factors of C. nigrodigitatus to fill a gap in the current biological knowledge of the species, and to contribute to fisheries management and the valorization of numerous sand-dragged artificial lakes of Southern Benin.
Study region
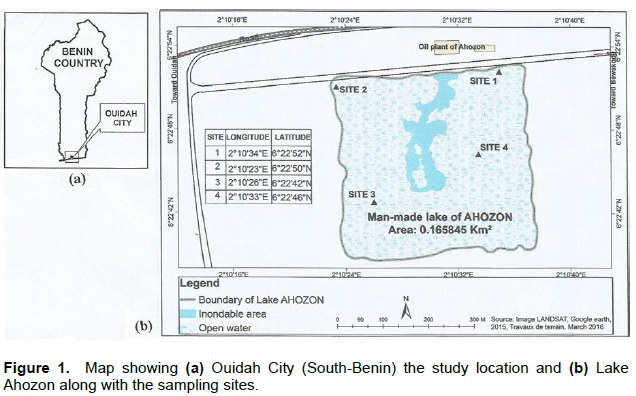
The study location is the artificial lake of Ahozon situated in Ouidah City in Southern Benin (Figure 1a). Lake Ahozon (06°22’52”N; 002°10’34”E) is a sand-dragged water body extending on about 0.165845 km2 (Figure 1b) and lies between a freshwater body, Lagoon Toho-Todougba, and a brackish water, the Benin coastal lagoon. The study area showed a sub-equatorial climate with two wet seasons (April-July; mid-September-October) and two dry seasons (December-March; August-mid-September). Annual average rainfall reached 1310 mm (Akoegninou et al., 1993) and ambient temperatures varied from 25 and 33.6°C (ASCENA, 2003). Intense agriculture using pesticides dominates the study area.
Lake Ahozon is an unmanaged, neglected and abandoned water body that receives every wet season, an important volume of running waters (Adite et al., 2017). However, as reported by Gbaguidi et al. (2016), evaluation of water physicochemical parameters indicated that Lake Ahozon is of good quality and favorable for the development of aquatic organisms, including fish resource.
Dominant phytoplankton genera identified in Lake Ahozon were Navicula, Peridinium, Scennedesmus, Pinnularia, Spirogyra, Cosmarium, Melosira, Synechocystis, Microcystis, Oscillatoria, Euglena, Phacus, Surirella and Lychmophora, and zooplanktons were dominated by Trichocerca, Keratella, Brachionus and copepods. Benthic macro-invertebrates recorded were chironomid larvae and a Gasteropod mollusk, Melanoïde tubercularis. Aquatic vegetation was dominated by Cyperus crassipes, Cyperus rotundus, Fuirena umbellata, Andropogon gayanus, Ludwigia perennis, Emilia praetermissa, Eleocharis complanata, Enydra fluctuans and Mariscus ligularis. Six species, Sarotherodon galilaeus, Oreochromis niloticus, Tilapia guineensis, Chrysichthys nigrodigitatus, Heterotis niloticus and Clarias gariepinus (Gbaguidi et al., 2016a, b) composed the fish community of Lake Ahozon, and were sporadically exploited by a couple of migrant fishermen.
Sampling sites
In Lake Ahozon C. nigrodigitatus individuals were sampled on four sites including two in the “aquatic vegetation” habitat and two in the “open water” habitat (Figure 1b). The “aquatic vegetation” habitat, the edge of the lake, is shallow and characterized by a low water velocity and relatively dense vegetation. The “open water” habitat is exempt of vegetation and exhibited a relatively high depth and high water velocity.
Fish samplings
Fish collections were done twice a month from August 2014 to October 2015. Samplings were performed both in the “aquatic vegetation” habitat and in the “open water” habitat using cast net (9.80 m-diameter, 4.90 m-height, 40 mm-mesh), seine (4.15 m-length × 1.77 m-width, 3 mm-mesh), hooks (90 m-length) and experimental gill nets (40 m × 1.05 m, 40 mm mesh). Seines were used at the lake’s edge whereas cast nets, gill nets and hooks were used in the “open water” according to Adite et al. method (2017). The two samplings per month and per habitat and the use of various fishing gears helped to get a relatively high sample size (1020 individuals) that reflects the population structure of C. nigrodigitatus.
Once collected, C. nigrodigitatus individuals were identified first in situ using references such as Needham and Needham (1962), Van Thielen et al. (1987) and Leveque et al. (1992). Individuals were then counted by habitat (open water, aquatic vegetation), gender (male, female), life stage (juvenile, sub adults, adults) and season (flood, dry, wet) to compute the relative abundance of each corresponding subpopulation. In addition, each of the 1020 individual was measured for its total length (TL) and standard length (SL) to the nearest 0.1 mm with a measuring board, and weighted to the nearest 0.1 g with an electronic scale. Fish samples were then preserved in 10% formalin and transported to the “Laboratoire d’Ecologie et de Management des Ecosystèmes Aquatiques (LEMEA)” and preserved in 90% ethanol.
Data analysis
Spatial, seasonal, sexual and ontogenetic biometric data including total length (TL), standard length (SL) and fish individual weight (W) were recorded in spreadsheets generated from SPSS computer software (Morgan et al., 2001). Range and average values of biometric data were also computed for C. nigrodigitatus population and for habitat, season, life stage, and gender subpopulations. The population size structure was examined by generating standard lengths (SL) frequency histograms of C. nigrodigitatus. Length-weight models were examined following Le Cren’s (1951) power function along with its logarithmic-transformed linear model:
W = aTLb (Le Cren, 1951) (1)
LogW = Loga + b LogTL (2)
where TL is the fish total length, W is the fish individual weight, a is the intercept, and b, the slope, is the allometry coefficient (Le Cren, 1951). One-way analysis of variance (ANOVA) was used to test significance of the regression. Fulton’s condition factors of C. nigrodigitatus population were computed following Tesch (1971) model:
where K is the Fulton’s condition factor, W, the fish individual weight (g), TL the total length (cm), and b = 3 is the allometry coefficient. In addition, Fulton’s condition factors (K) and growth trends (length-weight relationships) were examined by habitat types (open water; aquatic vegetation), genders (male, female), life stage categories (juveniles, sub adults, adults) and seasons (flood, dry, wet).
Population structure
Abundance
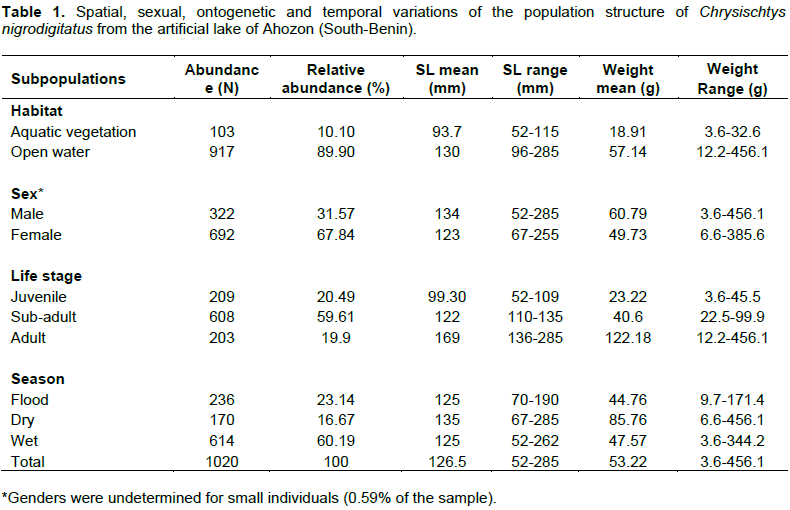
In the sand-dragged man-made lake of Ahozon, the introduced silver catfish, C. nigrodigitatus exhibited significant (P≤0.05) spatial and seasonal variations of the species relative abundance with the “open water” habitat comprising alone 89.90% of the population and the “wet season” subpopulation making alone 60.19% (Table 1). Sexual variations of abundance were depicted with females dominating the population and numerically made up 67.84% of C. nigrodigitatus assemblages. Ontogenetically, aggregated juveniles (mean SL: 99 mm) and sub- adults (mean SL: 122 mm) made about 80.10% of the population.
Length and weight frequency distributions
Figures 2 and 3 show standard length (SL) and body weight (W) frequency distributions of C. nigrodigitatus
from Lake Ahozon. Overall, the population showed, SL varying between 52 mm (TL: 67 mm) and 285 mm (TL: 324 mm). The length-frequency curve established for the whole population exhibited a unimodal size distribution with the modal class (120 to 180 mm) showing the highest frequency occurrence of 503 individuals and class (0 to 60 mm) showing the lowest frequency occurrence of one individual (Figure 2).
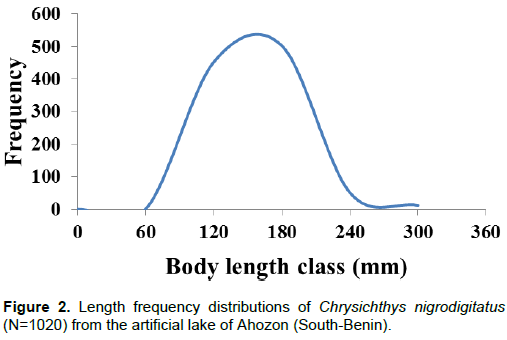
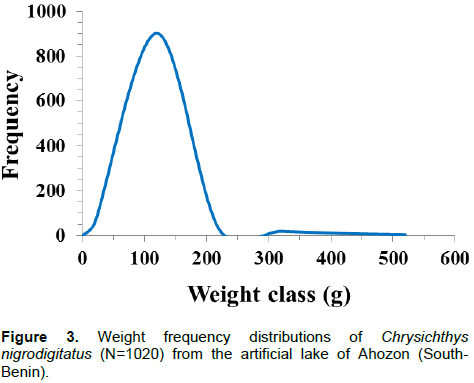
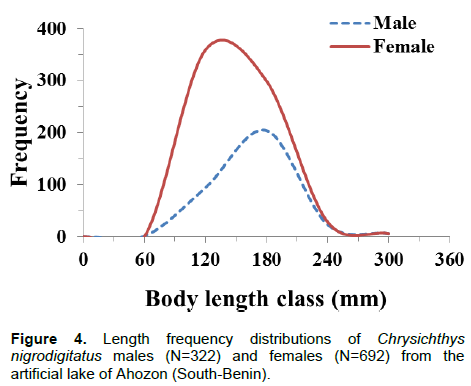
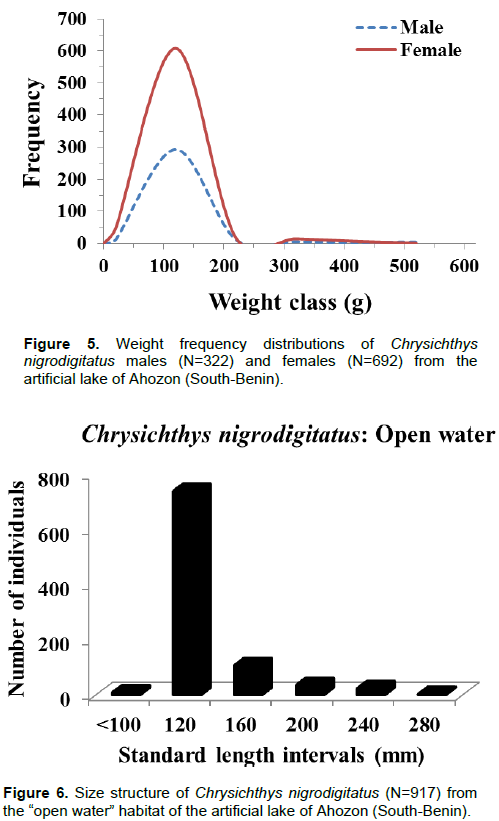
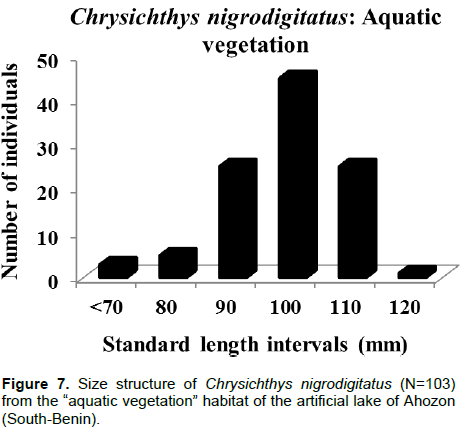
Body weight ranged between 3.6 g (SL: 52 mm) and 456.1 g (SL: 246 mm) and like body length, the weight-frequency curve established for the whole population showed a unimodal distribution with the modal class (20 to 120 g) showing the highest frequency of 902 individuals and class (420 to 520 g) showing the lowest frequency of four individuals (Figure 3). Sexually, the same trends of unimodal distributions for length and weight frequency were recorded for both genders (Figures 4 and 5). Spatially, standard length (SL) frequency histograms showed unimodal size distributions for both “open water” habitat and “aquatic vegetation” habitat (Figures 6 and 7).
Length-weight relationships
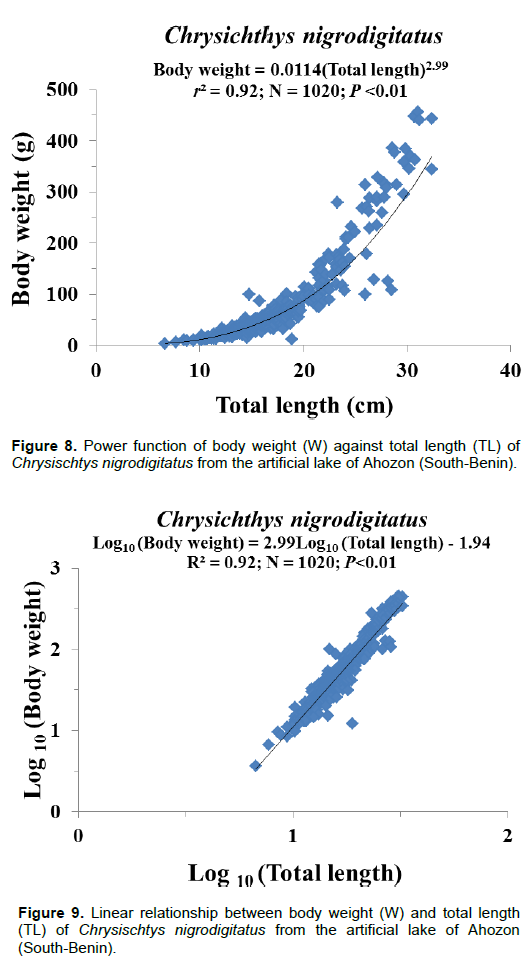
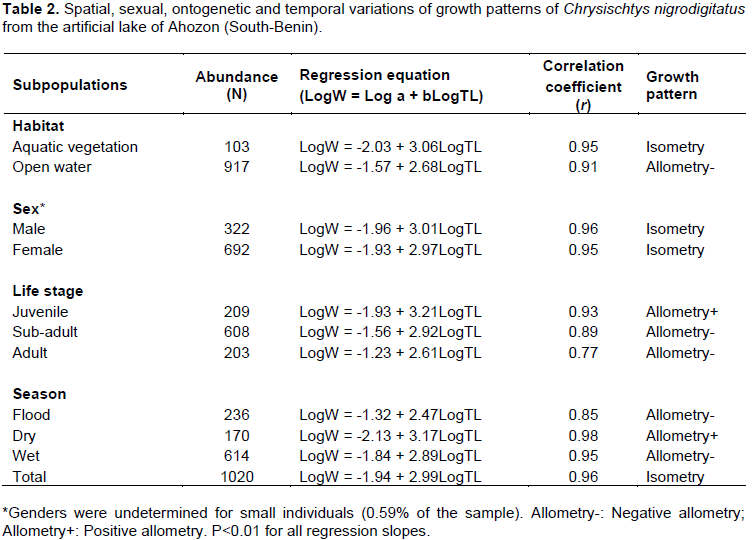
Figures 8 and 9 show the length-weight models established between C. nigrodigitatus total length (TL) and body weight (W). The resulting power function equation and the logarithm-transformed linear regression equation for the whole population were as follow:
W = 0.011TL2.99 (Le Cren, 1951); (4)
Log W = 2.99 Log (TL) - 1.94 (5)
r = 0.96; N=1020
where W is the individual weight, TL is the total length, R is the correlation coefficient and N, the number of individuals.
In addition, the linear regression equations established for habitat type, gender, life category and season sub-populations gave positive slopes ranging between b=2.47 for the flood season sub-population and b=3.21 for the juvenile life stage sub-population. The associated correlation coefficients “r” were significant (P≤0.05) and varied between 0.77 and 0.99 (Table 2). The results consistently indicated that the sub-population of dry period showed higher slope b = 3.17 compared with flood and wet seasons. Ontogenetically, juvenile sub-population exhibited higher slope b = 3.21 compared to sub-adults and adults. Spatial variations of growth were recorded with the “aquatic vegetation” sub-population showing higher slope b=3.06, whereas sexually, male sub-population exhibited higher slope b = 3.005, corresponding to an isometric growth pattern.
Condition factors (K)
The body condition of C. nigrodigitatus from Lake Ahozon was evaluated through Fulton’s condition factors (K) that were computed by season, life stage category, gender and habitat type. Overall, the population exhibited condition factors (K) ranging between 0.18 and 3.08 and averaging 1.12±0.19 (Table 3). Significant (P≤0.05) variations of average K were also recorded seasonally, ontogenetically, sexually and spatially. Seasonally, the mean condition factor was higher during the dry period with K= 1.21±0.22 compared to those of wet and flood period. Ontogenetically, juvenile sub-population exhibited better condition (mean K= 1.15±0.26) compared to sub-adult and adult sub-populations, whereas sexually, male sub-population exhibited higher mean condition indices (1.12±0.19). Spatially, the “aquatic vegetation” sub-population showed higher condition factor (Mean K= 1.16 ±0.17), compared to the open water subpopulation.
Fisheries management of numerous abandoned sand-dragged artificial lakes distributed throughout Benin is badly needed to valorize fish resources (Laleye et al., 2004), to increase grass-root revenues (Gbaguidi et al., 2016), to lessen degradation pressures and to contribute to restore natural aquatic biota (Iyabo, 2015). Two years and half after its introduction in the sand-dragged man-made lake of Ahozon, C. nigrodigtatus efficiently exploited the existing food resources and spawned actively to become one of the prominent fishable species in this special water body (Adite et al., 2017). Management evaluation tools such relative abundance, length and weight frequency, length-weight relationships and condition factors globally indicated a perfect establishment of this catfish in Lake Ahozon.
Abundance, length/weight frequencies and growth patterns
In Lake Ahozon, C. nigrodigitatus made about 29.40% of the fish biomass and numerically accounted for 11.22% of the fish community (Gbaguidi et al., 2016). As further reported by Gbaguidi et al. (2016), the favorable physicochemical traits of Lake Ahozon which is within the tolerance ranges of C. nigrodigitatus, the high availability and utilization of food resources, the active breeding and the multiple spawner behavior accounted for the relatively high propagation of this claroteid in Lake Ahozon. In particular, the high abundance (80%) of aggregated juveniles and sub-adults indicated that C. nigrodigitatus provided an important stock of offspring for recruitment in this water body. These results are comparable to those recorded on natural coastal lakes and lagoons of Benin where C. nigrodigitatus biomass reached 26% of the fish productions (Direction des Pêches, 1996). These findings indicated that this claroteid is well-established in the man-made lake of Ahozon as it is observed in most aquatic natural biota of Benin. In contrast, Akpan (2013) reported for these species, a lower percentage biomass from Uta Ewa Creek of Niger Delta Region of Nigeria, probably because of overfishing and environmental degradation.
In Lake Ahozon, the maximum total length (TL = 324 mm) recorded, though relatively high, is lower than TL = 590 mm reported by Laleye et al. (1995) in Lake Nokoué in Benin and TL = 1090 mm reported by Andem et al. (2013) in the Itu Head Bridge, in Akwa Ibom State of Nigeria. Inversely, the maximum TL = 324 mm recorded in Lake Ahozon was higher than that reported by Iyabo (2015) in the Ebonyi River, South Eastern of Nigeria. According to King (1996), the maximum lengths attained by fishes depend on the species, habitat conditions and its variability, season and environmental stochasticity. For example, Lake Nokoué, the largest water body in Benin, exhibited a high productivity because of the seasonal flooding of Oueme and Sô rivers (both connected to Lake Nokoué) that discharge a huge quantity of nutrients in this coastal lake. Also, Lake Nokoué was invaded by “acadja” that fertilizes this water body and hence, increases its primary productivity. The “acadja” is a park of tree branches constructed in the shallow parts of the lake by local fishermen as a fishery method (Laleye et al., 1995). In contrast, Lake Ahozon is an isolated young man-made water body of modest productivity because exempt of local management tool such as “acadja” and lacking connection with other inland waters from which this lake could receive nutrients to boost its productivity and to improve the growth of fishes.
Regardless of habitat, season, gender and developmental stage, length-weight regression equations of C. nigrodigitatus population gave significant (P≤0.05) slopes b=2.99, indicating that, globally, this claroteid exhibited an isometric growth pattern (Table 2 and Figures 8 and 9). Consequently, length and weight increased approximately at identical growth rates and hence, during life cycles, the shape of the fish remained nearly constant. The high correlation coefficient (r=0.96) recorded indicated that there was a strong association between total length and body weight (Nwani, 2006). These results agree with those reported by Offem et al. (2008) and Bolarinwa (2015) that have reported an isometric growth trend for C. nigrodigitatus population in the forest-savana of the Cross River and in the Epe Lagoon of Nigeria, respectively. Inversely, several authors such as Adeboyejo et al. (2016), Iyabo (2015), and Abu and Agarin (2016) reported positive and negative allometric growths for C. nigrodigitatus in Badagry Creek of Lagos, in the Ebonyi River of South- East and in the Lower Reaches of the New Calabar River Niger Delta of Nigeria, respectively. Positive allometric growth, as noted by Tesch (1971) and Bagenal (1978), suggests that body weight increased faster than length whereas negative allometric growth implies that length increased faster than weight and hence, fish become more slender and less valuable. In this study, the isometric growth trends depicted in C. nigrodigitatus is the result of the favorable water quality of Lake Ahozon characterized by a moderate temperature (mean: 33.25±2.20°C), a high water transparency (mean: 36.63±11.18 cm), a high conductivity (mean: 240±118.40 µ/cm), an alkaline pH (mean: 7.51±0.78), a relatively high concentration of dissolved oxygen (mean: 5.43±2.52 mg/L) and a relatively high percent of saturation (mean: 82.69±41.52%) (Gbaguidi et al., 2016).
In Lake Ahozon, length-weight models varied with habitats (Open water, Aquatic vegetation), seasons (flood, dry, wet), genders (male, female) and life stage category (juvenile, sub adults, adults) with slopes varying between 2.47 and 3.21 (Table 2). Spatially, the aquatic vegetation subpopulation showed an isometric growth (b = 3.06) compared to the open water habitat that exhibited a negative allometric growth (b = 2.68) and where preys were probably less available and less concentrated (Gbaguidi et al., 2017). Though females are generally known to exhibited lower growth rates compared to male because females mobilized their metabolic energy to reproduce, in this study, males and females showed an isometric growth rate and exhibited nearly identical slopes b = 3.01 and b = 2.97, respectively. Probably, the relatively high and identical growth rates recorded in both genders were the results of the high availability of food resources along with their high exploitation and utilization by both genders. Ontogenetically, the sub-populations of the three life stage categories, juveniles, sub-adults and adults showed allometric growth patterns. Particularly, and in contrast with sub-adults and adults, juvenile sub-population exhibited a positive allometric growth trend with b=3.21, the highest slope recorded during this study. As reported by Gbaguidi et al. (2016), juveniles utilize their entire metabolic energy to grow, whereas adults are mostly involved in spawning and mobilized most of their energy for gonad maturation, egg laying, hatching, and parental care of larvae and hence, exhibited in this study, negative allometric growth with slope b<3, indicating that, as length increased the fish became thinner. Seasonally, dry period sub-population showed a positive allometric growth pattern with b=3.17, probably because of the water reduction that caused a high concentration of food resources more available for C. nigrodigitatus. In contrast, the wet and flood season sub-populations exhibited a negative allometric growth trend with slopes b <3. During these periods, food resources were probably more diluted, less concentrated and hence, less available for individuals.
Body condition
In this artificial medium-environment, C. nigrodigitatus showed relatively high condition factors (K) ranging between 0.8 and 3.08 and averaging 1.12±0.19. These findings agree with those reported by Abowei and Ezekiel (2013) in the Amassoma River flood plains of Nigeria. However, Iyabo (2015) reported lower condition factors ranging between 0.58 and 1.74 from the Ebonyi River in South Eastern Nigeria. In contrast, higher condition factors were reported by Bolarinwa (2016) in the Mahin lagoon of Nigeria and by Abu and Agarin (2016) in the Lower Reaches of the New Calabar River Niger Delta. The differential niche breadth, which could be relatively reduced in Lake Ahozon, a young water body, may have been the cause of these variations in the condition factors (K).
For example, in Lake Ahozon, the dominant species Sarotherodon galilaeus (Cichlidae) is a pelagic alguivorous specialist that showed a mean condition factor K= 2.26±0.84 (Gbaguidi et al., 2016) higher than that (1.12±0.19) recorded for C. nigrodigitatus, a benthic feeder that foraged mainly on aquatic insects, substrate particles and detritus. This gap in the condition factor between these two sympatric species could be attributed to the distinctive ecological niche that was probably more available to S. galilaeus than to C. nigrodigitatus. Indeed, except in the aquatic vegetation habitat where the bottom was mostly constituted of benthic preys, the bottom of the open water, the major habitat, was sandy with less benthic substrates because Lake Ahozon is a “young” water body isolated from rivers and floodplains from which it could receive nutrients to increase its primary production and to improve the body condition of C. nigrodigitatus, a benthic feeder.
The study also revealed spatial, ontogenetic and seasonal variations of body condition in the sand-dragged man-made lake of Ahozon (Table 3). Indeed, one-way ANOVA on K across habitats (open water, aquatic vegetation), seasons (flood, dry, wet), and life stages (juveniles, sub adults, adults) subpopulations showed significant (P<0.01) variations of condition indices. The computed F-values, along with degrees of freedom and P-values were F1,1018 = 7.613, P = 0.006 for habitats, F2,1017 = 29, P = 0.0001 for seasons, and F2,1017 =5.583, P = 0.004 for life stages. Overall, condition factors were higher in the aquatic vegetation subpopulation where K averaged 1.16±0.17. As reported by Gbaguidi et al. (2017), foods are usually more available, more diversified and concentrated in aquatic vegetation habitats. Likewise, C. nigrodigitatus showed a higher well-being (mean K: 1.21±0.22) during the dry period when water volume was reduced and more concentrated in preys items. Ontogenetically, juvenile subpopulation exhibited a higher condition factors with K averaging 1.15±0.26, compared to sub-adults and adults showing lower values. Indeed, in general, juvenile mobilized its whole metabolic energy to grow whereas adults were mostly involved in spawning and consequently, mobilized their metabolic energy for reproduction and parental care (Murphy and Willis, 1996). In contrast with habitats, seasons and life stages, one-way analysis of variance on K across males and females showed insignificant (F1,1011 = 1.942, P = 0.164) sexual variation of condition factors, probably because preys were highly available to both genders that foraged ad libitum to compensate energy spent during spawning. Nevertheless, though insignificant, the well-being of male subpopulation tended to be higher than that of females.
The current study revealed that C. nigrodigitatus was well-conditioned and globally showed an isometric growth pattern, indicating that Lake Ahozon was suitable for the well-being and propagation of this valuable claroteid. The results showed spatial, seasonal and ontogenetic variations of growth factors. Length and weight frequency distribution evidenced the availability of fishable subpopulation composed of larger individuals along with an important stock of offspring for recruitment. Sustainable exploitation of Lake Ahozon fisheries requires an integrated management scheme including a periodic physico-chemical and biota monitoring of the water body.
The authors declare that there is no conflict of interest.
REFERENCES
|
Abowei JFN, Ezekiel EN (2013).The Length-weight relationship and condition factor of Chrysichthys nigrodigitatus (Lacepède, 1803) from Amassoma River flood plains. Sci. Agric. 3(2):30-37.
|
|
|
|
Abowei JFN, (2010b). The condition factor, length - weight relationship and abundance of Elops seneganensis (Regan, 1909) from Nkoro River, Niger Delta, Nigeria. Adv. J. Food Sci. Technol. 2:16-21.
|
|
|
|
Abu OMG, Agarin OJ (2016). Length-Weight Relationship and Condition Factor of Silver Catfish (Chrysichthys nigrodigitatus) from the Lower Reaches of the New Calabar River Niger Delta. Int. J. Innovative Stu. Aqu. Biol. Fish. 2(4):1-7.
|
|
|
|
Adeboyejo OA, Clarke OE, Ekele AS (2016). Length–weight relationship, condition factor and sex-ratio of fish fauna in Badagry Creek, Lagos, Nigeria. Int. J. Marine Sci. 6(24):1-8.
|
|
|
|
Adeyemi SO (2010). Length-weight relationhip and condition factor of Protopterus annectens (OWEN) in Idah area of River Niger, Nigeria. Anim. Res. Int. 7(3):1264-1266.
|
|
|
|
Adite A, Gbaguidi HMGA, Ategbo JM (2017). Reproductive Biology and Life History Patterns of the Claroteid, Chrysichthys nigrodigitatus (Lacépède: 1803) from a Man-made Lake of Southern Benin. J. Fish. Aquat. Sci. 12:106-116.
Crossref
|
|
|
|
Akpan II (2013). Species composition and abundance in Uta Ewa Creek, Niger Delta Region, Nigeria. J. Agric. Vet. Sci. 3(3):56-60.
|
|
|
|
Akoegninou A, Oyede LM, Toffi M (1993). La mangrove du Benin: Environnement physique, végétation, et essais de gestion. Document technique No 2, FAST-UNB, Abomey-Calavi.
|
|
|
|
Andem AB, George UU, Eyo VO (2013). Length Frequency Distribution of (Chrysichthys nigrodigitatus) (Lecepede, 1803) (Chrysichthys, Bagridae) from Itu Head Brigde, in Akwa Ibom State, Nigeria. Int. J. Sci. Res. 2(9):258-260.
|
|
|
|
ASCENA (2003). Données sur la pluviométrie des années 2002 et 2003. Agence pour la sécurité de la navigation Aérienne, Service de la Méréorologie Nationale, Cotonou.
|
|
|
|
Atobatele OE, Ugwumba AO (2011).Condition factor and diet of Chrysichthys nigrodigitatus and Chrysichthys auratus (Siluriformes: Bagridae) from Aiba Reservoir, Iwo, Nigeria. Int. J. Trop. Biol. 59(3):1233-1244.
|
|
|
|
Atobatele OE (2013).Growth pattern and morphometric discrimination of two congeneric species of Chrysichthys, C. nigrodigitatus and C. auratus (Siluriformes, Bagridae), from a small tropical reservoir. Pan-Am. J. Aqua. Sci. 8(1):62-67.
|
|
|
|
Ayoade AA, Ikulala AOO (2007). Length weight relationship, condition factor and stomach contents of Hemichromis bimaculatus, Sarotherodon melanotheron and Chromidotilapia guentheri (Perciformes: Cichlidae) in Eleiyele Lake, Southwestern Nigeria. Int. J. Trop. Biol. 55(3):969-977.
Crossref
|
|
|
|
Azeroual A, da Costa L, Lalèyè P, Moelants T (2010). Chrysichthys nigrodigitatus. In: The IUCN Red List of Threatened Species.
View
|
|
|
|
Bagenal TB (1978). Aspects of fish fecundity. In: Ecology of freshwater fish production, Gerking, S.D. (Eds.), Blackwell Scientific Publications, Oxford pp. 75-101.
|
|
|
|
Bolarinwa JB (2015). Length- weight relationships and condition factors of Tilapia zilli and Chrysichthys nigrodigitatus in Epe Lagoon, Nigeria. Sci. Eng. Perspect. 10:21-28.
|
|
|
|
Bolarinwa JB (2016). Length- weight relationships and condition factors of Oreochromis niloticus and Chrysichthys nigrodigitatus in Mahin lagoon, Nigeria. Resear. J. Agri. Environ. Manage. 5(3):075-080.
|
|
|
|
Direction des Pêches (1996). Statistiques des pêches continentales année 1995. Projet Pêche Lagunaire, Cotonou, Bénin.
|
|
|
|
Gbaguidi HMAG, Adite A, Abou Y (2017). Trophic Ecology and Establishment of the Silver Catfish, Chrysichthys nigrodigitatus (Pisces: Siluriformes: Claroteidae) Introduced in an Artificial Pond of Benin. J. Fish. Aquat. Sci. 12(1):42-53
|
|
|
|
Gbaguidi GAMH, Adite A, Sossoukpe E (2016a). Ecology and Fish Biodiversity of some Artificial Lakes of Southern Benin (West Africa): Implications for Fisheries Management and Aquaculture Valorization. J. Environ. Prot. 7(6):874-894.
Crossref
|
|
|
|
Gbaguidi HMAG, Adite A, Sossoukpe E (2016b). Abundance, length - weight relationships and Fulton's condition factor of the freshwater cichlid Sarotherodon galilaeus (Pisces: Teleostei: Perciformes) from a sand-dragged man-made lake of Southern Benin, West Africa. J. Biodivers. Environ. Sci. 8(5):75-87.
|
|
|
|
Iyabo UB (2015). Length-Weight Relationship and Condition Factor of Chrysichthys nigrodigitatus (Lacepede: 1803) of Ebonyi River, South Eastern Nigeria. Am. J. Agri. Sci. 2(2):70-74.
|
|
|
|
Kareem OK, Olanrewaju AN, Orisasona O (2015). Length-weight Relationship and Condition factor of Chrysichythys nigrodigitatus and Schilbe mystus in Erelu Lake, Oyo State, Nigeria. J. Fish. Livest. Prod. 3(4):1-4.
|
|
|
|
King RP (1996). Length-weight Relationship of Nigeria freshwater fishes. Naga ICLARM Quarterly 19 (3):49-52.
|
|
|
|
Laleye P, Chikou A, Philippart JC, Teugels GG, Van De Walle P (2004). Etude de la diversité ichtyologique du bassin du fleuve Ouémé au Bénin (Afrique de l'Ouest). Cybium 28:329-339.
|
|
|
|
Laleye P, JC Phillipart, Poncin P (1995). Biologie de la reproduction de deux espèces de Chrysichthys (Siluriformes, Bagridae) du lac Nokoué et de la lagune de Porto-Novo au Bénin. J. Afr. Zool. 109(3):213-224.
|
|
|
|
Lawal OM, Sangoleye OJ, Seriki BM (2010). Morphometry and diet of Chrysichthys nigrodigitatus (Lacepede, 1803) in Epe Lagoon, Nigeria. Afri. J. Biotechnol. 9(46):7955-7960.
Crossref
|
|
|
|
Le Cren ED (1951). The length-weight relationship and seasonal cycle in gonad weight and condition in the perch Perca fluviatilis. J. Anim. Ecol. 20:201-219.
Crossref
|
|
|
|
Leveque C, Paugy D, Teugels GG (1992). Faune des poissons d'eaux douces et saumâtres de l'Afrique de l'Ouest. Tome 2. Editions OSTORM/MRAC, Paris.
|
|
|
|
Leveque C (1997). Biodiversity dynamics and conservation: The freshwater fish of tropical Africa, Cambridge University Press.
|
|
|
|
Morgan GA, Grieggo OV, Gloeckner GW (2001). SPSS for Windows: An introduction to use and interpretation in research, Lawrence Erlbaum Associates, Publishers, Mahwah, New Jersey.
|
|
|
|
Murphy BR, Willis DW (1996). Fisheries Techniques. Second edition. American Fisheries Society, Bethesa, Maryland.
|
|
|
|
Needham GJ, Needham PR (1962). A guide to the study of fresh-water biology, Holden Day, San Francisco, California.
|
|
|
|
Nwani CD (2006). Length-Weight Relationship and Condition Factor of Distichodus species of Anambra River. Anim. Res. Int. 3(2):461-465.
|
|
|
|
Offem BO, Akegbejo-Samsons Y, Omoniyi IT (2008). Diet, size and reproductive biology of the silver catfish, Chrysichthys nigrodigitatus (Siluformes: Bagridae) in the Cross River, Nigeria. Rev. Biol. Trop. 56(4):1785-1799.
|
|
|
|
Seiyaboh EI, Harry GA, Izah SC (2016). Length-Weight Relationship and Condition Factor of Five Fish Species from River Brass, Niger Delta. Biotechnol. Res. 2(4):187-192.
|
|
|
|
Tesch FW (1971). Age and Growth. In: Methods for Assessment of Fish Production in Fresh Waters, Ricker, W.E. (Ed.). Blackwell Scientific Publications, Oxford, UK.
|
|
|
|
Van Thielen R, Hounkpe C, Agon G, Dagba L (1987). Guide de détermination des Poissons et Crustacés des Lagunes et Lacs du Bas-Bénin. Direction des Pêches, Cotonou, Bénin.
|