ABSTRACT
Fruits of persimmon are considered as one of the health promoting foods which has been established as one of the major fruits in Korea. Persimmon is one of the fruits processed mainly by drying. Quality characteristics of dried persimmon prepared with different fruit sizes of cultivar Sanggandungsi were investigated. Smaller sized fruits showed significantly high weight loss as compared to bigger ones due to drying. On the other hand, bigger sized fruits possessed significantly high value for hardness after 35 days of drying. Size of fruit was not found to be vital for soluble solid contents of dried fruits. One of the important bioactive molecules of persimmon fruits (tannin content) is significantly high in bigger sized fruits as compared to smaller ones. Significant differences in various parameters among different sized fruits indicate that, physicochemical characteristics of dried persimmon fruit could be affected by the fruit size.
Key words: Drying, fruit size variation, physicochemical properties.
Persimmon (Diospyros kaki Thunb.) fruit is rich in health promoting substances. It contains health promoting bioactive compounds like, ascorbic acid, condensed tannins, and carotenoids, which have antioxidant properties (Rao and Rao, 2007; Shahkoomahally et al., 2015; Fu et al., 2016). Consumption of persimmon fruit could be useful for the prevention of atherosclerosis (Zhang et al., 2016) because of its plasma lipid-lowering ability and antioxidant properties.
In China, it is widely consumed against health problems such as coughs, hypertension, dyspnoea, paralysis, burns, and bleeding because the fruit is rich in nutrients such as vitamin C, vitamin A, calcium, iron, and phenolic compounds (Nicoleti et al., 2007). It also shows inhibitory effect on human lymphoid leukemia cells, mutagenicity of C-nitro and C-nitroso compounds (Achiwa et al., 1997) as well as the incidence of stroke and the extension of the lifespan of hypertensive rats (Uchida et al., 1995; Xie et al., 2015). Reports suggest that consumption of the persimmon fruits is good for human health (Lia et al., 2013; Butt et al., 2015). However, regular availability of fruits is difficult since persimmon is grown only in a particular season in specific climatic regions. Moreover, some consumers are reluctant to buy fresh fruits because of the possibility of having its poor quality when consumed fresh (Byrne, 2002). Such circumstances favor producing high quality dried fruits.
Drying is one of the oldest methods of food preservation (Dehydration, 1998). Fruits can be dried to preserve and allow them to be available during the period, when fresh fruits could not be harvested. The dry fruits may retain the characteristics of natural products, offer reduced transportation cost due to reduced mass, and resist growth of mold and other microorganisms which cause deterioration in fresh fruits (Marques et al., 2006). Drying of persimmon fruits also reduces the astringency that comes from the phenolic compounds of some varieties. Astringency and color have been reported as important quality characteristics of dried persimmon (Akyildiz et al., 2004).
Persimmon, one of the best health promoting fruits, is processed mainly by drying. Few reports have been published describing the effect of drying on some fruits. However, there was no published reference report describing the quality characteristics of dried persimmon of different fruit sizes. The objective of this study was to investigate the quality characteristics of dried persimmon of different fruit sizes. The present study may provide information about selection of proper sized persimmon fruits, for drying.
Preparation of dried persimmon
Persimmon fruits were dried following the method of Kim et al. (2014). The fruits were washed with tap water and peeled off manually using a peeler. The pedicels of peeled fruits were tied to string and hanged for natural drying. Before peeling and subsequent drying, the fruits were divided into 4 categories based on their mass as: S-1 (200±5 g), S-2 (240±5 g), S-3 (280±5 g), and S-4 (320±5 g).
Seven fruits were tied on 1 m long strings and 2 to 3 strings were tied together while hanging for drying. The strings of persimmons were hung under the eaves of a roof, where they got plenty of sun and breeze but were protected from rain. The strings of persimmons fruits kept for drying were hung more than 3 m above the ground. In the present experiment, fruits were dried for 35 days. Average temperature and relative humidity during the fruit drying period were 7.2°C and 78.79%, respectively.
Fruit weight and soluble solid
Weight of raw and dried persimmon fruits (with pedicel) was measured using a digital balance (GT 480, Ohaus, Florham Park, NJ, USA). Soluble solid content was determined using a refractometer (RX-5000α, Atago, Tokyo, Japan) and expressed as °Brix. All measurements were made in triplicate and average values were reported.
Determination of moisture and hardness
Moisture content determined was measured following standard method (AOAC, 1995 method 950.46). The weight loss was calculated by subtracting the weight of the fresh peeled fruits and the weight of the dried peeled fruits, dividing the difference by the weight of the fresh peeled fruits, and then multiplying the quotient by 100. Hardness was measured using rheometer (Compac-100, Sun Scientific Co. LTD, Japan) under the following operational conditions: test type, mastication; adaptor type, circle; adaptor area, 0.20 cm2; and table speed, 60 mm/min.
Color measurement
L*(lightness), a*(redness, + or greenness, -), and b*(yellowness, + or blueness,-) values of dried persimmon were measured using a chroma meter (CR-300; Minolta Corp., Osaka, Japan). A Minolta calibration plate (YCIE=94.5, XCIE=0.3160, YCIE=0.330) and a Hunter Lab standard plate (L*=97.51, a*=-0.18, b*=+1.67) were used to standardize the instrument using a D65 illuminant. Color values were measured directly on 3 zones, of each dried persimmon and average values were reported.
Tannin content
Tannin content of dried persimmon was analyzed according to the method described by modified Prussian Blue assay (Graham, 1992; Price et al., 1988). One hundred milliliters of sample extract was added to 3 mL of distilled water, centrifuged at 10000× g for 15 min, and supernatant was collected. Absorbance was measured at 700 nm using a spectrophotometer (Hewlett- Packard, model 8452A, Rockvile, USA). The total phenolic contents were calculated from the calibration curve prepared from the absorbance of gallic acid standard solutions (Park, 1999). After which, phenolic content was expressed as tannin content.
Statistical analysis
Data were subjected to analysis of variance (ANOVA) and differences between means at P<0.05 were analyzed using the Tukey test. The statistics package version 4.0 (Analytical Software,Tucson, AZ, USA) was used for statistical analysis. Average values of triplicate measurements were considered for statistical analysis unless otherwise mentioned.
Weight loss
Four sorted persimmon fruit samples for each group with average weights of 200, 240, 280, and 320 g were peeled and dried at natural condition for 35 days. Weight loss of peeled persimmon at the end of drying was significantly (P ≤ 0.05) high for fruits of lower weights, S-1 (55.5%) and S-2 (55.7%) as compared to that of S-3 (52.0%) and S-4 (49.8%) (Table 1). Lower wet loss, in the present study, in the bigger fruits might be due to higher dry matter contents in bigger sized fruits. Results of Tittonell et al. (2001) suggest that, bigger sized leaves contain more dry matters as compared to that in smaller ones. Moreover, the weight loss increased with a decreasing sample size of apple fruit slices (Van Nieuwenhuijzen et al., 2001).
Hardness and moisture and soluble solid contents
Hardness of the small sized fruits was significantly low as compared to bigger fruits (Table 2). Hardness of S-4 (1.56 Kg/5 mm) was almost three times higher than that of S-1 (0.5 Kg/5 mm). The reason behind higher hardness value in bigger sized fruits might be due to, higher dry matter content in them. The results are in agreement with those of Tittonell et al. (2001) which suggest that, bigger sized leaves contain more dry matters as compared to that in smaller ones. Moisture contents of S-4 (50.1%) were significantly higher than that of other samples (Table 2). Higher moisture content in the bigger size fruits might be explained by the fact that, a smaller sample has a larger surface area/volume ratio as compared to the larger samples (Van Nieuwenhuijzen et al., 2001).
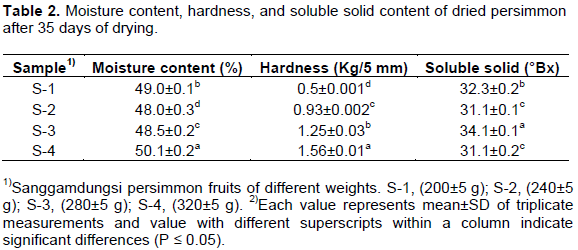
Fruit size could directly affect drying characteristics such as drying rate and moisture diffusivity in blueberry (Shi et al., 2008). MacGregor (2005) found that under similar drying conditions, larger fruits of blueberry took longer to reach the final moisture content than smaller ones, but the larger berries had a higher mass-losing rate than the smaller ones. Sample S-3 (34.1 °Bx) contained significantly high soluble solid contents as compared to other samples (Table 2). Blažková et al. (2002) also found positive correlations between the weight (size) of fruits and soluble solid contents.
Hunter color value and tannin content
Color value of the dried persimmon is known to play an important role in the consumer's acceptability. Hunter's color value of the dried persimmon of different size is shown in Table 3. S-4 possessed the highest (36.5) value for lightness and yellowness (26.2) while S-1 scored the highest value for redness (16.0). Fruit color changes to yellow or red according to kind and amount of carotenoid as persimmon matures and dried persimmon surface possesses more redness than unripe fruit (Kim et al., 1986). The highest tannin content was found in S-3 (0.1%) followed by S-4 (0.09%) (Table 3).
Tannins are one of the important bioactive molecules present in persimmon (Ahn et al., 2002; Vázquez-Gutiérrez et al., 2013). In the present study, bigger fruits were found to contain higher amount of tannins as compared to smaller ones. Results of the present study also agree with those of Roby et al. (2004) who also found higher tannin content in bigger sized wine grapes.
Different size of persimmon fruits varied in their physicochemical characteristics. Weight loss due to drying was high in smaller size fruits. Hardness value was high for bigger size fruits. However, soluble solid content was not found to be affected by fruit size.
Tannins, which are one of the vital bioactive molecules in persimmon, were higher in bigger fruits than in smaller ones. Results of the present study provide information about selection of proper sized fruits for drying. Overall results of the present study indicate that, physicochemical characteristics of dried persimmon fruit are affected by the fruit size.
The authors have not declared any conflict of interests.
REFERENCES
|
Achiwa Y, Hibasami H, Katsuzaki H, Imai K, Komiya T (1997). Inhibitory effects of persimmon (Diospyros kaki) extract and related polyphenol compounds on growth of human lymphoid leukaemia cells. Biosci. Biotech. Biochem. 61:1099-1101.
Crossref
|
|
|
|
Ahn HS, Jeon TI, Lee JY, Hwang SG, Lim Y, Park DK (2002). Antioxidative activity of persimmon and grape seed extract: In vitro and in vivo. Nutr. Res. 22:1265-1273.
Crossref
|
|
|
|
Akyıldız A, Aksay S, Benli H, Kıroğlu F, Fenercioğlu H (2004). Determination of changes in some characteristics of persimmon during dehydration at different temperatures. J. Food Eng. 65:95-99.
Crossref
|
|
|
|
AOAC (1995). Official Methods of Analysis of AOAC International. 16th ed. Method 950.46, Arlington, Virginia, USA: Association of Official Analytical Chemists.
|
|
|
|
Blažková J, HlušiÄková I, Blažek J (2002). Fruit weight, firmness and soluble solids content during ripening of Karešova cv. sweet cherry. Hort. Sci. (Prague). 29:92-98.
|
|
|
|
Butt MS, Sultan MT, Aziz M, Naz A, Ahmed W, Kumar N, Imran M (2015). Persimmon (Diospyros kaki) fruit: hidden phytochemicals and health claims. EXCLI J. 14: 542-561.
|
|
|
|
Byrne DH (2002). Peach breeding trends: a worldwide perspective. Acta Hortic. 592:49-59.
Crossref
|
|
|
|
Dehydration (1998). In. Encyclopaedia Britannica Online [online] Retrieved from https://global.britannica.com/topic/dehydration-food-preservation [15 December, 2016].
|
|
|
|
Fu L, Lu W, Zhou X (2016). Phenolic Compounds and In Vitro Antibacterial and Antioxidant Activities of Three Tropic Fruits: Persimmon, Guava, and Sweetsop. BioMed Res. Int. 2016. Article ID 4287461.
Crossref
|
|
|
|
Graham HN (1992). Green tea composition, consumption and polyphenol chemistry. Prev. Med. 21(3):334-350
Crossref
|
|
|
|
Kim ID, Lee JW, Kim SJ, Cho JW, Dhungana SK, Lim YS, Shin DH (2014). Exogenous application of natural extracts of persimmon (Diospyros kaki Thunb.) can help in maintaining nutritional and mineral composition of dried persimmon. Afr. J. Biotechnol. 3:2231-2239.
|
|
|
|
Kim JH, Kim JC, Ko GC (1986). Orchard Horticulture Outline, Hyangmoonsa, Korea. Pp. 220-229.
|
|
|
|
Lia C, Zou B, Dong X, Zhang Y, Du J (2013). Current progress on structure analysis and health benefits of persimmon tannin. Acta Hortic. 996:455-466.
Crossref
|
|
|
|
MacGregor W (2005). Effects of air velocity, air temperature, and berry diameter on wild blueberry drying. Dry. Technol. 23:387-396.
Crossref
|
|
|
|
Marques LG, Silveira AM, Freire JT (2006). Freeze-drying characteristics of tropical fruits. Dry. Technol. 24:457-463.
Crossref
|
|
|
|
Nicoleti JF, Silveira V, Tlis-Romero J, Telis VRN (2007). Influence of drying conditions on ascorbic acid during convective drying of whole persimmons. Dry. Technol. 25:891-899.
Crossref
|
|
|
|
Park YS (1999). Carbon dioxide-induced flesh browning development as related to phenolic metabolism in 'Niitaka'pear during storage. J. Korean Soc. Hortic. Sci. 40:567-570.
|
|
|
|
Price KR, Rhodes MJC, Barnes KA (1988). Flavonol glycoside content and composition of tea infusions made from commercially available teas and tea products. J. Agric. Food Chem. 46:2547-2522.
|
|
|
|
Rao AV, Rao LG (2007). Carotenoids and human health. Pharm. Res. 55:207-216.
Crossref
|
|
|
|
Roby G, Harbertson JF, Adams DA, Matthews MA (2004). Berry size and vine water deficits as factors in winegrape composition: anthocyanins and tannins. Aust. J. Grape Wine Res. 10:100-107.
Crossref
|
|
|
|
Shahkoomahally S, Ramezanian A, Farahnaky A (2015). Postharvest nitric oxide treatment of persimmon (Diospyros kaki L.) improves fruit quality during storage. Fruits 70:63-68.
Crossref
|
|
|
|
Shi J, Pan Z, McHugh TH, Wood D, Zhu Y, Avenaâ€Bustillos RJ, Hirschberg E (2008). Effect of berry size and sodium hydroxide pretreatment on the drying characteristics of blueberries under infrared radiation heating. J. Food Sci. 73:E259-E265.
Crossref
|
|
|
|
Tittonell P, De Grazia J, Chiesa A (2001). Effect of nitrogen fertilization and plant population during growth on lettuce (Lactuca sativa L.) postharvest quality. In. IV International Conference on Postharvest Science. 553:67-68.
Crossref
|
|
|
|
Uchida S, Ozaki M, Akashi T, Ysmsdhts K, Niwa M, Taniyama K (1995). Effects of (-)-epigallocatechin-3-o-gallate (green tea tannin) on the life span of stoke-prone spontaneously hypertensive rates. Clin. Exp. Pharmacol. Physiol. 1:302-303.
Crossref
|
|
|
|
Van Nieuwenhuijzen NH, Zareifard MR, Ramaswamy HS (2001). Osmotic drying kinetics of cylindrical apple slices of different sizes. Dry. Technol. 19(3-4):525-545.
Crossref
|
|
|
|
Vázquez-Gutiérrez JL, Hernando I, Quiles A (2013). Changes in tannin solubility and microstructure of high hydrostatic pressure–treated persimmon cubes during storage at 4° C. Eur. Food Res. Technol. 237:9-17.
Crossref
|
|
|
|
Xie C, Xie Z, Xu X, Yang D (2015). Persimmon (Diospyros kaki L.) leaves: A review on traditional uses, phytochemistry and pharmacological properties. J. Ethnopharmacol. 163:229-240.
Crossref
|
|
|
|
Zhang K, Zhang Y, Zhang M, Gu L, Liu Z, Jia J, Chen X (2016). Effects of phospholipid complexes of total flavonoids from Persimmon (Diospyros kaki L.) leaves on experimental atherosclerosis rats. J. Ethnopharmacol. 191:245-253.
Crossref
|