ABSTRACT
Terfezia claveryi is a species that belongs to the genera of Terfeziaceae or desert truffles, which is a family of truffles. In the present study, silver nanoparticles were synthesized from aqueous extract of T. claveryi which are in the range of 25 to 60 nm. The synthesized nanoparticles were characterized by ultraviolet-visible (UV-Vis) spectroscopy, fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), scanning electron microscopy (FESEM) and transmission electron microscopy (TEM). The effect of the silver nanoparticles on human breast cancer cell line has been tested. Peak absorption was recorded at 440 nm in UV-Vis spectra of silver nanoparticles. The XRD data reports that the silver nanoparticles are crystalline in nature and have face centered cubic geometry. FESEM showed the size range of synthesized silver nanoparticles as 25 to 50 nm. The TEM image represents that the majority of silver nanoparticles are in spherical shape with sizes ranging between 40 and 60 nm. The aim of the present study was to report for the first time fruit mediated synthesis of silver nanoparticles using the extract of T. claveryi and showed remarkable cytotoxicity activity against human breast MCF-7 cancer cell line.
Key words: Silver nanoparticles, Terfezia claveryi, fourier transform infrared spectroscopy (FT-IR), X-ray diffraction (XRD), scanning electron microscopy (FESEM), transmission electron microscopy (TEM), MCF-7 cancer cell line.
Truffles which are also called black diamond are a group of desert fungi, which grow in the northern part of Saudi Arabia bordering Kuwait, Iraq and Jordan (Hussain and Al-Ruqaie, 1999). These are rich in antioxidants such as vitamin A, vitamin C and β carotene (Murcia et al., 2002) and are used as convalescent (Janakat and Nassar, 2010). Especially, Terfezia claveryi species are rich in carbohydrates and proteins (Bokhary and Parvez, 1993)
and are commonly used as culinary agent. They look spherical in shape and pale brown in color. Antibacterial characters of T. claveryi were already studied as its aqueous extract inhibited the growth of P. aeruginosa by Janakat et al. (2004) and Aldebasi et al. (2013). Comparative pathological studies on the healing effect of natural (Terfezia claveryi) and synthetic (Vigamox) antimicrobials (Aldebasi et al., 2012) and in-vivo effect of T. claveryi extract on corneal ulcer of rabbit’s eye (Aldebasi et al., 2015). The aqueous extract of T. claveryi is hepatoprotective (Janakat and Nassar, 2010). The truffles are rich in flavonoids (Akyuz, 2013) which can be exploited in reducing AgNO3 to Ag0. The compounds of truffle aqueous extract have important therapeutic roles: anti-inflammatory, anti-carcinogenic, anti-mutagenic, immunesuppressor and anti-microbial properties (Hannan et al., 1989). As the T. claveryi is rich in proteins, the reducing activity of the amino acids can be applied for the green synthesis of silver nanoparticles by reducing AgNO3 to Ag0 (Aldebasi et al., 2014). There are different approaches for the synthesis of silver nanoparticles which include physical, chemical and biological approaches. Among all, biological approach is well preferred because of its eco-friendly, cheap and time saving factors. Nanoparticles are used in paints, waste water treatment (Tiwari et al., 2008), and drug delivery. Particularly in life sciences, nanoparticles have a great importance in drug delivery, gene delivery, photo-dynamics, imaging (MRI) and in vitro diagnostics (De Jong and Borm, 2008). The credit of nanoparticles is its non-toxicity which allows them to be used in drug/gene delivery when compared to traditionally method using chemical agents. Presently, silver nanoparticles are applied in diagnostic process in biosensors for quantitative detection (Majdalawieh et al., 2014; Li and Xu, 2014), antibacterial applications in wound dressings and cosmetics, conductive applications in conductive inks, optical applications in metal enhanced fluorescence (MEF) and surface enhanced Raman scattering (SERS). Also, silver nanoparticles can be applicable in the water filters to filter out pathogen free water, in the enhancement of latent finger prints (Sametband et al., 2007) and catalytic degradation of organic dyes (Vidhu and Philip, 2014).
Cancer is a major health problem and it arises from one single cell. According to WHO, if it continue rising without any immediate action, 13.1 million people may die in 2030. Tobacco use, alcohol use, lack of physical activity, low intake of fruit and vegetable are some of the important risk factors; the reason for 30% of worldwide cancer deaths. Diagnosis of tumors in the human body was very difficult (Gurunathan et al., 2013) at their earlier stage and there was a search of new treatment for treating this deadly disease. Radiotherapy, chemotherapy and surgery are some of the cancer treatments which are used to improve the patient’s life. Recently, nanoparticles are also used to overcome this problem. The nanoscale devices can easily enter the cells and they made an interaction with DNA, proteins, enzymes and cell receptors. The nanoparticles can detect the cancer disease in a very small volume of cells or tissue (Berrington and Lall, 2012). This study is focused on the cytotoxicity of silver nanoparticles on cultured MCF-7 cell line using different concentrations.
The present study was aimed at reporting for the first time fruit mediated synthesis of silver nanoparticles using the extract of T. claveryi. The optical absorption spectrum of synthesized silver nanoparticles is recorded by using UV-visible spectrophotometer. Morphological characterizations are performed using XRD, SEM and TEM. The spherical shaped silver nanoparticles showed excellent cytoxicity against MCF-7 human breast cancer cell lines.
Preparation of extract
Desert truffles (T. claveryi) were collected from Buraidah market, Al-Qassim region Kingdom of Saudi Arabia. The collected fruits were air dried and stored at cool temperature and used when needed. 10 g of fruit was mixed with 150 ml of distilled water and boiled for 8 to 10 min. After cooling, mixture was centrifuged at 5000 rpm for 10 min and the supernatant was collected for Ag nanoparticle synthesis.
Biosynthesis of silver nanoparticles
Fruit (10 g) was mixed with 150 ml of distilled water and boiled for 8 to 10 min and filtered through Whatman No.1 filter paper (pore size 25 μm). The filtrate was further filtered through 0.6 μm sized filters. The solution was decanted and stored at 4ËšC; it was used within a week of its preparation. 1 mM aqueous solution of silver nitrate (AgNO3) was prepared and used for the synthesis of silver nanoparticles. 10 ml of extract was added into 90 ml of aqueous solution of 1 mM silver nitrate for bioreduction of Ag+ ions in the solution and kept at room temperature for 24 h. The bio-reduction was analyzed by drawing the absorption maxima at 360 to 700 nm using a uv-vis spectrophotometer.
Characterization of Ag nanoparticles
Color change to brown color confirmed the synthesis of silver nanoparticles and were characterized by uv-visible spectroscopy (Thermo Scientific Evolution 201), FESEM ((SUPRA 55)-CARL ZEISS, Germany), XRD (XRD-SMART LAB (9kW)-RIKAGU, JAPAN), TEM (Hitachi H-7500 TEM, Japan) and fourier transforms infrared spectroscopy (FTIR-PERKIN ELMER Spectrum Two model, UK).
Determination of in vitro anticancer activity of synthesized AgNPs
Cell culture
Breast cancer cell line (MCF-7) was obtained from National Centre for Cell Science (NCCS), Pune, India. The MCF-7 cells were grown as monolayer in MEM, supplemented with 10% FBS, 1% glutamine, and 100 U/ml penicillin-streptomycin and incubated at 37°C in 5% CO2 atmosphere. Stocks were maintained in 75 cm2 tissue culture flask.
Measurement of cytomorphological changes in MCF-7
MCF-7 cells were pre-treated with different concentration of synthesized AgNPs and incubated for 24 h at 37°C in 5% CO2 atmosphere. After the incubation of cells, the gross morphological changes in the cells were observed under bright field microscope.
UV-Vis spectrum
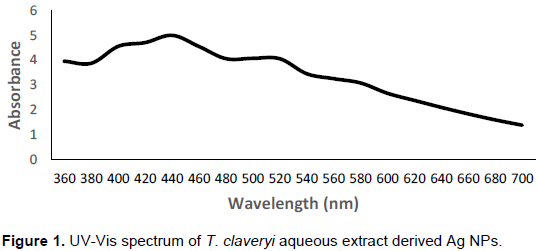
The synthesized silver nanoparticles maximum absorption range was measured using UV-visible spectrophotometry. The strong resonance for T. claveryi derived silver nanoparticles was clearly observed at 440 nm (Figure 1). The resultant is due to reduction of silver nitrate into silver and which suggests the presence of silver nanoparticles.
Fourier transform infra-red studies
FTIR spectrum of the synthesized AgNPs is shown in Figure 3 which reveals the possible biomolecules present in the fruit extract which is accountable for the reduction of silver ions and its interaction with the AgNPs.
XRD analysis
XRD studies were carried out to study the crystalline characteristics of the T. claveryi derived silver nanoparticles. The comparison between the standard and T. claveryi derived Ag nanoparticles confirms the crystalline nature of silver nanoparticles as evidenced by the peaks observed at 2θ values of 38.035, 44.21, 46.24, 64.39 and 77.21° corresponding to 111, 200, 220 and 311 Bragg reflections, respectively. The XRD pattern of Ag NPs is shown in Figure 2. The average size of Ag nanoparticles was calculated using XRD data and Scherrer equation and approximately found to be 20 nm. The Scherrer equation followed is, D = Kλ/ β cosθ where, D is crystallite size of Ag nanoparticles, λ is the
wavelength of the X-ray source (1.54056 nm) used in XRD, β is the width at the half maximum of the diffraction peak, θ is the Bragg angle and K is the Scherrer constant (0.94).
Scanning electron microscope (SEM)-EDX
Figure 4a depicts the FESEM image of silver nanoparticles. The size range of synthesized silver nanoparticles was found to be 25 to 50 nm. The silver nanoparticles showed spherical morphology under FESEM observation. The EDX characterization (Figure 4b) has shown absorption of strong silver signal along with other elements, which may originate from the biomolecules that are bound to the surface of silver nanoparticles.
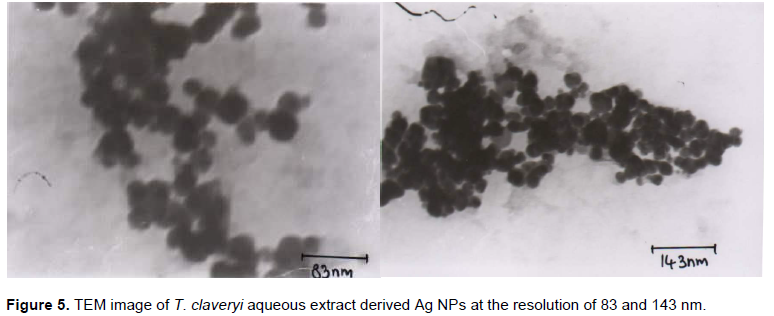
Transmission electron microscope (TEM)
Thin film of sample were prepared on a carbon coated copper grid by just dropping a small amount of sample on the grid and drying it under mercury lamp for 5 min. The TEM image of T. claveryi derived Ag nanoparticles shows that the majority of NPs are spherical shaped as presented in Figure 5. Also, TEM image at resolution of 83 nm represents that the Ag NPs size ranges between 40 and 60 nm.
Effect of silver nanoparticles against MCF-7 breast cancer cells
In vitro cytotoxicity of the silver nanoparticles was evaluated against MCF-7 breast cancer cells at different concentrations (10 to 50 g/ml). Our results unveils, that there is direct dose-response relationship with the tested cells at higher concentrations. In relation to cell death, a minimum of 10 g/ml of silver nanoparticles is well enough to induce 50% of cell mortality; shows the cytotoxicity of silver nanoparticles at various time intervals. The cell viability of the silver nanoparticles at different incubation time is depicted by Figure 6. The calculated IC50 value of this experiment is 10 mg/ml concentrations. Previously, synthesized AgNPs inducing cytotoxicity were discussed by (Safaepour et al., 2009; Sriram et al., 2010). However, there is no previous investigation data available on T. claveryi carrying nanoparticles.
Silver nanoparticles were synthesized from T. claveryi aqueous extract whose size falls in the range of 25 to 60 nm. It is convincing here that the aspartic acid, glutamic acid and other amino acids acted as reducing agents to convert AgNO3 to Ag° nanoparticles. In fact, T. claveryi is rich in amino acids and also truffles are rich in flavonoids. Initially, the plasmonpeakinUV-Visspectraof440nm confirmed the presence of silver nanoparticles, and the XRD derived crystal nature of Ag nanoparticles as face centered cubic. FESEM and TEM images suggest that the silver nanoparticles are in spherical shape. The present study shows significant cytotoxic effects by synthesized silver nanoparticles against MCF-7 breast cancer cells. The method could be exploited for developing economical biosynthesis of Ag nanoparticles in large scales and it would be useful in future nanomedicine.
The authors have not declared any conflict of interests.
REFERENCES
|
Akyuz M (2013). Nutritive value, flavonoid content and radical savenging activity of the truffle (Terfezia boudieri Chatin). J. Soil Sci. Plant Nutr. 13(1):143-151.
|
|
|
|
Aldebasi YH, Aly SM, Qureshi MA, Khadri H (2013). Novel antibacterial activity of Terfizia claveryi aqueous extract against clinical isolates of corneal ulcer. Afr. J. Biotechnol. 12:6340-6346.
Crossref
|
|
|
|
|
Aldebasi YH, Aly SM, Qureshi MA, Khan A (2015). Therapeutic Implication of Terfezia claveryi Extract on Corneal Ulcer of Rabbit's Eye. Br. J. Med. Med. Res. 8(10):863-873.
Crossref
|
|
|
|
|
Aldebasi YH, Aly SM, Riazunnisa K, Khadri H (2014). Noble silver nanoparticles (AgNPs) synthesis and characterization of fig-leaf (Ficus carica) extract and its antimicrobial effect against clinical isolates from corneal ulcer. Afr. J. Biotechnol. 13(45):4275-4281.
Crossref
|
|
|
|
|
Aldebasi YH, Wael GN, Abdel A, Mounir MS, Aly SM, Qureshi MA (2012). Comparative pathological studies on the healing effect of natural (Terfezia claveryi) and Synthetic (Vigamox) Antimicrobials. J. Pharm. Biomed. Sci. 2(6)66-77.
|
|
|
|
|
Berrington N, Lall N (2012). Anticancer Activity of Certain Herbs and Spices on the Cervical Epithelial Carcinoma (HeLa) Cell Line. Evid. Based Complement. Alternat. Med. 2012:564927.
Crossref
|
|
|
|
|
Bokhary HA, Parvez S (1993). Chemical composition of desert truffles Terfezia claveryi. J. Food Comp. Anal. 6:285-293.
Crossref
|
|
|
|
|
De Jong WH, Borm WH (2008). Drug delivery and nanoparticles: Applications and hazards. Int. J. Nanomed. 3(2):133-149.
Crossref
|
|
|
|
|
Gurunathan S, Han JW, Eppakayala V, Jeyaraj M, Kim J-H (2013) Cytotoxicity of Biologically Synthesized Silver Nanoparticles in MDA-MB-231 Human Breast Cancer Cells. BioMed Res. Intern. Article ID 535796, 10p.
|
|
|
|
|
Hannan MA, Al-Dakan A, Aboul-Enein H, Al-Othaimeen A (1989). Mutagenic and antimutagenic factor(s) extracted from desert mushroom using different solvents. Mutagenesis 4:111-114.
Crossref
|
|
|
|
|
Hussain G, Al-Ruqaie IM (1999). Occurrence, chemical composition, and nutritional value of truffles: an overview. Paki. J. Biol. Sci. 2(2):510-514.
Crossref
|
|
|
|
|
Janakat S, Al-Fakhiri S, Sallal AK (2004). A promosing peptide antibiotic from Terfezia claveryi aqueous extract against Staphylococcus aureus in vitro. Phytother. Res. 18:810-813.
Crossref
|
|
|
|
|
Janakat S, Nassar M (2010). Hepatoprotective activity of desert truffle (Terfezia claveryi) in comparision with the effect of Nigella sativa in the rat. Pak. J. Nutr. 9(1):52-56.
Crossref
|
|
|
|
|
Li H, Xu D (2014). Silver nanoparticles as labels for applications in bioassays. Trends Anal. Chem. 61:67-73.
Crossref
|
|
|
|
|
Majdalawieh A, Kanan MC, El KO, Kanan SM (2014). Recent advances in gold and silver nanoparticles: synthesis and applications. J. Nanosci. Nanotechnol. 14(24):4757-4780.
Crossref
|
|
|
|
|
Murcia MA, Martínez-Tomé M, Jiménez AM, Vera AM, Honrubia M, Parras P (2002). Antioxidant activity of edible fungi (Truffles and Mushrooms): Losses during industrial processing. J. Food Prot. 10(9):1614-1622.
Crossref
|
|
|
|
|
Safaepour M, Shahverdi AR, Shahverdi HR, Khorramizadeh MR, Gohari AR (2009). Green synthesis of small silver nanoparticles using geraniol and its cytotoxicity against Fibrosarcoma-Wehi 164," Avicenna J. Med Biotechnol. 1(2):111-115.
|
|
|
|
|
Sametband M, Shweky I, Banin U, Mandler D, Almog J (2007). Application of nanoparticles for the enhancement of latent fingerprints. Chem. Commun. 11:1142-1144.
Crossref
|
|
|
|
|
Sriram MI, Kanth SB, Kalishwaralal K, Gurunathan S (2010). Antitumor activity of silver nanoparticles in Dalton's lymphoma ascites tumor model. Int. J. Nanomed. 5(1):753-762.
|
|
|
|
|
Tiwari DK, Behari K, Sen P (2008). Application of nanoparticles in waste water treatment. World Appl. Sci. J. 3(3):417-433.
|
|
|
|
|
Vidhu VK, Philip D (2014). Catalytic degradation of organic dyes using biosynthesised silver nanoparticles. Micron 56:54-62.
Crossref
|
|
|
|