Full Length Research Paper
ABSTRACT
INTRODUCTION
MATERIALS AND METHODS
RESULTS
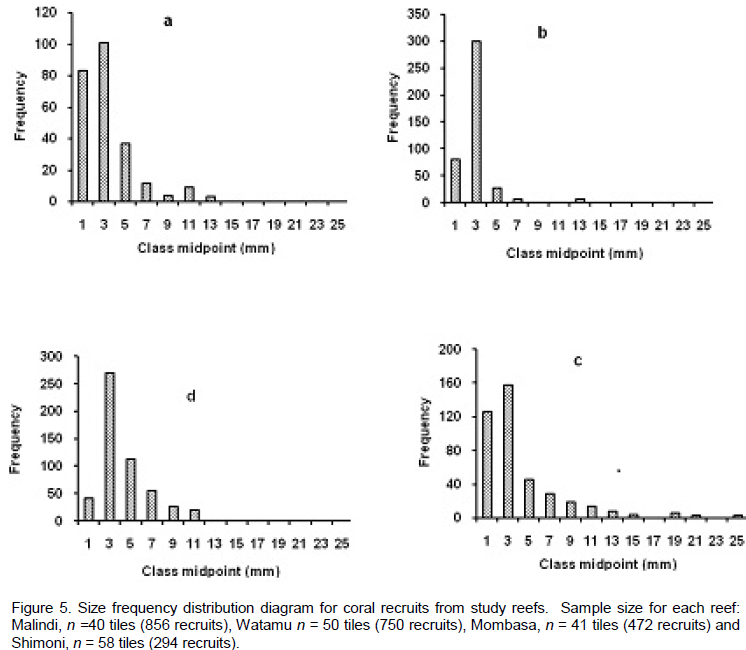
DISCUSSION
CONFLICT OF INTERESTS
The authors did not declare any conflict of interest.
ACKNOWLEDGEMENTS
This research was supported by grants from the British Ecological Society (BES), the World Wildlife Fund (WWF) and the International Development Research Centre (IDRC) Doctoral Research Award and the Simon Fraser University (SFU) President’s Award to the first author. Logistic support was provided by KWS rangers and laboratory and field assistance from the staff of Kenya Marine and Fisheries Research Institute (KMFRI). Excellent fieldwork assistance was provided by Masudi J. Zamu and Anthony M. Nzioka. We also greatly appreciate the reviewers’ comments that helped in improving this manuscript.
REFERENCES
|
Airoldi L, Virgilio M (1998). Responses of turf-forming algae to spatial variations in deposition of sediments. Mar. Ecol. Prog. Ser. 165:271-282. |
|
|
|
|
|
Arnold SN, Steneck RS, Mumby PJ (2010). Running the gauntlet: inhibitory effects of algal turfs on the processes of coral recruitment. Mar. Ecol. Prog. Ser. 414:91-105. |
|
|
|
|
|
Baker AC, McClanahan TR, Starger CJ, Boonstra RK (2013). Long-term monitoring of algal symbiont communities in corals reveals stability is taxon dependent and driven by site-specific thermal regime. Mar. Ecol. Prog. Ser. 479:85-97. |
|
|
|
|
|
Belleveau SA, Paul VJ (2002). Effects of herbivory and nutrients on the early colonization of crustose coralline and fleshy algae. Mar. Ecol. Prog. Ser. 232:105-111. |
|
|
|
|
|
Bellwood DR, Hughes TP, Folke C, Nyström M (2004). Confronting the coral reef crisis. Nature 429:827:833. |
|
|
|
|
|
Birrell CL, McCook LJ, Willis BL, Diaz-Pulido GA (2008). Effects of benthic algae on the replenishment of corals and the implications for the resilience of coral reefs. Oceanogr. Mar. Biol.: Ann. Rev. 46:25-63. |
|
|
|
|
|
Birrell, CL, McCook LJ, Willis BL (2005). Effects of algal turfs and sediment on coral settlement Mar. Pollut. Bull. 51:408-414 |
|
|
|
|
|
Bobcock RC, Smith L (2002). Effects of sedimentation on coral settlement and survivorship. Proc. 9th Int. Coral Reef Symp. 1:245-248. |
|
|
|
|
|
Brakel W (1984). Seasonal dynamics in suspended sediments plumes from the Tana and Sabaki rivers, Kenya: analysis of landsat imagery. Remote Sens. Environ. 16:165-173. |
|
|
|
|
|
Bruno JF, Petes L, Drew E, Harvell C, Hettinger A (2003). Nutrient enrichment can increase the severity of coral diseases. Ecol. Lett. 6(12):1056-1061. |
|
|
|
|
|
Burke L, Maidens J (2004). Reefs at Risk, World Resource Institute, Washington DC, 81 p. |
|
|
|
|
|
Carreiro-Silva M, Kiene WE, Golubic S, McClanahan TR (2012). Phosphorus and nitrogen effects on microbial euendolithic communities and their bioerosion rates. Mar. Pollut. Bull. 64:602-613. |
|
|
|
|
|
Casey JM, Choat JH, Connolly SR (2015). Coupled dynamics of territorial damselfishes and juvenile corals on the reef crest. Coral Reefs 34(1):1-11. |
|
|
|
|
|
Christiansen C, Gertz FM, Laima JC, Lund-Hansen LC, Vang T, Jürgensen C (1996). Nutrient (P, N) dynamics in the southwestern Kattegat, Scandinavia: sedimentation and resuspension effects. Environ. Geol. 29:66-77. |
|
|
|
|
|
Cornell HV, Karlson RH (2000). Coral species richness; ecological versus biogeographical influences. Coral Reefs 19:37-49. |
|
|
|
|
|
Darling ES, Alvarez-Filip L, Oliver TA, McClanahan TR, Cote IM (2012). Evaluating life history strategies of reef corals from species traits. Ecol. Lett 15:1378-1386. |
|
|
|
|
|
Darling ES, McClanahan TR, Corte IM (2013). Life histories predict coral community disassembly under multiple stressors. Glob. Chang. Biol. 19:1930-1940. |
|
|
|
|
|
De'ath, Fabricius KE, Sweatman H, Puotinen M (2012). The 27-year decline of coral cover on the Great Barrier Reef and its causes. Proc. Nat Acad. Sci. USA, 109:17995-17999. |
|
|
|
|
|
Devlin MJ, Fabricius KE (2005). Effects of terrestrial runoff on the ecology of corals and coral reefs: review and synthesis. Mar. Pollut. Bull. 50:125-14. |
|
|
|
|
|
Diaz-Pulido G, Garzón-Ferreira J (2002). Seasonality in algal assemblages on upwelling-influenced coral reefs in the Colombian Caribbean Bot. Marina 45:284-292. |
|
|
|
|
|
Diaz-Pulido G, Gouezo M, Tilbrook B, Dove S, Anthony KRN (2011). High CO2 enhances the competitive strength of seaweeds over corals. Ecol. Lett. 14:156-162. |
|
|
|
|
|
Diaz-Pulido G, Harii S, McCook LJ, Hoegh-Guldberg O (2010). The impact of benthic algae on the settlement of a reef-building coral. Coral Reefs, 29(1):203-208. |
|
|
|
|
|
Diaz-Pulido G, McCook LJ (2003). Relative roles of herbivory and nutrients in the recruitment of coral-reef seaweeds. Ecology 84:2026-2033. |
|
|
|
|
|
Doropoulos C, Ward S, Diaz-Pulido D, Hoegh-Guldberg O, Mumby P J (2012). Ocean acidification reduces coral recruitment by disrupting intimate larval-algal settlement interactions. Ecol. Lett.15: 338–346. |
|
|
|
|
|
Dunn JG, Sammarco PW, LaFleur Jr G (2012). Effects of phosphate on growth and skeletal density in the scleractinian coral Acropora muricata: A controlled experimental approach J. Exp. Mar. Biol. Eco. 41: 34-44. |
|
|
|
|
|
Edmunds PJ, Carpenter RC (2001). Recovery of Diadema antillarum reduces macroalgal cover and increases abundance of juvenile corals on a Caribbean reef. Proc. Nat. Acad. Sci. 98(9): 5067-5071. |
|
|
|
|
|
Fabricius KE (2005). Effects of terrestrial runoff on the ecology of corals and coral reefs: review and synthesis. Ma.r Pollut. Bull. 50: 125-146. |
|
|
|
|
|
Fabricius KE, De'ath G (2001). Environmental factors associated with the spatial distribution of crustose coralline algae on the Great Barrier Reef. Coral Reefs 19: 303-309. |
|
|
|
|
|
Fabricius KE, McCorry D (2006). Changes in octocoral communities and benthic cover along a water quality gradient in the reefs of Hong Kong. Mar. Pollut. Bull. 52:22-33. |
|
|
|
|
|
Fleitmann D, Dunbar RB, McCulloch M, Mudelsee M, Vuille M, McClanahan TR, Cole JE, Eggins E (2007) East Africa soil erosion recorded in a 300 year old coral colony from Kenya. Geophys. Res. Lett. 34:L04401, doi:10.1029/2006GLO028525. |
|
|
|
|
|
Gilmour JP, Smith LD, Heyward AJ, Baird AH, Pratchett MS (2013). Recovery of an isolated coral reef system following severe disturbance. Science 340:69-71. |
|
|
|
|
|
Glassom D, Celliers M, Schleyer MH (2006) Coral recruitment patterns at Sodwana Bay, South Africa. Coral Reefs 25:485-492. |
|
|
|
|
|
Glassom D, Zakai D, Chadwick-Furman NE (2004). Coral recruitment: Spatio-temporal analysis along the coastline of Eilat, northern Red Sea. Mar. Biol. 144:641-651. |
|
|
|
|
|
Godinot C, Tribollet A, Grove R, Ferrier-Pagès C (2012) Bioerosion by euendolith decreases in phosphate-enriched skeletons of living corals. .Biogeosci. Disc. 9:2424-2444 |
|
|
|
|
|
Goreau T, McClanahan TR, Hayes R, Strong A (2000). Conservation of coral reefs after the 1998 global bleaching event. Conserv. Biol. 14: 5-15. |
|
|
|
|
|
Gorgula SK, Connell SD (2004). Expansive covers of turf-forming algae on human-dominated coasts: the relative effects of increasing nutrients and sediment loads. Mar. Biol. 145:613-619. |
|
|
|
|
|
Harrington L, Fabricius FH, De'ath G, Negri A (2004). Recognition and selection of settlement substrata determines post-settlement survival in corals. Ecology 85:3428-3437. |
|
|
|
|
|
Harii S, Kayanne H, Takigawa H, Hayashibara T, Yamamoto M (2002). Larval survivorship, competency periods and settlement of two brooding corals, Heliopora coerulea and Pocillopora damicornis. Mar. Biol. 141:39-46. |
|
|
|
|
|
Harriot VJ, Banks SA (1995). Recruitment of sclectinian corals in Solitary Islans Marine Reserve, a high latitude coral-dominated community in Eastern Australia. Mar. Ecol. Prog. Ser. 123: 155-161. |
|
|
|
|
|
Harrison PL, Booth DJ (2007). Coral reefs: naturally dynamic and increasingly disturbed ecosystems. Mar. Ecol. Chapter 13: 316-377. |
|
|
|
|
|
Hoegh-Guldberg O, Mumby PJ, Hooten AJ, Steneck RS, Greenfiled P, Gomez E, Harvell CD, Sale PF, Edwards A J, Caldeira K, Knowlton N, Eakin CM, Iglesias-Prieto R, Muthiga N, Bradbury RH, Dubi A, Hatziolos ME (2007). Coral Reefs under rapid climatic change and ocean acidification. Science 318: 1737-1742. |
|
|
|
|
|
Hughes TP, Graham NA, Jackson JB, Mumby PJ, Steneck RS (2010). Rising to the challenge of sustaining coral reef resilience. Trends Ecol. Evol. 25(11):633-642. |
|
|
|
|
|
Hughes TP, Tanner JE (2000). Recruitment failure, life histories and long term decline of Caribbean coral reefs. Ecololgy 81: 2250-2263. |
|
|
|
|
|
Irving AD, Connell SD (2002) Interactive effects of sediment and microtopography on the abundance of sub-tidal turf-forming algae Phycologia 41:517-522 |
|
|
|
|
|
Karisa JF, Kaunda-Arara B, Obura DO (2008). Spatial and temporal variation in coral recruitment and mortality in coastal Kenya. In Obura DO, Tamelander J, Lindahl O (eds.) Coastal Ocean Research and Development in the Indian Ocean (CORDIO). |
|
|
Katwijk MM, Meier NF, van Loon R, van Hove EM, Giesen WBJT, van der Velde G, den Hartog C (1993). Sabaki River sediment load and coral stress: correlation between sediments and condition of the Malindi-Watamu reefs in Kenya (Indian Ocean). Mar. Biol. 117: 675-683. |
|
|
|
|
|
Kayanne H, Iijima H, Nakamura N, McClanahan TR, Behera S, Yamagata Y (2006). Indian Ocean Dipole index recorded in Kenyan coral annual density bands. Geophys. Res. Lett. 33. |
|
|
|
|
|
Kendrick GA (1991). Recruitment of coralline crusts and filamentous turf algae in Galapagos archipelago: effects of sediment scour, erosion and accretion. J. Exp. Mar. Biol. Ecol. 147: 47-63 |
|
|
|
|
|
Kitheka JU, Nthenge P, Obiero M (2003). Sabaki estuary sediment transport dynamics and resultant sediment fluxes in the monsoon-driven Malindi Bay, Kenya. LOICZ-START AFRICAT Project Report-II-Sabaki p 1-57. |
|
|
|
|
|
Kuffner IB, Walters LJ, Becerro MA., Paul VJ, Ritson-Williams R, Beach KS (2006). Inhibition of coral recruitment by macroalgae and cyanobacteria. Mar. Ecol. Prog. Ser. 323:107-117. |
|
|
|
|
|
Kuta K, Richardson L (2002). Ecological aspects of black band disease of corals: relationships between disease incidence and environmental factors. Coral Reefs 21(4):393-398. |
|
|
|
|
|
Lirman D, Fong P (2007). Is proximity to land-based sources of coral stressors an appropriate measure of risk to coral reefs? An example from the Florida Reef Tract. Mar. Pollut. Bull. 54:779-791. |
|
|
|
|
|
Logan CA, Dunne JP, Eakin CM, Donner SD (2014). Incorporating adaptive responses into future projections of coral bleaching. Glob. Chang. Biol. 20:125-139. |
|
|
|
|
|
Maina J, McClanahan TR, Venus V, Ateweberhan M, Madin J (2011). Global gradients of coral exposure to environmental stresses and implications for local management. PLoS ONE, 6: e23064. |
|
|
|
|
|
Mangubhai S, Harrison PL, Obura DO (2007). Patterns of larval settlement on lagoon reefs in the Mombasa Marine National Park and Reserve, Kenya. Mar. Ecol. Prog. Ser. 348:149-159. |
|
|
|
|
|
Madsen JD, Chambers PA, James WF, Koch EW, Westlake DF (2001). The interaction between water movement, sediment dynamics and submersed macrophytes. Hydrobiologia 444:71-84. |
|
|
|
|
|
McClanahan, TR (2014). Decadal coral community reassembly on an African fringing reef. Coral Reefs 33:939-950. |
|
|
|
|
|
McClanahan TR, Obura DO (1997). Sedimentation effects on shallow coral communities in Kenya. J. Exp. Mar. Biol. Ecol. 209:103-122. |
|
|
|
|
|
McClanahan TR (1988). Seasonality in East Africa's coastal waters Mar. Ecol. Prog. Ser. 44(2): 191-199. |
|
|
|
|
|
McClanahan TR, Donner SD, Maynard JA, MacNeil MA, Graham NAJ, Maina J, Baker AC, Beger M, Campbell SJ, Darling ES, Eakin CM, Heron SF, Jupiter SD, Lundquist CJ, McLeod E., Mumby PJ, Paddack M, Selig ER, van Woesik R. (2012). Prioritizing key resilience indicators to support coral reef management in a changing climate. PLoS ONE. 7: e42884. |
|
|
|
|
|
McClanahan TR, Maina J, Starger CJ, Herron-Perez P, Dusek E (2005). Detriments to post-bleaching recovery of corals. Coral Reefs 24: 230-246. |
|
|
|
|
|
McClanahan TR, Maina JM, Muthiga NA (2011). Associations between climate stress and coral reef diversity in the Western Indian Ocean. Glob. Chang. Biol. 17: 2023-2032. |
|
|
|
|
|
McClanahan TR, Nugues M, Mwachireya S (1994). Fish and sea urchin herbivory and competition in Kenyan coral reef lagoons: the role of reef management. J. Exp. Mar. Biol. Ecol. 184:237-254 |
|
|
|
|
|
McClanahan TR, Shafir SH (1990). Causes and consequences of sea urchin abundance and diversity in Kenyan coral reef lagoons. Oecologia 83:362-370. |
|
|
|
|
|
McCook LJ, Jompa J, Diaz-Pulido G (2001). Competition between corals and algae on coral reefs: a review of evidence and mechanisms. Coral Reefs 19: 400-417. |
|
|
|
|
|
Miller MW, Weil E, Szmant AM (2000). Coral recruitment and juvenile mortality as structuring factors for reef benthic communities in Biscayne National Park, USA Coral Reefs 19: 115-123. |
|
|
|
|
|
Nakamura M, Ohki S, Suzuki A, Sakai K (2011). Coral larvae under ocean acidification: survival, metabolism, and metamorphosis. PLoS ONE, 6:e14521. |
|
|
|
|
|
Novak V, Santodomingo N, Rösler A, Di Martino E, Braga JC, Taylor PD, Johnson KG, Renema W (2013). Environmental reconstruction of a late Burdigalian (Miocene) patch reef in deltaic deposits (East Kalimantan, Indonesia). Palaeogeogr. Palaeoclimat. Palaeoecol. 374:110-122. |
|
|
|
|
|
Nozawa Y, Harrison PL (2008). Temporal patterns of larval settlement and survivorship of two broadcast-spawning acroporid corals. Mar. Biol. 155: 347-351. |
|
|
|
|
|
Nugues MM, Roberts CM (2003a). Partial mortality in massive reef corals as an indicator of sediment stress on coral reefs. Mar. Poll. Bull. 46:314-323. |
|
|
|
|
|
Nugues MM, Roberts CM (2003b). Coral mortality and interaction with algae in relation to sedimentation Coral reefs 22(4): 507-516. |
|
|
|
|
|
Nugues MM, Smith GW, van Hooidonk RJ, Seabra MI, Bak RPM (2004). Algal contact as a trigger for coral disease. Ecol. Lett. 7:919-923. |
|
|
|
|
|
Nyström M, Folke C, Morberg F (2000). Coral reef disturbance and resilience in a human-dominated environment. Trend Ecol. Evol. 15: 413-417. |
|
|
|
|
|
Nzali LM, Johnstone RW, Mgaya YD (1998). Factors affecting scleractinian coral recruitment on a nearshore reef in Tanzania. Ambio 25: 717-721. |
|
|
|
|
|
O'Leary J, Potts D, Braga J, McClanahan T R (2012). Indirect consequences of fishing: reduction of coralline algae suppresses juvenile coral abundance. Coral Reefs 31: 547-559. |
|
|
|
|
|
Obura DO (1995). Environmental stress and life history strategies, a case study of corals and river sediment from Malindi Kenya. PhD Thesis, University of Miami p326. |
|
|
|
|
|
Obura DO, Furaha J, Mwaura J (2005). Coral settlement patterns in the Mombasa Marine Park. Coral Reef Degradation in the Indian Ocean (CORDIO) Status Report 2005, 167-173. |
|
|
|
|
|
Ohowa BO (1996) Seasonal variations of nutrient fluxes in the Indian Ocean from the Sabaki River, Kenya. Discov. Innov. 8:265-274. |
|
|
|
|
|
Palumbi SR, Barshis DJ, Traylor-Knowles N, Bay RA (2014). Mechanisms of reef coral resistance to future climate change. Science 344:895-898. |
|
|
|
|
|
Pandolfi JM, Connolly SR, Marshall DJ, Cohen AL (2011). Projecting coral reef futures under global warming and ocean acidification. Science, 333(6041):418-422. |
|
|
|
|
|
Pandolfi JM, Jackson JBC, Baron N, Bradbury RH, Guzman HM, Hughes TP, Kappel CV, Micheli F, Ogden, JC, Possingham HP, Sala E (2005). Are US coral reefs on the slippery slope to slime? Science 307: 1725-1726. |
|
|
|
|
|
Perry CT, Larcombe P (2003). Marginal and non-reef building coral environments Coral Reefs 22:427-432. |
|
|
|
|
|
Richmond RH, Rongo T, Golbuu Y, Victor S, Idechong N, Davis G, Kosta W, Neth S, Hamnett M, Wolanski E (2007). Watersheds and coral reefs: Conservation Science, Policy and Implementation. BioScience 57:598-607. |
|
|
|
|
|
Rodgers CS (1990). Response of coral reefs and reef organisms to sedimentation. Mar. Ecol. Prog. Ser. 62:185-202. |
|
|
|
|
|
Santodomingo N, Novak V, Pretkovic V, Marshall N, Di Martino E, Capelli ELG, Johnson KG (2015). A diverse patch reef from turbid habitats in the middle Miocene (East Kalimantan, Indonesia). Palaios, 30(1):128-149. |
|
|
|
|
|
Selig ER, Casey KS, Bruno JF (2012). Temperature-driven coral decline: the role of marine protected areas. Glob. Chang. Biol., 18: 1561–1570. |
|
|
|
|
|
Shannon CE, Weaver W (1949). The Mathematical Theory of Communication. University of Illinois Press, Urbana, IL, 125 pp. |
|
|
|
|
|
Thomason JC, LeTissier MDA, Thomason PO, Filed SN (2002). Optimizing settlement tiles: the effect of surface texture and energy, orientation and deployment duration upon the fouling community. Biofouling 18:293-304. |
|
|
|
|
|
Thomsen, MS., McGlathery K (2006). Effects of accumulations of sediments and drift algae on recruitment of sessile organisms associated with oyster reefs. J. Exp Mar Biol. Ecol. 328: 22– 34. |
|
|
|
|
|
Todd PA (2008). Morphological plasticity in scleractinean corals. Biol. Rev. 83:315-337. |
|
|
|
|
|
Todd PA, Sanderson PG, Chou LM (2001). Morphological variation in polyps of the scleractinean coral Favia speciosa (Dana) around Singapore. Hydrobiologia 444:227-235. |
|
|
|
|
|
Tribollet A, Golubic S (2011). Reef Bioerosion: Agents and Processes In Z. Dubinsky and N. Stambler (eds) Coral Reefs: An Ecosystem in Transition, Springer Science + Business Media BV. |
|
|
|
|
|
Umar MJ, McCook LJ, Price IR (1998). Effects of sediment deposition on the seaweed Sargassum on a fringing coral reef. Coral Reefs 17:169-177. |
|
|
|
|
|
Venera-Ponton DE, Diaz-Pulido G, McCook LJ, Rangel-Campo A (2011). Macroalgae reduce growth of juvenile corals but protect them from parrotfish damage. Mar. Ecol. Prog. Ser. 421: 109-115. |
|
|
|
|
|
Watson M, Ormond RFG (1994). Effects of an artisanal fishery on the fish and urchin populations of a Kenyan coral reef. Mar. Ecol. Prog. Ser. 109: 115-129. |
|
|
|
|
|
West K, van Woesik R (2001). Spatial and temporal variance of river discharge on Okinawa (Japan): inferring the temporal impact on adjacent coral reefs. Mar. Pollut. Bull. 42:864-872. |
|
|
|
|
|
Wolanski E, Martinez JA, Richmond RH (2009). Quantifying the impact of watershed urbanization on a coral reef: Maunalua Bay, Hawaii. Estuar Coast Shelf Sci 84: 259-268. |
|
|
|
|
|
Xie LQ, Xie P, Tang HJ (2003). Enhancement of dissolved phosphorus release from sediment to lake water by Microcystis blooms—an enclosure experiment in a hyper-eutrophic, subtropical Chinese lake. Environ. Pollut. 122:391-399. |
|
Copyright © 2024 Author(s) retain the copyright of this article.
This article is published under the terms of the Creative Commons Attribution License 4.0