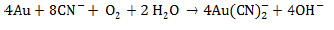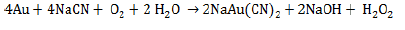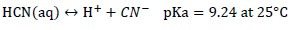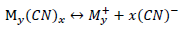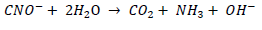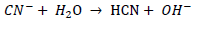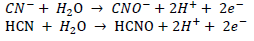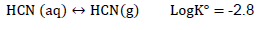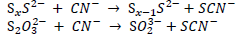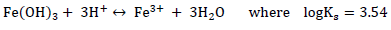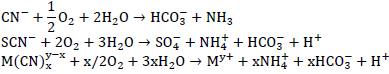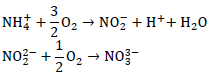Cyanide is a chemical that is widely distributed in the environment, mainly as a result of anthropogenic activities. Only small quantities are naturally produced. Most industrial activities use this chemical compound for manufacturing a product as electroplating or for extracting gold. Exposure to cyanide results in negative health impacts to the wildlife and humans. In nature, cyanide occurs in several species and fates, of which the free cyanide forms are the most toxic ones. Cyanide can be removed by chemical or biological processes. Biological treatment called bioremediation, which is cost-effective and eco-friendly, is the most applied process to remove cyanide from contaminated environments. This technology focused on the use of microorganisms to remove pollutants. Many microorganisms have been reported to transform the cyanide in another less toxic compound, or to consume cyanide for their growth. The reactions are influenced by environmental parameters such as pH and temperature and by the nutriment availability. Bioremediation technologies were few applied in most of African Countries. Future works should focus on how to adapt the bioremediation technologies that already applied in other parts of the World in African context.
Cyanide is used by humans in many cases. Every year, in industry, massive quantities of cyanides are used in metal extraction, electroplating, pesticides, metal hardening, photography, printing, dyeing, and many other manufacturing processes. It is also used in the production of organic chemicals such as nitrile, nylon, and acrylic plastics (Aazam, 2014).
The use of cyanide also facilitates the storage of salt. Potassium ferrocyanide (K4Fe(II)(CN)6) and sodium ferrocyanide (Na4Fe(II)(CN)6) in maximum concentrations of 200 mg kg-1 have been used as anti-clumping additives in road salt in order to facilitate handling and distribution (Ohno, 1990; Kjeldsen, 1999).
Gold mining use
Cyanide is also used in the chemical extraction of gold from low-grade ores by the heap leach process (Kjeldsen, 1999; White and Markwiese, 1994). This is the predominant process in the gold extraction industry that has been applied commercially since 1887 (Adams and Lloyd, 2008). Gold occurs in multi-phase mineralization including base-metal-rich mesothermal and base-metal-poor epithermal assemblages; it is recovered by a combination of amalgamation with mercury and leaching with cyanide (Carling et al., 2013).
The first recorded use of cyanide to extract gold from ores was in 1889 at the Crown Mine in New Zealand (Dorr, 1936; Johnson, 2014). A typical cyanide heap leach process has a series of steps (White and Markwiese, 1994). The gold-bearing ore is extracted, crushed to a nominal size, and piled on a constructed liner. An alkaline cyanide solution is sprayed on the pile. The cyanide solution is buffered to a pH of about 11 to keep cyanide from forming hydrogen cyanide, which can be lost through volatilization. The cyanide in solution complexes with gold (and other metals) extracting the metals from the ore. The cyanide solution is captured by the liner and is recirculated through the pile to further extract cyanide complexes metals (Kjeldsen, 1999). The stoichiometry by which this reaction occurs is known as the Elsner (1846) equation (Jeffrey, 2000):
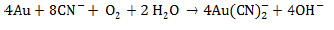
The fundamental process of gold extraction involves the formation of , a stable ion (Sadler, 1990) which brings gold into solution form and then be recovered by adsorption onto carbon. This aqueous ion is exploited by the gold industry most commonly through carbon in leach or carbon in pulp processes (Earls et al., 1995; Environment Australia, 1998; Minerals Council of Australia, 1996; Souren, 2000; Staunton and Jones, 1989). The current accepted mechanism describing gold dissolution in cyanide is shown in its simplest form by this
equation (Donato et al., 2007):
After leaching, the crushed ore is either left on the pad or removed to another area for disposal (Kjeldsen, 1999). Therefore, artisanal gold mining is a substantial source of mercury and cyanide contamination in developing countries worldwide (Appleton et al. 1999; Carling et al. 2013; Cordy et al. 2011; Taylor et al., 2005).
War use
Another use of cyanide is for war. Cyanide is a likely weapon for terrorists due to its notoriety, lethality, and availability. Battlefield use of cyanides was proposed by Napoleon III during the Franco-Prussian war, to improve the lethality of bayonets. The French introduced gaseous HCN to World War I in 1915, and used 4000 tons in battle (Morocco, 2005). HCN gas was used in the gas chambers in the World War II holocaust, in prison for the execution of criminals with death sentences, and also as a chemical warfare agent (Nsimba, 2009).
In general, cyanide has three forms: CNfree, CNWAD and CNSAD (Botz et al., 2005). Free cyanide is the CN- and HCN. Silver (Ag), Cadmium (Cd), Copper (Cu), Mercury (Hg), Nickel (Ni) and Zinc (Zn) are some examples of metals that form CNWAD and while iron (Fe) forms C NSAD (Botz et al., 2005; Nsimba 2009). In nature, these forms of cyanide are present depending on the region.
In air
In air, cyanide is present mainly as gaseous hydrogen cyanide. A small amount of cyanide in air is present as fine dust particles. This dust eventually settles over land and water. Rain and snow help remove cyanide particles from air. The gaseous hydrogen cyanide is not easily removed from the air by settling, rain, or snow. The half-life (the time needed for half of the material to be removed) of hydrogen cyanide in the atmosphere is about 1 to 3 years. Most cyanide in surface water will form hydrogen cyanide and evaporate. However, the amount of hydrogen cyanide formed is generally not enough to be harmful to humans (U.S. Agency for Toxic Substances and Disease Registry (ATSDR) 2006).
In water
Cyanide in water
In water, there are various forms of cyanide such as: CNfree (examples: CN- and HCN), CNWAD (examples: KCN, NaCN, cyanogen calcium (CaCN), cyanogen chloride (CNCl), cyanogen bromide (CNBr)) (ATSDR 1997; Xie and Hwang, 2000), CNSAD, cyanate (CNO-) and thiocyanate (SCN-).
In most natural waters, HCN is predominate (Doudoroff, 1976; Flynn and Haslem, 1995; Moran, 1998), but it is transported by rainwater in negligible quantities because of its solubility in water (ATSDR, 1997).
CNCl can be formed in drinking water during the interaction between humic substances and chloramines, occuring during chlorination of water (ATSDR, 1997; Boening and Chew, 1999). The half-life of cyanide in water is not known. Cyanide in water does not build up in the bodies of fish (ATSDR, 2006).
Physical properties
Cyanide in water takes gaseous and aqueous forms (Nsimba, 2009). Three gaseous forms of cyanide are present in water: HCN, CNCl, and CNBr (Nsimba, 2009; Xie and Hwang, 2000).
Thiocyanates are often present in the form of ferricomplexes, which can give a red coloration to water at concentrations of approximately 5 mg L-1 (Environmental Resources, 1987; Kjeldsen, 1999).
Chemical properties and reactivity
In water, free cyanide takes the form CN- and aqueous HCN (HCN(aq)). Carbon (C) is associated with nitrogen (N) by triple bond.
The CN- is a versatile ligand that reacts with many metal cations to form metal-cyanide complexes. These species, which are typically anionic, have a general formula , where M is a metal and x is the number of cyano-groups, dependant on the valence number of M (Donato et al., 2007).
Dissociation
HCN (aq) is a weak acid and can dissociate into cyanide ion according to the following dissociation reaction (Sharpe, 1976; Smith and Mudder, 1991):
The distribution of HCN and CN- species is a function of pH for a simple aqueous solution at 25°C. At pH 9.2; CN- and HCN are in equilibrium. At pH 11; over 99% of the cyanide remains in solution as CN-. At pH 7; over 99% of the cyanide exists as HCN (Nsimba, 2009). The stability of metal-cyanide complexes is variable and requires moderate to highly acidic pH conditions in order to dissociate. Weak metal-cyanide complexes dissociate under mildly acidic conditions (pH 4 to 6). Strong metal-cyanide complexes require strong acidic conditions pH < 2 to dissociate (Nsimba, 2009).Depending on the type of metal, some simple cyanides can dissolve in water, forming metal ions and cyanide ions ( Donato et al., 2007; Klenk et al., 1996):
Dissolution
The gaseous forms of cyanide are soluble in water, but degrade by hydrolysis very rapidly at high pH. The hydrolysis degradation product is the cyanate ion (CNO-), which can subsequently hydrolyze to carbon dioxide (CO2) and ammonia (NH3) at alkaline pH conditions (CDC, 2005; IPCS/INCHEM 2005; Nsimba 2009). The solubility of metal-cyanide complexes is influenced by pH and temperature (Botz et al., 1995) and the presence of ammonia (Donato et al., 2007; Franson, 1992).
Thiocyanates are frequently quite soluble in water and can produce relatively high SCN- concentrations in groundwater. The potassium- and sodium ferrocyanides (which are added to road salt to prevent clumping) are readily soluble, but can re-precipitate in the presence of iron (III) (Kjeldsen, 1999).
Photo decomposition
If groundwater contaminated with ferrocyanide is exposed to daylight, iron-complexed cyanides may be transformed to free cyanide. Therefore, samples for cyanide analysis (total or weak acid dissociable cyanide) must be stored in dark containers (Kjeldsen, 1999).
Hydrolytic reactions
Cyanate hydrolyzes rapidly under acidic conditions to carbon dioxide and ammonia (FMC, 2005 in Nsimba, 2009)
HCN is formed in solutions of cyanide by hydrolytic reaction of CN- in water (Doudoroff, 1976; Flynn and Haslem, 1995; Moran, 1998; Nsimba, 2009):
HCN is miscible with water, giving a weak acid. The CN triple bond is readily hydrolyzed by strong alkali or acid giving formic acid and ammonia, with higher temperature favoring these reactions (Donato et al., 2007).
Hydrolytic reactions are catalyzed by some enzymes, such as cyanide hydratase, nitrile hydratase, cyanidase and nitrilase (Dash et al., 2008)
Oxidation
Free cyanide can be oxidized to form cyanate (CNO-) or, depending on the pH, its protonated form fulminic acid (HOCN) (pKa=3.45 at 25°C) (Bard et al., 1985; Nsimba, 2009).
Volatilization
HCN is quite volatile with a vapor pressure of 84 000 Pa at 20°C. Henry’s constant, (KH) is given as 1.7×10-3 (mg HCN (L air)−1)-1 (Gmelius Handbuch, 1977). Therefore if the concentration is 100 mg HCN L-1 in the pore water, and all the cyanide is assumed to be dissociated, then the equilibrium concentration in the gas phase in the pores will be 0.17 mg HCN (L air)-1(Kjeldsen, 1999).
Sorption
The only significant cyanide sorption observed was for iron cyanides at low pH-values, where the total cyanide concentration in the water phase is low and thus insignificant (Kjeldsen, 1999).
Substitution/transfer
Free cyanide can react with various forms sulfur to form thiocyanate, which is relatively less toxic than free cyanide. The reactions of polysulfide and thiosulfate with the CN- are as follows (Dash et al., 2009; Luthy and Bruce, 1979; Smith and Mudder, 1991; Zagury et al., 2004):
In soil and groundwater
Cyanide in soil and groundwater
The prevalent form of cyanide occurring in soils and groundwater are the following (Alesii, 1976; ATSDR, 1997; Shefchek et al., 1995):
1. HCN, also called cyanogen, hydrocyanic acid and prussic acid.
2. Simple cyanides (inorganic salts, for example NaCN, KCN).
3. Iron-complexed cyanides: Ferrocyanide (Fe(II)(CN)63−), also called hexacyano- ferrate(II) and Ferricyanide (Fe(III)(CN)64−), also called hexacyanoferrate(III).
An additional group is the so called nitriles, organic material with a R-CN composition where R refers to the organic radical. Various pesticides containing cyanides, as well as naturally occurring nitriles, originating from plants and microorganisms fall into this group (Fuller, 1984). A related group of compounds to the cyanides is the thiocyanates containing the (SCN) group (Kjeldsen, 1999).
Physical properties
The iron-complexed cyanides are often found as a Prussian blue colouration in the soil (ferri ferrocyanide Fe(III)4(Fe(II)(CN)6)3-) and other similar iron-complexed cyanides. Cyanide concentrations of 100 to 500 mgCN kg-1 can produce bluish/greenish coloration in soils (Environmental Resources Ltd, 1987). Bog iron ore can also contain precipitated ferricyanides (Meeussen et al., 1990). Spent bog iron ore seems to contain very little free cyanide (Kjeldsen, 1999) SCN- are often present in the form of ferricomplexes, which can give a red coloration to soil. The red coloration is visible at concentrations of 50 mg kg-1 in soil (Environmental Resources Ltd. 1987; Kjeldsen, 1999)
In soils, cyanides are moderately mobile. The mobility depends on the pH condition and the percentage of iron oxides and the clay. They have a low mobility when pH is low and the percentage of iron oxides and the clay is high (ATSDR, 1997).
Chemical properties and reactivity
The behavior of cyanide compounds in soil and groundwater is governed by many interacting chemical and microbial processes (precipitation, dissolution, sorption and degradation). Therefore, cyanides in soil and groundwater have an extremely complex chemical behavior (Kjeldsen, 1999).
Dissolution and precipitation
If pH in soils is high, dissolution of iron cyanide can result. And then, iron Fe (III) and Fe(II)(CN)63-) are reform and they could be to raise to soil surface by capillary. On the surface, water is eliminated by evaporation and the ion could to precipitate, resulting in novel iron cyanide complexes (Bureau et al.,2011). Therefore, the rate of dissolution is affected by (1) buffer capacity of the soil, (2) transport of water through soils, and (3) alkalinity of water being transported through the soil (Kjeldsen, 1999; Meeussen et al., 1995).
Adsorption
Cyanide adsorption onto soils is dependent on cyanide speciation and soil mineralogy. Natural soils contain a mixture of many minerals varying greatly in physical characteristics. Aluminum oxides are ubiquitous soil components and important adsorbents in soil systems. Gibbsite (Al(OH)3(s)) is the most common form of aluminum oxide found in soils (Sposito, 1984).
Sorption
Sorption of cyanide is limited, because most soil particles are also negatively charged. This limited sorption has been confirmed by column experiments with various cyanide compounds (Alesii, 1976; Zhang and Hendrix, 1991).
Retardation of cyanide due to sorption processes is generally of minor importance in most soils (Kjeldsen, 1999).
Solubility
The solubility of iron cyanide in soil is dependent on pH. Thus, reprecipitation of cyanide can occur if conditions change. Most near surface soils contain various iron hydroxides, which dissolve according to the following reaction (assuming that iron is in the Fe(OH)3 form) (Kjeldsen, 1999):
Transformation/mineralization
Biological oxidation and ultra-violet (UV) irradiation can be transformed CNSAD or CNWAD and SCN- into NH3, sulphate (SO4), CO2, metal (Fe2+, Fe3+, Na+,…) (Kjeldsen, 1999).
Complexation
This was done to obtain iron-cyanides like ferricyanide (Fe III) and ferrocyanide (Fe II) (Kjeldsen, 1999)
In artisanal small scale gold mining catchment area
In general, three cyanide forms are observed in gold mining process waste solutions: CNfree, CNSAD and CNWAD. These forms are shown in Table 1. However, other compounds derived from cyanide are present such as cyanate (CNO-), CNCl and SCN-(Souren, 2000; Donato et al., 2007). The main WAD cyanide complexes in mining tailings waste are Cu(CN)31−, Zn(CN)42−, Ni(CN)42− and Fe(CN)6 3− (Botz et al., 1995; Ou and Zaidi, 1995; Donato et al., 2008). Iron cyanides present in the metallurgical process are Fe(II)(CN)63− and Fe(III)(CN)64− (Donato et al., 2007; Franson, 1992; Klenk et al., 1996).
Transports and fate
In artisanal and small scale gold mining, cyanide can exist in tailings ponds, surface soil, surface water, atmosphere, soil and groundwater.
Water bearing cyanide in tailings pond is in contact with tailings solids and sediment, and the atmosphere. The fate and transport of aqueous cyanide is represented by a cyanide cycle encompassing a complex set of chemical reactions involving free cyanide radical and various metal-cyanide complexes. Biodegradation of free and WAD cyanide dominates in the tailings sediment, while metal-cyanide complexation, precipitation, biological oxidation (BO), photolysis, and volatilization are the primary fate mechanisms operating in the tailings pond water. Figure 1 shows that cyanide recycling occurs between the tailings pond water and the atmosphere (A à B àC à A). For example, a portion of free cyanide in the water column (A) can volatilize as gaseous HCN (B) into the atmosphere where it can be adsorbed into moisture droplets (C) and re-enter the ailings pound via precipitation. The other portion can be biologically degraded to ammonia, carbone dioxide, and nitrate (Donato et al., 2007; Smith and Mudder, 1991).
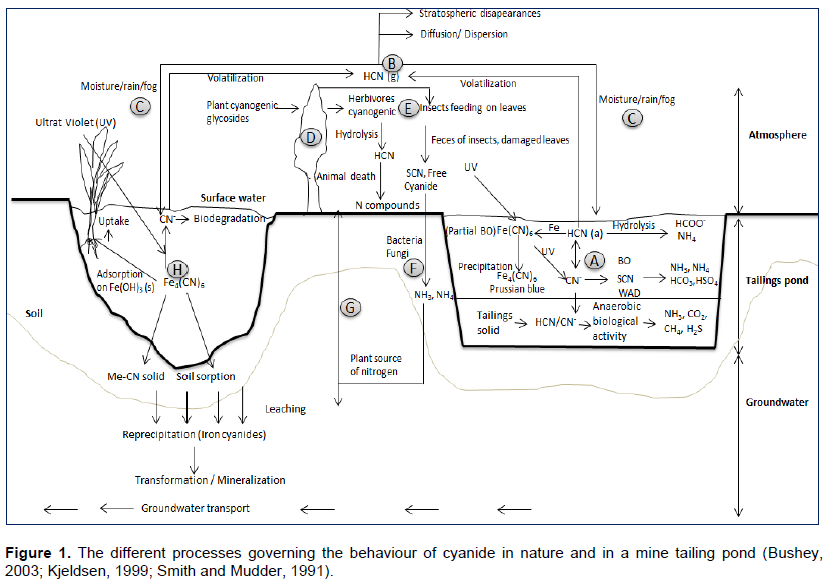
Cyanide can be transported into surface water. Both biological and physicochemical processes control the fate of cyanide in this zone. These include: Transport processes (advection and dispersion), photolysis (dissociation of iron/other metal-cyanid complexes to free cyanide via UV irradiation), volatilization of free cyanide into the atmosphere, microbiological degradation of free cyanide and weakly-complexed cyanide, plant uptake and assimilation of free and metal-complexed cyanide, rhizosphere mediated degradation of cyanide species, adsorption of free and metal-cyanide complexes onto sediments, precipitation/dissolution of iron and other metal-cyanide complexes (Bushey, 2003; Donalto et al., 2007).
Similar to the tailings pond, cyanide recycling occurring between the surface water and the atmosphere can be represented by:
H à B àC à H.
At the soil surface, cyanide can be present, if it was often land disposed on site during past mining exploitation. Leachates from this disposed material may contain iron-cyanide complexes and small amounts of other weak acid dissociable complexes depending on geochemical conditions. These soluble cyanide species can be taken up by plants or can be transported along with the groundwater as shown in Figure 1. Furthermore, cyanide in the plants can be transformed by animal metabolism (herbivores, insects). Residuals of these animals (feces, rest of animal death) and damaged leaves can be metabolized by soil microorganisms and taken up again by plants. Therefore, there is a cyanide recycling process between soil surface and groundwater following (Donato et al., 2007):
D àE à F à G àD
Physicochemical properties and reactivity
Cyanide, because of its high dielectric constant (Klenk et al., 1996), complexes readily in metallurgical circuits to dissolve other metals, preferentially gold from ores (Donato et al., 2007; Smith and Mudder, 1991; Staunton and Jones, 1989)..
Cyanide allows gold to be dissolved at a range of pH levels, although the stability of cyanide is greater at a higher pH. For gold mining metallurgical purposes, elevated pH levels of 9.5 to 10 maintain an appropriate cyanide-stability for gold dissolution (Donato et al., 2007; Souren, 2000).
Free cyanide is not persistent in the tailings environment (Botz et al., 1995; Resource Assessment Commission, 1991) and will degrade through physical, chemical and biological processes, into other less toxic chemicals (Environment Australia, 2003). Natural degradation, primarily by volatilization of cyanide in tailing storage facilities, is the most common method of removing cyanide in the gold mining waste (Botz et al., 1995; Donato et al., 2007; Environment Australia, 1998; John, 1988; Longe and Devries, 1988; Minerals Council of Australia, 1996; Ou and Zaidi, 1995; Sadler, 1990; Simovic et al., 1985; Smith and Mudder, 1995; Souren, 2000). A shallow pond with a large surface area increases the rate of conversion to gaseous HCN from the liquid phase (Environment Australia, 2003), although such spatial features attract wildlife (Donato, 1999). Aeration and mixing have a similar effect (Environment Australia, 2003). Once the pH of a tailings dam drops below 8 (Ou and Zaidi, 1995) or 9.2 (Staunton and Jones, 1989), the majority of cyanide is liberated via the gaseous state into the atmosphere (Donato et al., 2007). Unlike CNfree, WAD forms of cyanide, such as the copper complex, are resilient in the tailings dam environment (Brooks and McGlynn, 1987; Donato et al., 2007; Minerals Council of Australia, 1996; Sinclair et al., 1997; Staunton and Jones, 1989) and subsequently release cyanide ions under varying environmental conditions (Botz et al., 1995; Donato, 1999; Henny et al., 1994) and absorption by wildlife (Donato et al., 2007; Reece, 1997; Ryan and Shanks, 1996).
In addition the specific chemical profiles of metal cyanide complexes are important. Copper and zinc cyanide complexes are insoluble in water (Franson, 1992), but soluble in ammonia (Klenk et al., 1996).
Heavy-metal cyanide complexes are characterized by their stability, insolubility or being slightly soluble (Donato et al., 2007; Franson, 1992; Klenk et al., 1996).
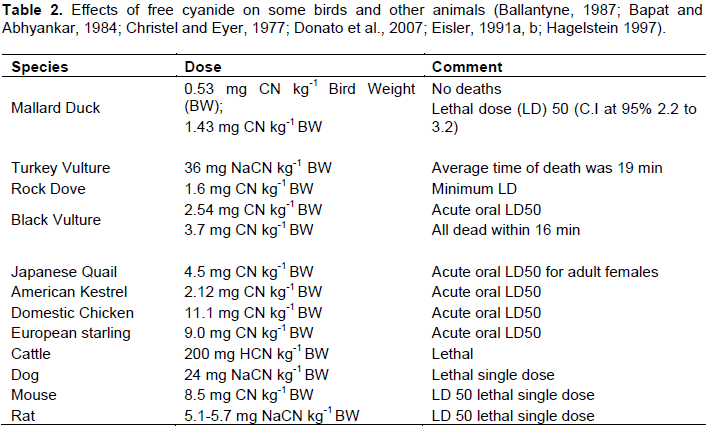
The concentrations of bioavailable cyanide complexes to wildlife in the tailing environment depends strongly on the type and valence of the complex metal ion, as well as temperature, the pH of the solution, the pH of wildlife digestive tracts, dissociation rates and the concentration of the solution (Donato et al., 2007; Hallock ,1990; Klenk et al. 1996; Smith and Mudder, 1995).
Cyanide could be removed by physical, chemical or biological treatments. Natural cyanide attenuation is also possible.
The physical and chemical treatments of cyanide operate on the principle of converting cyanide into a less toxic compound through an oxidation reaction. Several destruction processes are well proven to produce treated solutions or slurries with low levels of cyanide as well as many metals: alkaline chlorination process (Botz 2001b; Dash et al., 2009; Dubey and Holmes, 1995; Parga et al., 2003; Young and Jordan, 1995), sulfur dioxide and air process, copper-catalyzed hydrogen peroxide process, Caro‘s acid process, the iron-cyanide precipitation, activated carbon polishing, ion exchange, reverse osmosis, ozonation, etc. (Ackil, 2003).
Most of these methods are expensive and have several disadvantages (Wild et al., 1994). For example, alkaline chlorination process is not effective in the case of cyanide species complexed with metals such as nickel, silver, etc. due to slow reaction rates (Patil and Paknikar, 2000). The process also produces sludge, which requires specific license for disposal. Another disadvantage is that it is relatively expensive due to the quantity of chlorine required. Further reason, the addition of excess chlorine increases the total solids content of water, making it undesirable for recycling and reuses purposes and leaves a residue with a high chlorine content which is toxic to aquatic life (Kao et al., 2003, 2006). In addition, various chlorinated organics may be produced if the wastewater contains organic substances (Dash et al., 2009).
Natural cyanide attenuation
It is well reported that cyanide solutions placed in ponds or tailings impoundments undergo natural attenuation reactions, which result in the decrease of the cyanide concentration. These attenuation reactions are dominated by natural volatilization of hydrogen cyanide, but other reactions such as biological degradation, oxidation, hydrolysis, photolysis and precipitation also occur (Botz et al., 2005). At several sites, ponds or tailings impoundments are intentionally designed to maximize the rate of cyanide attenuation. Advantages of natural attenuation include lower capital and operating costs when compared to chemical-oxidation processes (Ackil, 2003)
Bioremediation of entity polluted by cyanide
Cyanide is a chemical compound that microorganism or plants can transform to another compound less toxic. Usually, microorganisms or plants are used for remediating environments polluted by cyanide. Bioremediation refers to the use of microorganism (Elkins, 2013) and phytoremediation refers to the use of plants. Biological methods are preferred for cyanide removal because of their low operation cost, their ability to remove a wide range of cyanide compounds, and their ability to produce high quality effluents (Botz et al., 2005).
Biodegradation mechanism
There are many groups of microorganism discovered which can transform simple or complex cyanide compounds, including bacteria such as Klebsiella oxytoca (Chen et al., 2008), Pseudomonas fluorescens P70 (Dursun et al., 1999), fungus such as Fusarium solani (Barclay et al., 1998), Fusarium oxysporum (Akinpelu et al., 2015) and algae such as Scenedesmus obliquus (Gurbuz et al., 2009).
Cyanide is used as a nutrient by the bacteria for their growth, acting as nitrogen source. Some bacteria are able to use cyanide compounds as both a carbon and nitrogen source. Therefore, supply of external carbon source is no longer needed for these bacteria. Other bacteria need glucose as carbon source for survival in presence of cyanide (Bouari, 2012; Dursun et al., 1999).
The biodegradation occurred into two steps:
The first step is the oxidative breakdown of cyanides, and subsequent sorption and precipitation of free metals into the biofilm. Cyanide and thiocyanate are then converted to ammonia, carbonate and sulfate (Ackil, 2003):
In the second step, ammonia is converted to nitrate through the conventional two step nitrification process shown as:
The ease of degradation of metal cyanides depends on their chemical stability: Free cyanide is the most readily degradable, followed by metal cyanide complexes of Zn, Ni, and Cu; iron cyanide the least degradable (Mudder et al., 1998).
Bioremediation capacity
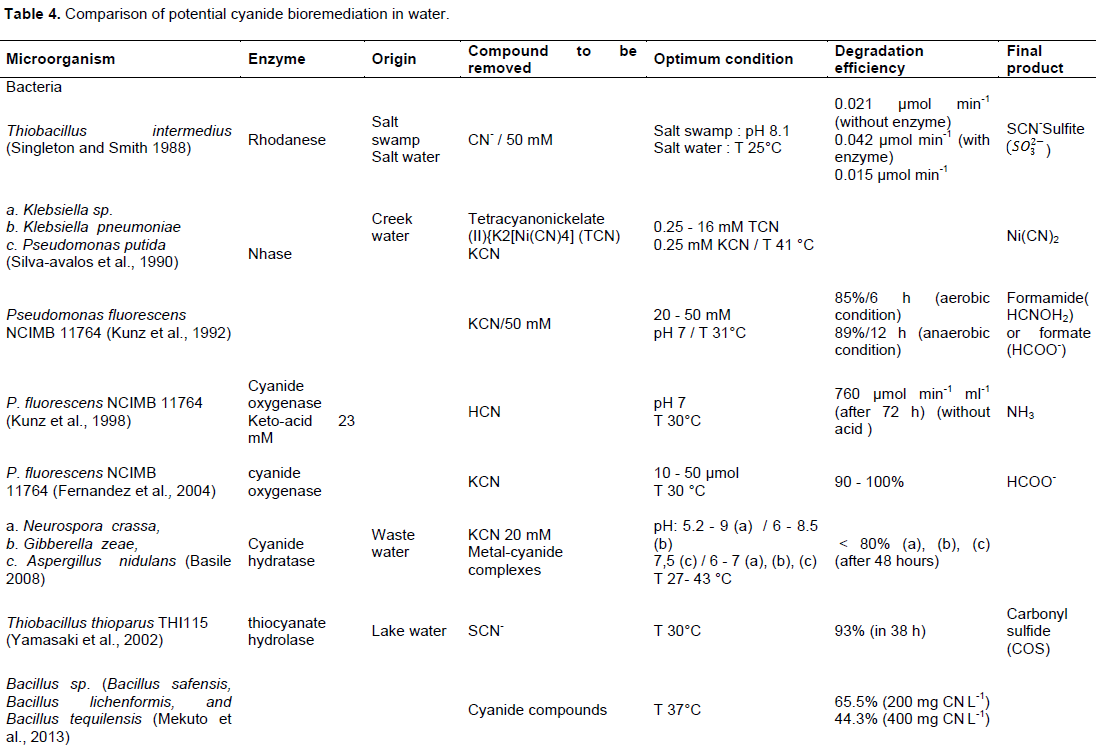
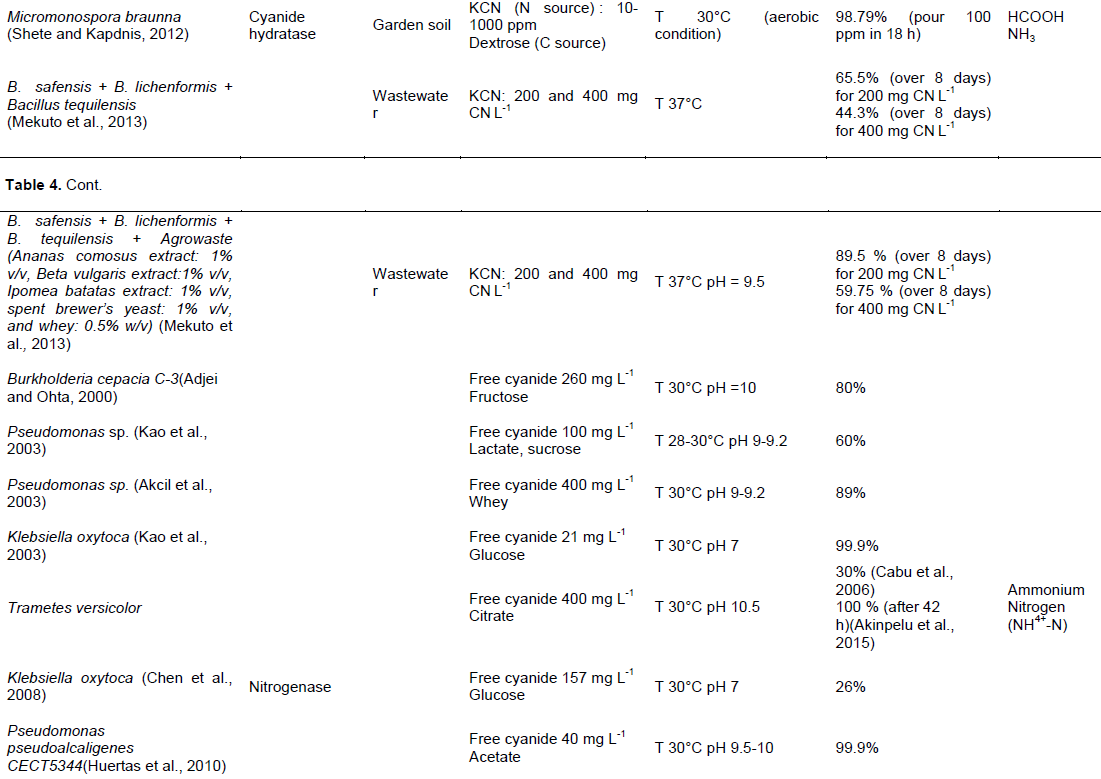
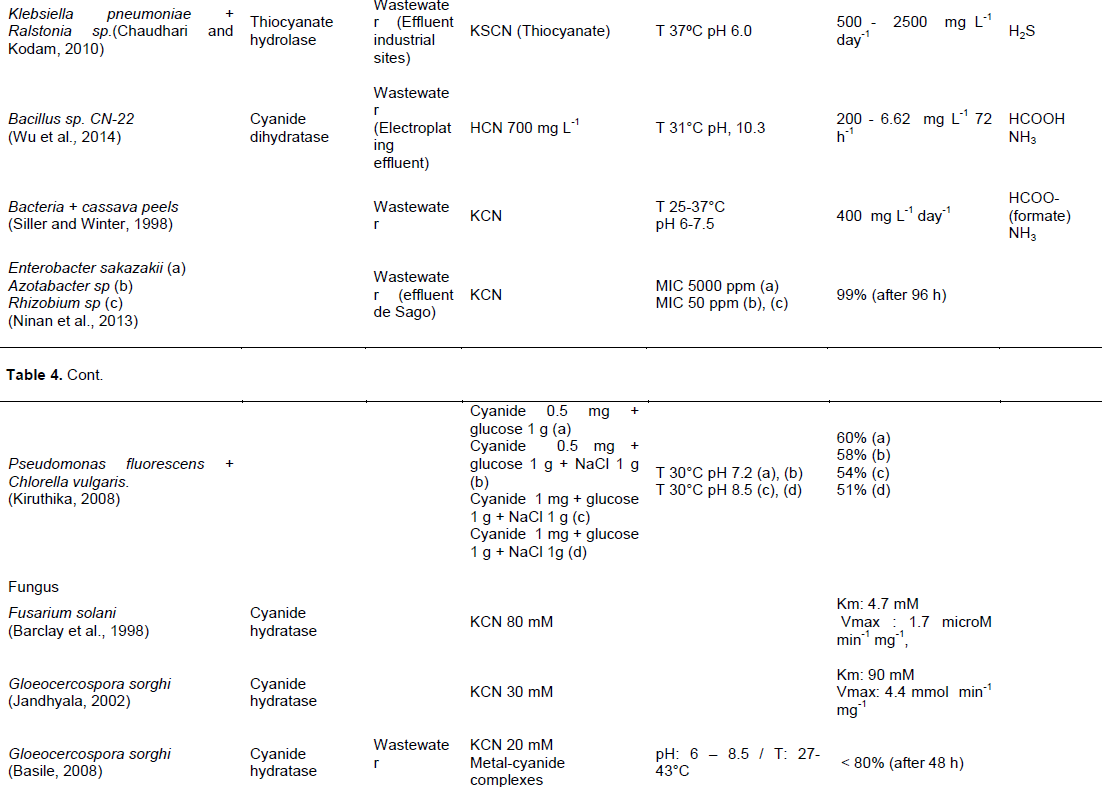
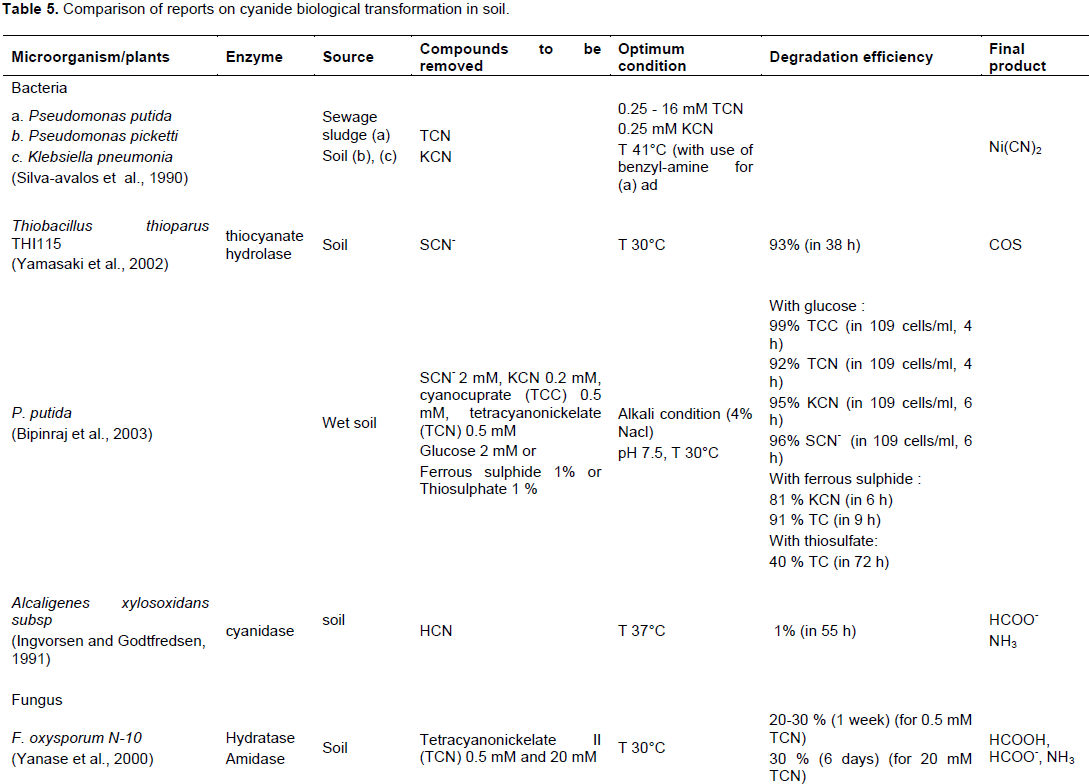
focused on: Correlation of the growth kinetics of the bacteria and the rate of cyanide removed, evaluation of the environmental parameters on the degradation of different cyanide compounds or determination of the minimum inhibitory concentration (MIC) cyanide compounds for the microorganism. Tables 4 and 5 show a comparison of studies evaluating cyanide biological transformation, respectively in water and in soil In Tables 4 and 5, the same bacteria were used for biological treatment of cyanide but the potential effectiveness varies depending on the composition of the medium, type and initial concentration of cyanide and organic matter, pH, and temperature.

In the water, the optimal condition is formed by a pH ranging from 5.2 to 10.5 for bacteria, from 6 to 8.5 for fungus and pH 12 for plant. Temperature is usually held between 25 and 50°C, with the majority around 30°C for bacteria, 43°C for fungus and 40°C for plants. Cyanide hydratase is often used by microorganism as enzyme for degrading cyanide. Pseudomonas Fluorescens NCIMB 11764 was identified as the potential cyanide degrading bacteria, its performance is between 105 and 706 µmol min-1 for degrading free cyanide with or without enzyme as catalyzer (Kunz et al. 1992; Kunz et al., 1998). The mixed bacteria composed by Klebsiella pneumoniae and Ralstonia sp. have also a high potential with a velocity 1042 µg L-1 min-1 for degrading thiocyanate (Chaudhari and Kodam 2010). For Fungus, Gloeocercospora sorghi is the most effective. Its maximal velocity is 4.4 mmol min-1 mg-1 (Basile, 2008; Jandhyala, 2002).
In the soil, the optimal condition was formed by a pH around 7 for bacteria and pH 4 for fungus. The cyanide degrading bacteria have a temperature between 30 and 37°C. But, for fungus, it stays around 30°C. For degrading cyanide, bacteria and fungus use various enzymes as: Thiocyanate hydrolase, cyanidase and hydratase amidase. Microorganisms have a faculty to degrade strong acid dissociable cyanide in the soil than in the water. Pseudomonas putida. was identified as the potential cyanide degrading bacteria (Bipinraj et al., 2003). The fungus, F. oxysporum N-10 is the most effective with a velocity 0.02 and 1 mM day-1 respectively in the mixed (Barclay et al., 1998) and single culture (Yanase et al., 2000).
Glucose was often used as organic matter in water and soil and the final product of biodegradation is formed by ammonia or ammonium. While earlier studies only focused on the microorganism application, after the discovery of co-culture bacteria or fungus, many current studies are focused on the addition of agricultural wastes or wastes in the microorganism mixed culture.
Research about phytoremediation is not investigating deeply.
Bioremediation technologies
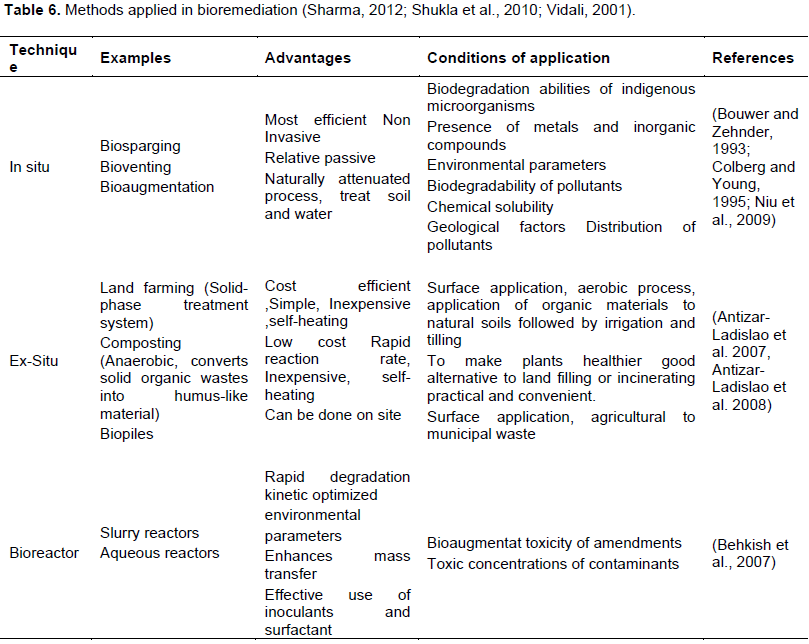
More choice of bioremediation technologies were existedfor removing cyanide. It could be conducted in-situ or ex-situ or by using bioreactors (Sharma, 2012). Each method has its specificity and most of them are cost-effective as shown as in Table 6, which summarize the advantages of the different technologies, and their conditions of application.
Bioremediation application in the world
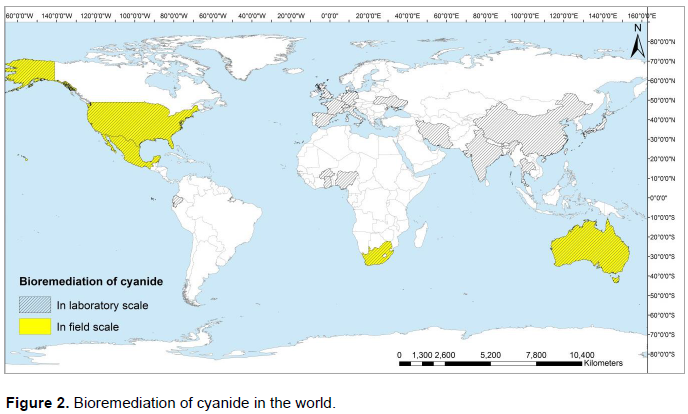
Most of researches were focused on the bioremediation of water, soil and solid wastes in lab-scale. Application of bioremediation in situ or ex-situ is few limited. Figure 2 show the bioremediation applied on the entity polluted by cyanide in the World. Most of them were observed in America such as United States (Akcil and Mudder, 2003; Fernandez et al., 2004; Kunz et al., 1992; Kunz et al., 1998; Pinedo-Rivilla et al., 2009; Silva-avalos et al., 1990; Singleton et al., 1988; Wang et al., 1996), Mexico (Meehan et al., 1999), Ecuador (Diaz and Caizaguano, 1999); Europa such as France (Dumestre et al., 1997; Ferret, 2012), Germany (Ingvorsen en Godtfredsen, 1991), Switzerland (Brandl et al., 2003), Italy (Cipollone et al., 2007), Spain (Luque-Almagro et al., 2005; Luque-Almagro et al., 2008; Quesada et al., 2007), United Kingdom (Baxter and Cummings, 2006; Ezzi and Lynch, 2005), Ukraine (Podolska et al., 2003); Asia such as China (Zhou et al., 2007), India (Bipinraj et al., 2003; Shete and Kapdnis, 2012), Japan (Yamasaki et al., 2002), Iran (Mirizadeh et al., 2014; Naghavi et al., 2012), Malaysia (Maniyam et al., 2011), Taiwan (Kao et al., 2003), Thailand (Potivichayanon and Kitleartpornpairoat, 2010) and Oceania such as Australia (Markwiese and White 1991). Few Africans countries as Ghana (Adii, 2011), Nigeria (Oyedeji et al., 2013), South Africa (Akinpelu et al., 2015; Dent et al., 2009; Jandhyala et al., 2003; Mekuto et al., 2013; Ntwampe and Santos, 2013; Santos et al., 2014; Van Zyl et al., 2011) and Burkina Faso (Razanamahandry et al., 2016) have been applied bioremediation in laboratory scale. Only South Africa country had applied the cyanide bioremediation in field scale (Shumba, 2008). But, cyanide is widely used in the artisanal gold process extraction in African countries (Adii, 2011).