Maize (Zea mays L.) is one of the most widely grown crops in the world, ranking third next to wheat and rice (Reeves et al., 2016). In sub-Saharan Africa (SSA), maize is the most widely-grown staple food crop occupying more than 33 million hectares each year (FAOSTAT, 2015). It is among the most important and widely grown crops in Ethiopia, ranking first in total production with over 7.23 million tones of produce and second in area coverage next to Teff (Eragrostis tef (Zucc.) Trotter) (Mosisa et al., 2012; Central Statistical Authority (CSA), 2014). Considering its importance, wide adaptation, total production and productivity, maize is regarded as one of the high priority food security crops in Ethiopia, the second-most populous country in SSA after Nigeria (CSA, 2011). However, maize production has remained low, with the estimated national average yield of 3.4 t ha-1 (CSA, 2014) compared to the world average yield estimated at 5 t ha-1 (FAOSTAT, 2015), due to several major diseases, including foliar diseases.
Turcicum leaf blight (TLB) is a major foliar disease of maize in most production areas worldwide (Jakhar et al., 2017). It is a fungal disease caused by Exserohilum turcicum (Pass.) K. J. Leonard and E. G. Suggs. The pathogen was formerly known as Heliminthosporium turcicum Pass (Khedekar et al., 2010). The disease is more prevalent in humid areas with moderate temperatures (Pataky et al., 2006). It is widely distributed, however, sporadic in nature and its development mostly depends on weather conditions, stage of plant growth and level of resistance in maize cultivars (Perkins and Pedersen, 1987). The pathogen has wide host range and a high pathogenic variability (Muiru et al., 2010).
The pathogen attacks all parts of the plant but the most conspicuous symptoms/lesions are found on the foliage. Lesions destroy the leaves, resulting in yield losses due to lack of carbohydrate to fill the grains. Heavily infected fields present a scorched or burnt appearance resulting in premature death of leaves (Harlapur et al., 2007). TLB causes extensive leaf damage and defoliation during the grain filling period, and yield losses due to necrosis or chlorosis of leaves premature death of the leaves and loss of nutritive value even as fodder (Patil et al., 2000) has been reported. Yield losses of up to 28 to 91% due to TLB have been reported in Italy, mostly when heavy infection occurred before tasselling (FAO, 2010).
Ramathani et al. (2011) reported the prevalence of TLB in highlands and wetter areas of the Kenya and Uganda. Previous reports have also shown that E. turcicum is a serious pathogen in highlands associated with cool, high relative humidity, mid-altitudes and cloudy weather conditions (Palaversic et al., 2012). Extreme impact of TLB on maize in the highland agro-ecologies have also been reported in Uganda, Kenya, Ethiopia and Zambia and South Africa (Ramathani et al., 2011).
In Western Ethiopia, TLB (E. turcicum) is the most important maize disease reported. Farmers in this area (46.7%) indicated TLB as the major leaf disease on maize while gray leaf spot Cercospora zeae-maydis, GLS, is ranked as the second most important leaf disease in the area (Wende et al., 2013). Therefore, TLB ranked as the number one problem and is considered as a high research priority of maize in Ethiopia (Wende et al., 2013).
TLB incidence ranges from 95 to 100% in areas with constant moisture and high humidity and the yield loss can reach up to 70% (Tewabech et al., 2012). It is reported to cause devastating damage on most commercial varieties of maize released in the country (Tewabech et al., 2012).
TLB varies in incidence and severity from year to year and from one locality to another depending largely on genetic makeup of the plants and prevailing environmental conditions. It is a multiple cycle disease and new repeated inoculations are needed for disease development making it highly dependent upon sporulation from other lesions (Ullstrup, 1966).
Previously, the TLB disease was limited to specific areas and varieties; but currently the disease has become very important almost in all maize growing agro-ecologies. Most released varieties under production are affected by the disease. Although, it becomes very important in the nation, there is limited quantified data that reflect the extent of its distribution across highlands and mid-altitude maize growing agro ecologies and the reaction of maize varieties to this disease under this production system need to be studied. Thus, the present investigation was undertaken with the following objectives:
1) To assess the incidence and severity of TLB in major highland and mid-altitudes of maize growing agro-ecologies of Western part of Oromia, Ethiopia.
2) To map the geographical distribution of the TLB disease in the study area.
3) To assess the reaction of maize varieties under production to TLB in surveyed area.
Study areas and sampling system
Field surveys were carried out during the 2017 growing season in major maize growing agro-ecologies of Western part of Oromia region of Ethiopia (Figure 1). West Shewa, South West Shewa, Horro Guduru Wollega, East and West Wollega zones were assessed to determine the incidence and severity of TLB of maize. Zones and districts were selected based on the differences in production (farming) systems, weather condition (Relative humidity, maximum and minimum temperature), altitudes and major vegetation cover (availability of maize crop) (Ramathani et al., 2011).
However, some farming systems tend to overlap between districts.
From each representative zone, 2 - 12 districts were surveyed based on production of maize crop and 8 - 73 fields each averaging 1 ha at the interval of 5 - 10 km along road side were assessed per zone. Such fields were randomly selected on both sides of the road. A total of 172 farmers’ fields, 29 districts and 5 zones were surveyed when the crop was at flowering to grain filling stage. Each sampling point was marked with the global positioning system (GPS) using GPS receiver for altitude and co-ordinates. In each field, 9 m² areas were marked out in three randomly selected points in a diagonal form using meter tap. Ten stands of maize plant in the middle of each marked area were randomly selected and assessed for incidence and severity (Nwanosike et al., 2015). Each marked area was regarded as a replicate for disease assessment.
Assessment of Turcicum leaf blight
Diseases incidence
Disease incidence was assessed as proportion of plants showing symptoms in the field. The number of plants within 10 randomly selected stand showing TLB symptoms were counted and expressed as a percentage of the total number of stands per plot using the following formula (Nwanosike et al., 2015).
Disease severity
Disease severity on whole plant basis was rated using a visual scale of 0-5; where, 0 = all leaves free from infection, 1= a few restricted lesions on the lower leaves (≤ 5%), 2= several small and large lesions on many leaves (5.1 - 10 %), 3= numerous small and large lesions on many leaves (10.1 - 25%), 4 = many enlarged and coalesced lesions on many leaves above the cob (25.1 - 50%), 5 = several coalesced lesions, leaf showing wilting, tearing and blotching typical blight symptoms (>50%) (Muiru et al., 2007; Durrishahwar et al., 2008). Severity scores were converted to percent disease index (PDI) (Wheeler, 1969; Kumar et al., 2011).
Where: PSI is percent severity index. Varieties with ratings; 0 - 1.4 (<30%) were considered resistant, 1.5 - 2.4 (30 - 50%) = moderately resistant, 2.5 - 5 (>50%) = susceptible (Muriithi, 2001).
Data analysis
Disease data (incidence, severity and PSI) were entered into Excel to calculate means and percentages. Correlation analysis was carried out between disease data and weather parameters using the Proc. Corr. procedures of the SAS software (SAS, 2002). Disease distribution maps were generated using the Arc GIS 10.3 software with spatial analyst by interpolating the surface from GPS points and the associated field severity data using the inverse distance weighted (IDW) interpolation method.
Status of Turcicum leaf blight of maize (TLB) in surveyed areas
Turcicum leaf blight of maize was prevalent in all surveyed agro-ecologies in 2017 cropping season at varying levels (Table 1). The mean TLB incidence in the surveyed districts ranged between 16.3% (Abay chomen district of Horro Guduru Wollega Zone) and 96.7% (Wayu Tuka district of East Wollega Zone). Amongst all the surveyed districts, the highest disease incidence (96.7%) was recorded in Wayu Tuka followed by Lalo Assabi, Sibu Sire, Diga and Bako tibe with disease incidence of 94, 92.5, 91.8 and 90%, respectively. On the other hand, Abay chomen, Horro, Bacho and Jima rare districts had relatively lower TLB incidence of 16.25, 20, 25 and 26.7%, respectively.

Percent severity index (PSI) ranged from 3.13% in Abay chomen district to 57.50% in Lalo Assabi district. The highest mean PSI (57.5%) was recorded in Lalo Assabi district followed by Sibu sire, Bako Tibe, and Toke Kutaye, which had 50.7, 46 and 45% PSI, respectively. In contrast, the mean minimum PSI was recorded in the Abay Chomen district (3.13%) followed by Bacho (4%), Dandi (6.33%) and Jima rare (6.67%).
The highest maize TLB incidence and PSI were recorded in areas with a mean maximum temperature of 31.12 - 34.93°C and humid areas (Table 1). The finding of the present study is in agreement with Nwanosike et al. (2015) who stated that Turcicum leaf blight of maize is widely distributed, and the level of intensity varied across locations in Tanzania and the highest TLB incidence and percent severity index recorded at maximum temperature up to (31 - 35°C) and high relative humidity (75 - 100%). Earlier researchers (Harlapur, 2005; Khedekar et al., 2010; Rani, 2015)suggested that prevailing environmental conditions (relative humidity, maximum and minimum temperature) during cropping season could be reasons for higher disease incidence. Ullstrup (1966) also reported that TLB incidence varies in prevalence and severity from year to year and from one locality to another, depending largely on environmental conditions (that is, humid weather along with heavy dew favored the spread and development of the disease in an epidemic form). TLB has particularly been noticed to cause significant maize yield reduction in many production regions including in sub-Saharan Africa (SSA), especially in the humid mid-attitude and highland regions (Latterell and Rossi, 1983; De Vries and Toenniessen, 2001).
Beshir et al. (2015) reported that E. turcuicum was the causative agent of leaf blight observed in central Sudan. Furthermore, the TLB occurred in all the study locations with incidence and severity ranging from 45 to 100, and 65 to 100, respectively.
In Africa, where maize and sorghum are the staple foods, TLB is reported to be widespread in the warm and humid growing regions of Ethiopia, Tanzania and Uganda (Adipala et al., 1993a; Tewabech et al., 2001).
Geographical distribution of TLB of maize in surveyed areas during the season
Disease incidence, PSI and GPS records were used to construct disease map in surveyed zones and districts.The disease map illustrates severity and incidence levels over the agro-ecologies and was used to study epidemic patterns at the time of the study. Results showed spatial pattern of epidemics.
TLB incidence was categorized on the map as described by Harlapur (2005), where 0 - 5% = slight/trace infection, 5.1 - 15% = light infection, 15.1 - 30% = moderate infection, 30.1 - 75 = severe infection, >75 = very severe infection.
In terms of incidence, virtually all districts had very high level of disease. The TLB incidence in vast majority of districts studied recorded disease incidence of 75% and above (Figure 2). In Ambo Zuria, Ilu Galan, Dire Inchini, Nono, Dandi, Dawo, Waliso, Guduru, Kiramu, Gobu Sayo, Guto Gida, and Gimbi districts severe infection (30.1 - 75%) of TLB were recorded which was indicated by orange colour on the map. Toke Kutaye, Sasiga, Diga, Wayu Tuka, Sibu Sire, Bako Tibe, Jibat, Lalo Assabi, Dano, and Chelia recorded above 75% which were very severe infection indicated by red colour on the map (Figure 2). On the other hand, moderate infections (15.1 - 30%) of TLB were recorded in a few districts such as Becho, Abay Chomen, Horro and Jimma Rare. This was due to moderately resistant to resistant hybrid varieties or cultivar grown in the districts.
The disease map also illustrates spatial patterns of epidemics with some districts showing high variation in PSI. PSI levels ranged from as low as 3.13% in Abay Chomen to as high as 57.7% in Lalo Assabi district. It was described on the map according to Nwanosike et al. (2015), where, 0 – 29 = low severity, 30 – 49 = moderate severity and 50 – 100 = high severity (Figure 3).
In general, low value of disease incidence and severity suggests that a number of varieties grown by farmers appear to be resistant or tolerance to TLB. Second possible explanation for the observed data could be crop rotation (especially preceding crop).
The distribution of TLB and pathotypes of E. turcicum have been identified in Kenya (Ngugi et al., 2000) and Uganda (Sserumaga et al., 2013). According to Wende et al. (2013), Turcicum leaf blight is ranked as the number one problem and is considered a high research priority of maize in Ethiopia.
Status of TLB in relation to maize varieties under production
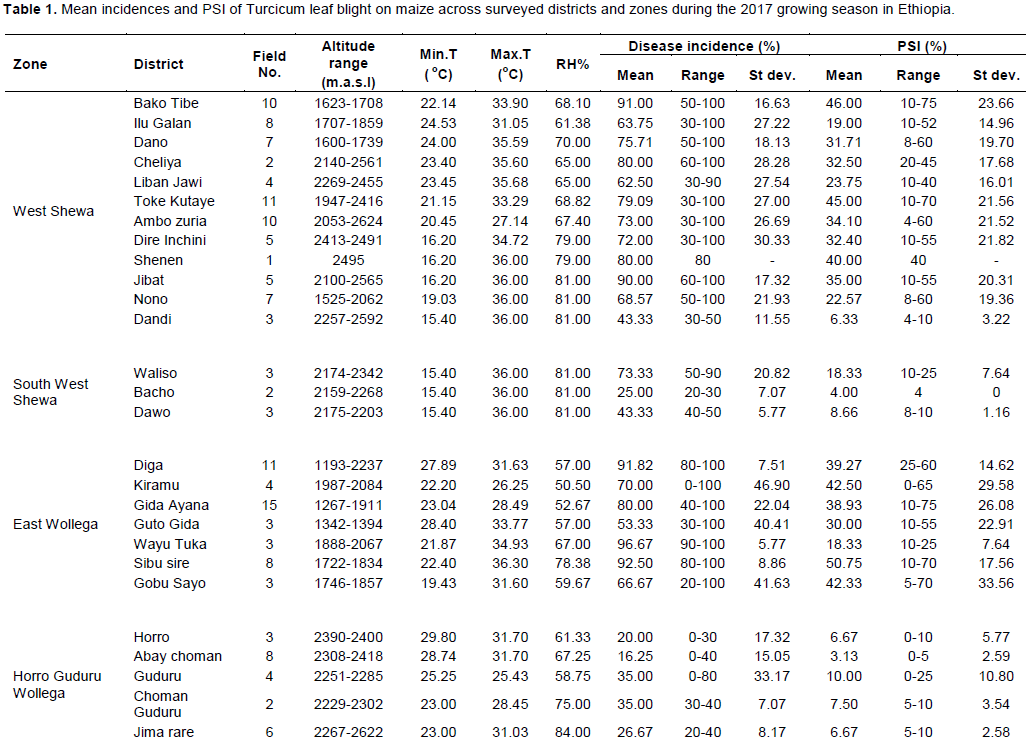
Survey result on incidence and percent severity index of TLB revealed that maize varieties under production varied in their reaction to TLB. The highest mean disease incidence (90%) was scored on variety designated as Oromia, whereas the lowest (30%) was recorded on variety denominated as Shashamane (Figure 4). Varieties such as Limu, Wenchi and BH-540 had 84, 80 and 79.1% disease incidences, respectively. TLB incidences of 37.5 and 47.5% were recorded on unspecified hybrid and Jibat varieties, respectively.
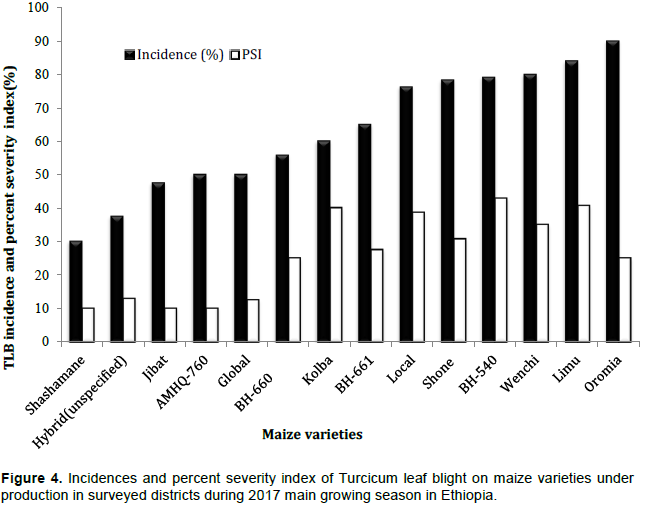
TLB index expressed as PSI ranged from 10% on variety Shashamane to 42% on BH-540. Maize varieties Limu and Kolba also had higher PSI of around 40%. The study revealed that TLB was more common and severe on varieties such as BH-540, Limu, Kolba and Local. In contrast, varieties such as Shashamane, Global and Jibat performed relatively better than others with respect to the TLB reaction in the surveyed areas (Figure 4). This indicated that the upsurge in the TLB on maize was highly influenced by the nature of susceptibility/resistance of the varieties and conducive environmental conditions for the development of the disease in the location. Earlier survey reports (Harlapur et al., 2000; Ramathani, 2011) indicated that cultivar susceptibility and weather parameters play an important role in influencing TLB severity. Turcicum leaf blight is reported to cause devastating damage on most commercial varieties of maize released in Ethiopia (Tewabech et al., 2012).
Krausz et al. (1993) reported an epidemic of turcicum leaf blight from Texas and reported loss in grain yield of susceptible hybrids ranging from 40 to 50%, whereas hybrids with either Ht1 resistance gene or significant polygenic resistance had minimum disease incidence. Babu et al. (2004) reported TLB incidence on maize at Almora and it attained epidemic proportion resulting in 83% yield reduction.
Intensity of TLB at different growth stages of maize
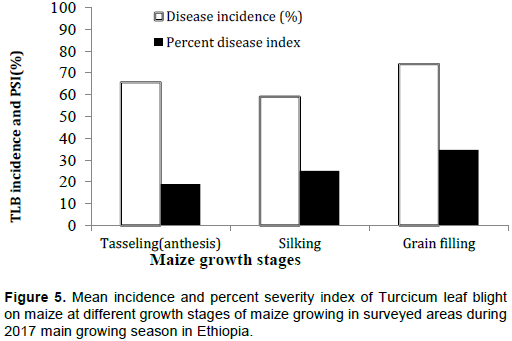
TLB incidence and PSI showed marked variations across the different maize growth stages. This revealed that the development of TLB also relies on the maize growth stage. The overall mean incidence and PSI were high (up to 74.1 and 34.7%, respectively) during the grain filling stage of maize compared to tasseling and silking stage of the crop (Figure 5). Kalappanavar (2017) conducted survey in India and reported that the maximum disease severity was recorded at grain filling stage (22.06 and 15.50%) and minimum at vegetative stages (14.31 and 9.49%).
From the current study, data higher yield loss due to TLB could be expected in many of the surveyed location because the disease occurred before silking stage of maize. This is in agreement with the study of Raymond and Hooker (1981) who stated that if the TLB disease establishes before silking, yield reduction of up to 40% may occur but if infection delays until 6 - 8 weeks after silking, yield losses would be minimal. The present study confirmed wider distribution of TLB in surveyed areas as associated with plant stages. This is in line with Perkins et al. (1987) who stated that TLB disease is widely distributed; however, sporadic in nature and mostly depends on weather conditions, stage of plant growth and level of resistance in maize cultivars.
In Uganda, overlapping of growing seasons and presence of off-season maize resulted in infection before tasseling stage, consequently resulting to higher yield loss (Adipala et al., 1993b). Yield loss is caused predominantly through loss of photosynthetic leaf area due to blighting (De Vries and Toenniessen, 2001). Loss of photosynthetic tissue can result in decreased yield, and silage quality can be affected (Jakhar et al., 2017).
Effects of cropping system and preceding crop on maize TLB incidence and PSI
Types of cropping system, previous crop, and residue left in the field played important role for the development of maize TLB in the surveyed areas. PSI ranged from 12.3 to 60% during the current survey (Table 2). The maximum PSI was recorded in fields where maize is intercropped with sorghum while maize-kale intercropping was associated with the lowest PSI. On the other hand, most of the intercrops failed to reduce TLB level as compared to mono cropping. The maximum TLB severity in sorghum-maize intercropping might be due to sorghum’s ability to serve as collateral host for E. turcicum, the cause of TLB. However, no definite pattern was established in the current experiment as far as the effect of intercropping on TLB is concerned. Assefa (1997) also reported that planting maize and sorghum together showed higher TLB intensity and lower common rust (CR). Ramathani et al., (2011) also suggested that cropping systems significantly influenced the patterns of spread of TLB disease.
With respect to effect of preceding crop, the current work failed to establish a pattern with respect to effect of rotation on TLB development. The only exceptions were maize following fenugreek, which had 30% incidence and 5% PSI, a much lower score as compared to other systems. TLB is residue born disease; as a result, alternatively sowing non cereal crops in the same field can reduce the availability of host residue from the field, thereby affecting the chance of the pathogen to over season. This obviously reduces the inoculum level (source) and the incidence of early season development of the disease. Studies on TLB on maize indicate that maize residues are an important factor for the survival of the pathogen and initiation of the epidemic (Fullerton and Fletcher, 1974). Assefa (1997) stated that maize planting after maize would result in high TLB intensity, while planting maize following noug-maize-noug (Niger Seed) system result in lower severities of TLB. High percent severity index on maize was due to intensive cultivation of maize crop season after season, every year, narrow genetic makeup of the commercial hybrids and non-adoption of disease management practices by the farmers (Kalappanavar, 2017). Increased severity of Turcicum leaf blight in Africa associated with continuous cultivation of maize and susceptible maize cultivar has also been reported by Danson et al. (2008). However, lack of marked reductions in TLB level across most of the rotation schemes, included in the current survey, might be because of short durations for rotating maize with other crops. The fact that most of the rotating crops were cereals and might have also contributed to the lack of any significant effect by the rotation scheme on TLB development.
TLB incidence and percent disease index on maize in relation to fertilizer application
Fertilized and non-fertilized maize fields assessed during the survey varied in the level of TLB incidence and PSI.
The TLB disease incidence and PSI were relatively low in fertilized fields compared to non-fertilized fields (Figure 6). Mean minimum TLB incidence (67.7%) and PSI (29.9%) were recorded from fertilized fields. On the other hand, maximum disease intensity, 86.7% TLB incidence and 44.8% PSI were recorded in non-fertilized maize fields (Figure 6). It is known that plants suffering from nutrient deficiencies are weaker, slow in growing, and faster in aging. Such plants are susceptible to pathogens (Agrios, 2005). Assefa (1998) studied the effects of farm-yard manure (FYM), and nitrogen and phosphorus rates (N/P) on the intensity and frequency of Turcicum leaf blight and reported that the incidence was low (26.4%) at 20/46 kg/ha N/P2O5 plus 24 t/ha FYM as compared to the other combinations (with a maximum incidence of 33.6%) and the two checks (40/46 and 75/75 kg/ha N/P2O5). Plant population and fertilize application influence the incidence and severity of Turcicum leaf blight of maize. TLB of maize percent disease index was significantly reduced in treatments that received optimum fertilizer dosage compared to others (Kumar et al., 2017).
Maize TLB incidence and percent severity index in relation to planting date
Disease incidence and percent severity index were low in maize fields planted late April. The minimum disease incidence (50%) and percent severity index (7%) were recorded in maize fields planted in this time followed by mid-May where TLB incidence and percent severity index were 51.5 and 18.25%, respectively. The maximum disease incidence and percent severity index of TLB were observed on maize fields sown at early April and mid-June. At early April and mid-June the percent severity index recorded was 55%. But, disease incidence showed slight difference 100 and 80%, respectively (Figure 7). This very high and low severity of TLB disease is due to the environmental conditions that favour the development of disease and increase the incidence and severity on the maize during this growing season. This study indicated that early and late planting date favoured TLB development (both TLB incidence and severity high in maize). Knowing proper planting date can help the farmers to escape the maize crop from the time when the epidemics of this disease outbreaks. Timely planting can often help cultivars escape the most severe damage from TLB if crop development outpaces normal disease progression. The latest-planted maize in an area may be infected when plants are smaller, resulting in the disease progressing more rapidly relative to the crop. However, in cases of high disease incidence, both early- and late-planted maize may be severely damaged by TLB (Jakhar et al., 2017). Assefa (1997) reported that planting maize before May 18 could result in a lower incidence of TLB.
Correlation between Turcicum leaf blight of maize and weather parameters
TLB incidence and PSI positively correlated with relative humidity although not significantly differ (Table 3). On the other hand, TLB incidence negatively correlated with maximum and minimum temperatures. Similarly TLB PSI was negatively correlated with maximum and minimum temperatures, while altitude had significant and negative relationship with both TLB incidence and PSI. In general, the disease levels were significantly influenced by altitude but not weather conditions according to results of the current survey. Reddy et al. (2013) observed negative correlation between disease incidence and maximum temperature and positive correlation with high relative humidity. They reported that the mean minimum temperature had no effect on the disease development.
The results are contrary to those reported by Nwanosike (2015) in which TLB was highly influenced by relative humidity. Rani (2015) also reported positive and significant relationship between relative humidity and disease intensity and significantly negative correlations between minimum and maximum temperature on one hand, and disease intensity on the other. Rai et al. (2002) have also revealed definite relations between environmental parameters and disease development with the maximum initial disease incidence (100%) and significant disease severity (4.00) recorded during February.
The deviations of current survey results from those of previous studies might be because weather data were recorded only using portable devices during disease assessment dates in the current survey. It should be noted that disease development in general is influenced by long term and short term weather conditions that span longer than the few particular dates. However, absence of weather stations in the survey areas, make it impossible to have weather records throughout the growing period and beyond. Other reason for this deviation of the area covered in this investigation were mid and highland altitude parts of major maize growing areas in western parts of Oromia regional state only (that is, the lowlands major maize growing areas were not covered in this study).