ABSTRACT
Countries with the highest HIV prevalence, Botswana, Eswatini, Lesotho and South Africa are also experiencing an increase in climate variability, rising temperatures and drought. This study investigates perceived challenges of high temperatures and water scarcity/rationing on monthly clinic attendance and ARV adherence for people living with HIV in Greater Gaborone, Botswana. A systematic random sample of adult women [n=419], including 67 pregnant women and mothers, and men [n=239] attending 21 HIV clinics were surveyed. Health care workers [n=84] were interviewed. Close-ended and open-ended questions were analyzed using logistic and multilevel models and content analysis. Women and men reporting exposure to high temperatures were at increased odds of ARV non-adherence, especially in rural areas; missing clinic appointments due to heat in rural areas and water rationing in peri-urban and rural areas increased the odds of ARV non-adherence; almost half of respondents reported water rationing negatively affected their health; and perceptions of available clinic services during water shortages, differed for some men and health care providers. Infrastructural and health education messaging improvements are needed in HIV clinics, particularly in rural villages to adapt to climate variability and ensure ARV adherence for people living with HIV in these areas.
Key words: HIV, antiretroviral therapy adherence, climate variability, water shortages, water rationing, ARV clinics, treat all program, Botswana.
The region of Southern Africa is experiencing profound climate variability and change, with climate models predicting increases in temperature, rainfall reductions, and decreases in available water resources in the upcoming decades (International Panel of Climate Change (IPCC), 2007). Southern Africa is also home to four countries with the highest adult HIV prevalence in the world: Botswana (rate 20.3 per 100 population, range 17.3-21.8), Eswatini (27.3, range 25.1-29.1), Lesotho (23.6, range 21.2-24.7) and South Africa (20.4, range 17.4-22.5) (United Nations AIDS (UNAIDS), 2020). In 2006, the President’s Emergency Plan for AIDS Relief (PEPFAR) implemented a Safe ‘Water, Sanitation, and Hygiene’ (WASH) campaign in PEPFAR country-programs (United States Agency for International Development) (USAID, 2011). WASH was intended to promote biological health, while also preventing HIV-comorbidities and opportunistic diseases by focusing on three hygienic improvement practices: safe drinking water, washing hands with soap, and safe handling and disposal of feces (Marriott et al., 2018). For people living with HIV, safe drinking water promotes hydration as well as gastrointestinal absorption of antiretroviral (ARV) medications, including Prevention of Mother-to-Child Treatment (PMTCT) for postpartum women and infants. Diarrheal infections caused by unclean water can interfere with ARV and PMTCT absorption, contribute to malnutrition and reduced immunity, as well as contribute to new HIV strains resistant to treatments (USAID, 2011). Clean water is also necessary for bathing and keeping the house clean to reduce the potential of secondary (opportunistic) infections, and clean water and sanitation provisions promote the dignity of people living with HIV and their caretakers (Haftu et al., 2017).
In 2017, the “Treat All Program” was implemented in PEPFAR country-programs in Southern Africa (PEPFAR, 2019) with the aim to eliminate HIV by 2030 with 90-90-90 treatment targets: 90% of all people living with HIV will know their HIV status (via expanded HIV testing); 90% of all people who know their HIV positive status will receive sustained ARVs (via monthly clinic visits); and 90% of people receiving ARVs will achieve viral suppression (via ARV adherence), thereby, eliminating the potential of HIV transmission. ARV and PMTCT adherence is thereby dependent upon (a) attending monthly clinic appointments for testing to monitoring HIV progression via the CD4 blood count (biological measure of immunological health, optimal > 350 cells/mm3) and viral load (potential for HIV transmission, optimal < 200 copies/mL) and prescription renewal and (b) between clinic visits, taking the ARV drugs as prescribed, without missing dosages.
While the WASH program has been successful in improving hygienic practices in resource-poor settings (Mills and Cumming, 2016), there is a need to further understand how high ambient temperatures and water scarcity and rationing, adversely impact ARV and PMTCT adherence. ARV adherence may be compromised if people living with HIV miss their monthly clinic appointment due to extreme heat while walking or traveling long distances and/or the inability to bath and prepare for their appointment. ARV non-adherence may also occur if people are turned away from their clinic due to limited services e.g., services are denied if health care workers are unable to wash their hands prior to patient exams and blood draws, store blood specimens and clean equipment (Cronk and Bartram, 2018) and administer patient care including medications.
Botswana was the first country to provide free ARVs to its residents living with HIV but it continues to be among countries with the highest HIV prevalence in the world and is therefore, the focus of this study. The following is an overview of climate variability in Botswana.
Temperature changes and rainfall patterns
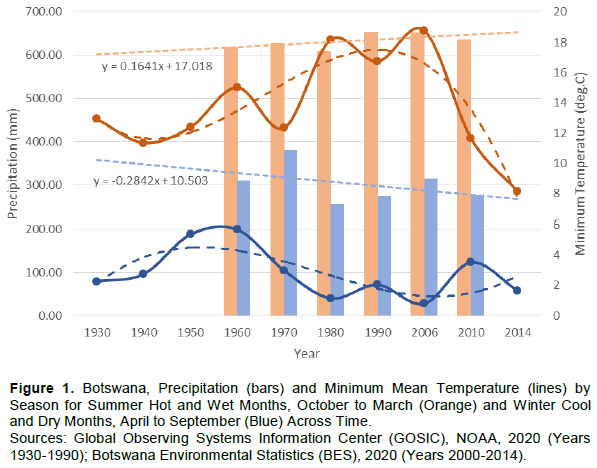
Botswana covers approximately 581,729 km2, and is situated between Namibia, Zimbabwe, and South Africa. Botswana is an arid to semi-arid country, that is drought-prone, with highly erratic rainfall (Botswana Environmental Statistics (BES), 2020) and high evaporation rates that exceed precipitation (BES, 2020). Comparable to other arid to semi-arid countries, the diurnal range in temperature is high. Most rainfall occurs between October and March, which is also the summer or time-period of hotter temperatures. Across Botswana, the minimum summer temperatures have risen slightly since 1930; and after 1990 a decline in rainfall is observed (Figure 1). During the winter months, April to September there is very little to no rainfall with cooling of temperatures. The temporal trend in minimum temperatures during these months decreased slightly after 1930; with a slight upward trend in rainfall since 2006 (Figure 1).
Water supply for domestic and clinic use
Botswana’s surface water resources are restricted to ephemeral and perennial rivers, dams and small lake-reservoirs. Water Utilities Corporation (WUC) under the Ministry of Minerals, Energy and Water Resources (MMWER) is responsible for water production in Botswana. Most surface water run-off cannot be captured due to high variability of run-off in-time and high evaporation (Department of Water Affairs (DWA), 2005). The Okavango and Chobe Rivers in the north are the only two perennial rivers. Ephemeral rivers are in the east, interior and south. There are nine main dams operated by WUC along ephemeral rivers. In general, surface water resources supply urban area and some peri-urban villages. Botswana’s groundwater resources provide 55.5% of the national demand for fresh water, while rivers and dams 5.6 and 39.9%, respectively (UNDP, 2012). The reliability is reduced during droughts (when groundwater levels drop and dam inflows are less) and when supply systems are over-stretched. The estimated combined sustainable yield of Botswana’s boreholes and storage dams are 2,165 mm3/annum or 216 L/person/day, based on the 2011 national population census estimate of n=2,024,904 people (MMWER, 2013). This is less than the current water demand of around 200 mm3/annum or 262 L/person/day (MMWER, 2013).
Power (electricity) plays an important economic role in the extraction of water from boreholes and dams countrywide. The national power grid is mainly concentrated along the railway line in the eastern side of the country where a majority of people live (BES, 2017). Rural populations residing on the outskirts of the grid receive diesel-powered generators to fuel the boreholes that supply water in these remote villages (BES, 2017). Due to Botswana’s population growth in recent years, power demand has increased leading to frequent power cuts (Sekantsi and Timuno, 2017).
Rainwater harvesting
In Botswana, rainwater harvesting has been used at a small scale in households. The adoption of the traditional knowledge and practices in regards to rainwater harvesting is very limited due to modern provisions by the government (Selabe and Minyoi, 2018). It is only recently that there has been a renewed interest in rainwater harvesting projects in Botswana, but this initiative is still in its infancy stage (Botswana Technology Centre (BTC), 2006). To cultivate this culture, the Government of Botswana has built rainwater resevoirs at many schools, clinics and public offices across the country (Selabe and Minyoi, 2018). Selabe and Minyoi (2018) found that a reluctance to practice rainwater harvesting was based on a false impression that water is abundant per the government’s supply and also readily available (Selabe and Minyoi, 2018). Based on the limited literature on this topic, there appears to be a need for additional incentives to encourage rainwater harvesting as well as an evaluation of current rainwater harvesting initiatives in Botswana.
Water rationing
In addition to water production, the WUC is responsible for water rationing, that is, limiting water use due to concerns of low water supply, in particular during droughts and/or maintenance of the water distribution infrastructure, which can be short or long-term. Water management centers that serve villages, will give notice of the days during the week that water will be rationed, that is, taps will be turned off. In 2015-2016, the risk of drought was high (BES, 2017) and many parts of the country experienced water rationing to preserve the limited water supply. According to the MMWER (2015) the Greater Gaborone area’s average demand surpassed supply by 32.9 million liters a day (MMWER, 2015). Even with rationing, the demand surpassed supply by 18.2 million liters a day (Gondo and Kolawole, 2019). During water rationing in very remote areas that are off the electrical grid, water management centers will provide their respective villages with water bowsing to augment water supply deficits (Electrical Generation and Distribution (EGD), 2018). Water in bowsers (big tanks) are transported to different villages by road. Parastatals and the private sector assist the WUC to provide water bowsers, flatbed trucks and water tanks to avail water to water stressed villages during times of water rationing.
Purpose of study
The purpose of this study is to investigate the impacts of climate variability, that is, high ambient temperatures and water scarcity and rationing (herein, also referred to as environmental exposures) on ARV non-adherence, for adults living with HIV in Greater Gaborone, Botswana. Botswana is studied because it is a landlocked arid to semi-arid country in Southern Africa that is experiencing rising temperatures and water scarcity, with increasing water rationing alerts. Botswana’s population is also relatively small, with about 2 million people and HIV prevalence rate 20.3 (range, 17.3-21.8), that is, about one-fifth of Botswana’s population is HIV infected (UNAIDS, 2019). ARV adherence is thus, a very important strategy for eliminating HIV in Botswana.
Gaps in the literature
Studies that have investigated the relationship between HIV/AIDS and water have focused on hygiene and sanitation (Obi et al., 2006; UNICEF, 2008; Nkongo and Chonya, 2009) and the reported benefit of potable water and improved sanitation to reduce diarrheal comorbidity (Obi et al., 2006). Contaminated water supply and crowded living conditions (Makaudze, 2016) especially where tuberculosis (TB) and pneumocystis are endemic, lead to favorable conditions for opportunistic infections (Hsu et al., 2005; Fewtrell et al., 2005; Yates et al., 2015; Nkongo and Chonya, 2009). Other direct and indirect factors that may explain the relationship between water scarcity and HIV/AIDS include multiple stressors as defined by Mbereko et al. (2016) such as high ambient temperatures, food insecurity, poverty and other structural barriers. A study in fishing villages (n=22) of Ngamiland, Botswana found that 53.0% of 248 households, reported caring for individuals with HIV in the last five years (Ngwenya and Mosepele, 2007). Of these, 29.0% of households reported the illness had a serious impact on their fishing activities, which indirectly impacted their ability to care for their HIV infected family member. The authors concluded that social protection measures that prevent the poor from becoming more marginalized should be prioritized for households in high HIV prevalence villages (Ngwenya and Mosepele, 2007). Literature on the environment and ARV medications specifically, have focused on food insecurity (Drimie and Mullins, 2002; Frega et al., 2010; Singer et al., 2015; Rawat et al., 2014; Weiser et al,. 2014) and ARV absorption and transportation systems (Young et al., 2014; Faturiyele et al., 2018; Hong et al., 2014) by which to access HIV services. There is a timely need to further understand how high ambient temperatures and water shortages and rationing impact ARV adherence, for people living with HIV in Botswana. The findings from this study will inform the “Treat All Program” in other arid to semi-arid countries also experiencing high HIV prevalence and increasing climate variability.
Study area
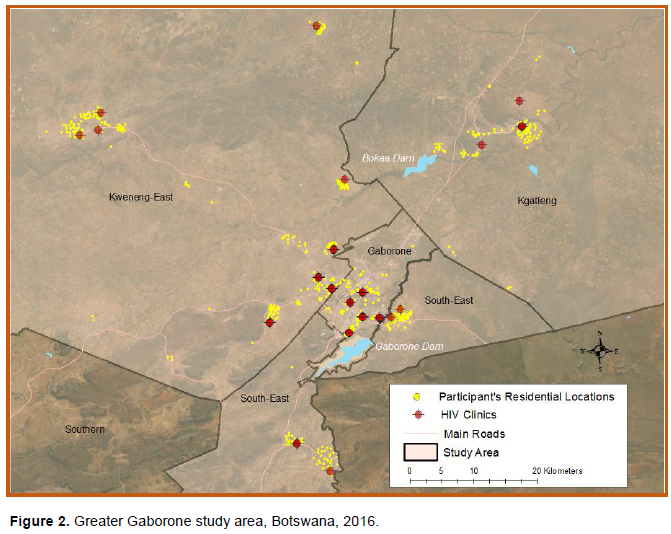
Greater Gaborone, Botswana includes Gaborone, a district and capital city, and three surrounding districts: South East, Kgatleng and Kweneng East (Figure 2) comprising about 421,907 people (Botswana Census, 2011). While Gaborone is classified as urban, South East district contains peri-urban villages and Kgatleng and Kweneng East are rural districts. Characteristics of the population in Greater Gaborone include HIV prevalence ranging from 16.6 per 100 population in South East district to 21.5 in Kweneng East. Although considered rural, Kweneng East has the largest population (n=256,759) and number of clinics (n=30) due to migration from Gaborone. Gaborone however, has the most residents with piped indoor water (58.4%) followed by South East (51%) and Kweneng East (23.2%) districts (Botswana Census, 2011).
Sampling of clinics and repondents
This study utilized a cross-sectional survey conducted in 2016 and face-to face interviews with (n=658) adults attending 21 HIV clinics in Greater Gaborone. The study took place during the winter season (May-August) when temperatures were cool (Figure 1) and there was no rainfall. The primary data was collected in 2016 when Botswana was still using the CD4 count threshold system to enroll the HIV patients into care. Since early 2017, the CD4 count system has been replaced with the “Treat All Program” (PEPFAR, 2019). The CD4 count therefore, plays a vital role in understanding ARV adherence.
The clinics were purposively selected from urban, peri-urban, and rural villages. To calculate the number of clinics needed, the national HIV prevalence (22.0) was multiplied by the number of ARV clinics (n=84) in the study’s area (0.22×84) = 18 clinics. To account for under sampling, this number was increased to 21 clinics for this study. The number of required adult women and men (n=667) attending these clinics was calculated using the formula adopted from a national survey conducted on HIV/AIDS stigma in 2014 (Botswana Stigma Index Survey Report, 2014) (Supplemental File 1). Following the selection of clinics and participants, a systematic random sampling was employed to select patients at their respective clinics. At each clinic, every second patient/mother to enter the door was selected for interview until the minimum required sample for each clinic was reached. The selection of pregnant women and mothers also used purposive sampling, with at least two women selected at each clinic. To complement the information that was gathered from patients, semi-structured interviews were conducted with four purposively selected health care workers from each clinic (doctor, two nurses and a pharmacist). These four health workers in each clinic were selected also because they were the only primary contacts that patients saw on their monthly appointments. After removing 9 outliers, the final dataset consisted of 658 adult women (n=419) including 67 pregnant women or mothers, and men (n=239) and health care workers (n=84).
Ethical clearance
All participants were asked to sign an informed consent to participate in the study. For inclusion criteria, only participants 18 years old and older who were HIV positive and enrolled in a HIV clinic took part in the study. The questionnaires were administered by the principal author and four research assistants in Setswana (local language) and English. The study was approved by the Michigan State University Internal Review Board (IRB). After obtaining IRB approval from the Ministry of Health and Wellness (MoHW) in Botswana, separate IRBs were also sought for each HIV clinic from the District Health Management Teams in Botswana.
Statistical analyses
In 2015, treatment and management of HIV in Botswana was available through a fixed dose combination of Atripla-combination of tenofovir (TDF) TDF/emtricitabine (FTC) or 3TC/ efavirenz (EFV) for all HIV positive patients regardless of their CD4 count and/or reproductive status (MoHW Botswana, 2016). In 2016, recommendations were that all HIV positive naïve pregnant women be placed on ARV for themselves and their baby’s welfare (PMTCT) as soon as possible. PMTCT is a comprehensive therapy, which means that other than the drugs provided for the woman, there is prophylaxis for HIV exposed children, periodic HIV testing to monitor the baby as well as partner testing and counselling. The risk of mother-to-child transmission can be significantly reduced by providing ARVs as lifelong therapy or as prophylaxis for the mother during pregnancy and labor and delivery, with antiretroviral prophylaxis for the infant, and antiretroviral drugs to the mother or child during breastfeeding (if breastfeeding), and use of safe delivery practices and safer infant feeding. In the context of this study, PMTCT is referred to as both a lifelong therapy and prophylaxis.
ARV adherence was modelled as a composite dichotomous variable defined by two questions: (1) “Are you taking your pills as instructed by your doctor?” (No vs. Yes) and (2) “Do you miss a dosage sometimes?” (No vs. Yes) and a biological measurement (3) CD4 count. If the participant did not take their pills as instructed, or missed dosage(s) of their medication or their CD4 count from their last clinic visit was < 350 copies/dL, they were considered ARV non-adherent (code=1). ARV non-adherence was the dependent variable used in subsequent regression models. This composite non-adherence variable was used because literature (Mannheimer et al., 2006; Atanga et al., 2018; Bermudez et al., 2016) has shown that rigorous study of measurement techniques of ARV adherence in clinical care settings is limited. This is generally because measurement of ARV adherence to long-term ARV ntherapy is complex (Pednekar et al., 2019) and requires longitudinal assessment. Additionally, there is no gold standard for ARV adherence and evaluation usually entails an assessment of construct validity (Liu et al., 2001).
The environmental exposures were measured using participant’s responses from the survey (Supplementary File 2). The participants were asked to recall their environmental experiences over a period of 30 days prior to their enrollment of this study. This time frame was used as a threshold because patients attended clinic appointment on a monthly basis. The environmental exposures were studied in relation to the participant’s residential location as well as at the clinic they attended.
Other potential confounding variables included gender (female=0, male=1), mode of transport with public transport=1, walking=2 and private car=3, type of clinic the patient attended, urban=1, peri-urban=2 or rural=3, and residence, urban=1, peri-urban=2 or rural=3. The unadjusted and adjusted odds of each environmental exposure on ARV non-adherence and 95% confidence intervals were estimated using logistic regression and for the adjusted models, controlling for potential confounding variables. These analyses were also stratified by gender. Two-level multilevel models were estimated to further understand the impacts of the environmental exposures on participant’s ARV non-adherence nested within the clinics they attended, controlling for potential confounding variables. The full maximum likelihood was used examining the improvement in model fit using the Akaike’s Information Criterion (AIC) with smaller values representing better fitting models.
Finally, the health care workers were asked open-ended questions (Supplemental File 2) to study emerging themes about clinic water shortages. To conduct a content analysis, steps recommended by Braun and Clarke (2006) were followed. Direct quotes were used where necessary. Pregnant women and mothers were asked three open-ended questions (Supplemental File 2) centered around their concerns and concerns for their babies. The residential locations of participant’s and the clinics they attended were geocoded and offset to preserve confidentiality in ArcGIS v.10.6 (ESRI, 2020). All survey data were translated from paper to digital format in SPSS (IBM Corp., 2017) and coded and analyzed in SAS v. 9.3 (SAS, 2020).
Table 1 shows the unadjusted odds of ARV non-adherence for adults attending HIV clinics in urban, peri-urban and rural locations. In urban areas, there were n=126 participants, of which 52 (41.3%) were ARV non-adherent. Very few participants residing in urban areas missed a clinic appointment due to heat or water rationing. While 68 participants reported that water rationing affected their health, less than half of these n=29 (42.6%) reported to be ARV non-adherent, which was not significantly different from those who where ARV adherent (OR=1.13, 95% CI 0.55-23.41). In peri-urban areas, there were n=160 participants, of which 69 (37.5%) were ARV non-adherent. For participants who missed a clinic appointment due to water scarcity, the odds of ARV non-adherence was OR=5.4 (95% CI, 1.06-27.91). Of the 94 participants who reported that water rationing affected their health, there were non-significant differences between those who were ARV non-adherent, n=37 (39.4%) and those who were ARV adherent (OR=1.21, 95% CI 0.63-2.33). In rural areas, there were n=372 participants, of which 120 (32.3%) were ARV non-adherent. Participants in rural areas were significantly more likely to be ARV non-adherent if they missed a clinic appointment due to heat (OR = 11.86, 95% CI = 3.36-41.81) or were turned away from a clinic due to water shortage (OR = 4.98, 95% CI = 1.69-14.69).
Table 2 shows that both women and men had a higher odds of ARV non-adherence if they missed a clinic appointment due to heat (OR = 3.49, 95% CI 1.43-8.52 and OR = 4.67, 95% CI 1.26-17.18). A higher percentage of men compared to women reported that water rationing affected their health (45.7% vs. 28.5%). While there were a similar number of women (n=14) and men (n=13) who were turned away from clinics due to water shortage, men who were turned away were significantly more likely than those not turned away, to be ARV non-adherent (OR = 4.20, 95% CI 1.12-15.67).
Multilevel modelling results
Table 3 shows that missing clinic appointments due to heat was the most significant risk factor for ARV non-adherence ( 0.2224 p-value 0.008), controlling for participant’s sex, distance traveled from home to the clinic and the other environmental exposures. Table 4 shows that the effect of missing a clinic appointment due to heat on ARV non-adherence was exacerbated in rural areas ( 0.4264 p-value 0.003). Missing a clinic appointment due to water scarcity in rural areas ( 0.1110 p-value 0.0600) or being turned away from a clinic due to water shortage in rural areas ( 0.2143 p-value 0.0790) were borderline but not significant, controlling for sex, distance traveled and rural residency.
Content analysis of open-ended responses by mothers
Mothers indicated that 2016 was drier (n=55) and very hot (n=47) compared to the previous years. Concerning the ambient heat and missing clinic appointments, a young pregnant woman of Hatsalatladi Village, Kweneng East district stated:
“On really hot days it’s tiring to take the bus to the clinic, and then wait in line to see the doctor, so sometimes I don’t go” (young woman age, 29 Kweneng East-Rural).
In addition, three mothers reported their experience with water rationing as, missed an appointment due to lack of water (n=1); sent home without service at the clinic due to water shortage (n=1) and unable to prepare baby formula because of lack of water (n=1). When a mother of two children was asked how water rationing affected her, she replied:
“When the water is dry in my village and I can’t Bathe my baby and myself, I feel embarrassed to go to the clinic somedays” (mother, age 34, Taung Village, South East-Peri-urban).
A young woman who was unable to prepare baby formula because of water rationing further said:
“The nurses told me to avoid mix feeding because my CD4 count was fluctuating for some time after giving birth. But because water went [cut] without any warning for the whole day, I could not starve my baby. I breastfed her. I did not want to do it, but I had no choice” (mother, age 39, Kgatleng-Rural).
Content analysis of open-ended responses by healthcare providers
Healthcare providers (n=84) reported that high temperatures posed a constraint in their health care facility. A pharmacist in Gaborone clinic reported:
“Our drugs expire really quickly because of being exposed to high temperatures” (Pharmacist, in Gaborone, Urban Clinic).
Meanwhile a doctor in peri-urban clinic of Mmopane, Kweneng East commented:
“On very hot days, blood samples expire even before we have the chance to send them to the technical labs” (Doctor, Peri-Urban Clinic).
High temperatures also slowed health care services in other departments within the clinic, as reported by a nurse in a rural clinic:
“The women’s section run slow when it’s too hot because they [pregnant women] are required to provide a lot of fluid samples” that take time (Nurse, Rural Clinic).
Healthcare providers were asked about clinic attendance on hot days, and whether missing a clinic appointment due to heat can lead to ARV non-adherence. While a small (n=19) number of healthcare providers indicated that heat did not have any impact on clinic attendance there were more, n=65 (77.3%) healthcare providers who indicated heat did have an impact on clinic attendance.
“Yes, they miss appointments because of the heat particularly the women with small babies” (Nurse, Rural Clinic).
Most health providers (n=70) were particularly concerned about mothers ‘attendance during hot days’. A nurse in the Kgatleng district rural clinic commented:
“Mothers also tend to skip their appointment when it’s too hot because most of the women walk to this clinic “(Nurse, Rural Clinic).
Of the 84 health care providers, n=17 (20.2%) from seven HIV clinics indicated that water was not an issue in service provision because a water reservoir used as a backup for water was replenished with rainfall or purchased water. These providers also said that they never turned away a patient, without providing services because of lack of water. Other health care workers, n=67 (79.7%) reported that water rationing was a hindrance in their HIV clinic. Doctors across clinics reported of a poor water supply that could be shutdown “at any time” without prior notification from Water Utilities Corporation. Pharmacists also reported unreliable water supply, that “could sometimes go for days with just slow drops from the taps”. Nurses indicated that they “longed for a backup water plan such a Jojo” (water reservoir, locally called by its brand name) that are found in urban clinics and hospitals.
When asked how water rationing interrupted their day-to-day activities at the clinic, a doctor in a rural clinic replied:
“We tend to see weak patients when there is no
water because facilities such as toilets are unusable, and this forces patients to use unsanitary toilets that are a breeding places for opportunistic infections” (Doctor, Rural Clinic).
Other services such as blood screening were halted on water rationing days.
“We do not screen for blood which is important for their CD4 count and viral load when there is no water, so it really throws us out of schedule” (Nurse, Rural Clinic).
While high temperatures and water scarcity and rationing did not have a significant impact on ARV non-adherence in urban areas; water rationing had a significant impact on ARV non-adherence in peri-urban areas; and heat and being turned away from clinics due to water shortages had a significant impact on ARV non-adherence in rural areas. Across all residential locations, participants reported their health was affected by water scarcity, which may have contributed to ARV non-adherence for some, but overall this was not a significant risk factor for ARV non-adherence. This finding suggested that people living with HIV continued to take their ARV medications despite not feeling well during periods of water scarcity.
Missing clinic appointments due to heat
Missing clinic appointments due to heat was a risk factor for ARV non-adherence for both women and men, and people living with HIV in rural areas in the unadjusted models. In the adjusted models, missing clinic appointments due to heat was the most significant risk factor for ARV non-adherence, controlling for participant’s sex, distance traveled from home to clinic and water scarcity and rationing. The effect of missing clinic appointments due to heat on ARV non-adherence was further exacerbated in rural areas. The reasons participants gave that made it difficult to comply with clinic appointments, were the long waiting lines and how tired one woman felt when she had to wait in the clinic heat for her appointment. That experience of waiting in the heat left an impression on her, such that on days of her appointment, she reported not wanting to attend if it was very hot, anticipating the long wait time in the heat.
From the health care workers perspective, women with children also have difficulty waiting for their appointments in the heat. Although some women commented that taking the bus or walking to the clinic in the heat was a reason for missing their clinic appointment, travel distance was not a significant risk factor for ARV non-adherence. This finding is corroborated by Munyaneza et al. (2018) in their study in rural Rwanda, who found that cost surface distance was not associated with either program outcome or with missing three consecutive visits (Munyaneza et al., 2018). The cost surface distance was defined as the distance between location of village and ART health center weighted by topography, calculated using a 2008 (90 m × 90 m resolution) digital elevation model (Munyaneza et al., 2018).
These findings suggest that adding an air conditioner or fan in rural clinic’s waiting rooms could be an incentive for women and mothers of children to attend their monthly clinic visit on hot days, knowing that when they arrive they can wait in comfort for their appointment. Air conditions and fans are already present in storage areas of rural clinics to protect the blood samples and ARV medications from heat damage, and they are in some examination rooms. Therefore, the electrical infrastructure of most clinics is capable of adding an air conditioner or fan. A programmatic needs assessment of rural clinic’s infrastructure, to assess the electrical capability however, may be warranted to ensure ARV adherence for people living with HIV, in rural Botswana. Furthermore, during times of electrical outages, each clinic could be provided with a generator(s) to ensure continued operation of the clinic during hot days or when there is water scarcity and rationing.
Missing clinic appointments due to water scarcity
Missing a clinic appointment due to water scarcity was a significant risk factor for ARV non-adherence in peri-urban villages in the unadjusted model; however there were few participants in peri-urban villages who missed clinic appointments due to water scarcity, to confidently confirm this relationship. In the adjusted model, missing a clinic appointment due to water scarcity was not a significant risk factor for ARV non-adherence when studied independently; and for participants residing in rural areas that relationship was also not significant, controlling for participant’s sex, distance traveled from home to the clinic and other environmental exposures. This implied that participants had water connections in their homes, but they experienced water rationing on other days. During the days where water was not available, they relied on other sources other than the home supply for water. This finding was supported by a participant who expressed concern about not being unable to bathe her baby and herself, in preparation for her appointment and thus, she was embarrassed to attend her clinic appointment. In a South African study (Galvin and Masombuka, 2020), these authors found that many peri-urban areas and townships have formal water connections in their houses but can experience water ration for hours or even days. Households (45%) reported water rations of longer than two days (Galvin and Masombuka, 2020). Expanding the WASH program to include concerns about hygiene and dignity in relation to monthly clinic visits, would help to promote ARV adherence. For example, clinics could provide free showers for patients who arrive from rural areas, where water rationing is occurring. At the individual/population level, patients can have reservoirs ready for rainwater harvesting during rainy seasons.
Mothers in this study reported feeling that year 2016 was hotter and drier compared to previous years, an observation supported by climatological records (BES, 2017) and report (Rural Development Council (RDC), 2017). Mothers and children are at greatest risk of poor health due to lack of water, as children’s lives are in danger if mothers do not have clean water to prepare sanitary formula milk and children’s body size is sensitive to dehydration. When there is water rationing, mothers may be forced to mix-feed (breastfeed and bottle feeding) which is not advisable when a mother is ARV non-adherent and does not have a suppressed viral load. Aibibula et al. (2017) found that incomplete HIV viral suppression is a threat to both individual health status and HIV control measures by destroying the immune system and the potential for ongoing HIV transmission (Aibibula et al. 2017). In their Cameron study, Atanga et al. (2018) found that HIV positive breastfeeding mothers on treatment who had high viral load, also had low ARV adherence (Atanga et al., 2018).
Even though in Zambia, water scarcity did not cause patients to miss HIV clinic appointments, challenges related to fetching water did (USAID, 2014). A study conducted in Zambia showed that people queued for water at communal taps caused further challenges for people living with HIV who often missed their medication schedules, whilst waiting for their turn to fetch water (USAID, 2014). Furthermore, water availability was worsened by erratic electricity supplies that affect the pumping of water (USAID, 2014). Other challenges related to water were voiced by the mothers in this study. These findings suggest that the WASH program can be expanded to include continued assessment of borehole placement as rural populations grow and become move mobile in rural Botswana. Women in this study can make sure they always have extra storage of water in their homes to be ready for when there is unannounced water rationing in their area.
Water scarcity affecting health
Over half of women (57.0%) and men (57.7%) reported that water rationing negatively affected their health, although this effect was not statistically associated with ARV non-adherence. Over half of all participants residing in urban (53.9%), peri-urban (58.8%) and rural (57.7%) areas also reported that water rationing negatively affect their health, but this effect was also not statistically associated with ARV non-adherence. This finding highlights that water rationing affect people’s lives even if it may not directly be linked to medication adherence. In 2014, USAID showed that rural areas still faced the greatest challenges in accessing clean water and sanitation facilities, such as toilets (USAID, 2014). The country-specific findings illustrated that many factors were involved in limiting improvements in water and sanitation services in rural areas, including low socioeconomic status, culture and drought. Botswana has frequent water shortages and rationing. The WUC (2016) water rationing schedule illustrated that water supply was cut in the Greater Gaborone area for seven hours, three days a week during 2016. These hours were from 0830 in the morning to 1530 in the afternoon. ARV clinics in the area operated from 0800 am to 1630 pm, which means that patients had no water available to them at their home or clinic. Rural areas further carry the burden of water shortages, with some villages without water supply at all and others that rely on bowsers provided by WUC. Future research should continue to investigate the health impacts of water scarcity for people living with HIV in rural Botswana.
Clinical services availability during water shortages
Health care providers are the caretakers of the patients, thus their reflections on how high temperatures and water shortages impact the care of their patients, was important for this study. These findings showed that men where significantly more likely to be ARV non-adherent after being turned away from their clinic during a water shortage. Monthly appointments constitute a checkup by the doctor, blood work for CD4 count and viral load, and based on those results, a medication refill. Even though men indicated that they had been turned away from receiving services due to water shortage, some health care providers affirmed that they continued to provide daily services to their patients regardless of water shortages; while other health care providers commented that “water shortages were a hindrance to their day-day running of the health facility” and services such as blood screening for CD4 count and viral load were not provided, which impacted ARV adherence levels of their patients. These disparate findings suggest that limited services during water shortages should be communicated effectively by health care providers to men in particular, to ensure additional follow-up of care.
These findings also confirm that even when patients have a high level of commitment to ARV adherence, high temperatures and water scarcity may impede their ability to attend their monthly clinic visit, particularly among women with children, thereby reducing the likelihood of achieving ARV adherence. Future strategies to adapt to rising temperatures could include adding an air conditioner or fan in rural clinic’s waiting rooms as an incentive for women and mothers of children to attend their monthly clinic visit, knowing that when they arrive they can wait in comfort for their appointment. Air conditions and fans are already present in storage areas of rural clinics to protect the blood samples and ARV medications from heat damage, and they are in some examination rooms. Therefore, the electrical infrastructure is most likely already available in clinics. During times of electrical outages, each clinic could be provided with a generator(s) to ensure continued operation of the clinic. Further expanding the WASH program to include concerns about hygiene and dignity in relation to monthly clinic visits, would help to promote ARV adherence. For example, clinics could provide free showers for patients who arrive from rural areas, where water rationing is occurring. These improvements in clinic infrastructure, would help people living with HIV, adapt to climate variability.
Healthcare providers also expressed concern that when there was no water, patients tended to use dirty toilets which was conducive to opportunistic infections, such as diarrhea. These findings are corroborated by Haftu et al. (2017)’s evaluation study of WASH in Ethiopia that showed in areas where water was not improved, diarrhea was 10 times higher among people living in these areas. The authors emphasized that special attention should be given to people living with HIV as they often experience a slower recovery from opportunistic illness such as diarrhea when compared to the general population (Haftu et al., 2017). In Kibera, Kenya, 94% of HIV positive people had suffered from an opportunistic illness due to exposure to contaminated water and poor environmental sanitation (Aketch et al., 2016). Another study in Ethiopia (Yallew et al., 2012) found that patients who have had diarrhea in the past 24 hours were six times more likely to have unimproved water status as compared to those who did not have diarrhea for the past 24 hours. For those suffering from diarrhea, there is an urgent need for accessible and frequent use of a toilet, in addition to an adequate supply of safe drinking water to prevent dehydration and further illness. Therefore, it is necessary to have a backup water supply such as a reservoir tank for those ARV clinics that do not have it. If toilets are not accessible at clinics, patients may be deterred from attending particularly when they are not feeling well, and this increases their medication non-adherence.
As previously mentioned, WUC rations water up to three times per week during water supply cuts in Greater Gaborone (WUC, 2016). It is recommended that this frequency be reduced to one or two times per week for ARV clinics. Clinics could also reduce the number of hours they provide services or start later and extend services into the evening. Warnings about water supply cuts could also be communicated more effectively to health care facilities so healthcare providers are prepared for those shutoffs. Most of the information about water rationing is found online in the WUC website, and social media pages. Most of Batswana listen to the radio and own cellphones, therefore, these communication platforms could be used to reach a wider community. At the individual level, patients should learn the culture of harvesting rain water during the rainy season because they will boil during time of need it to ensure its safety. Patients should also be encouraged to not rely on communication from water authorities, it should be a way of life to always have extra water reserve in their homes.
This study had some limitations. First, the stratified samples of participants (by location of residence and gender) who were ARV non-adherent were relatively small compared to similar participants who reported adhering to their HIV treatment regime. Therefore, the 95% confidence intervals around the odds of ARV non-adherence were inflated in the stratified unadjusted models. Stratification of the population however, provided some additional information about the characteristics of ARV non-adherent participants that was not captured in the multilevel models. In addition, the recall bias of those who participated in the study could have influenced their responses in terms of magnitude of environmental exposures, since the question was targeted to 2016 (time of the primary data collection) and the interview was conducted in Botswana’s cool and dry months. Finally, while the sampling methodologies for clinics and participants were thoughtful and robust, it is possible that adults who more routinely miss HIV clinic appointments were under-represented, thus knowledge of their specific risks for ARV non-adherence are still not fully understood. Despite these limitations, this study showed that high ambient temperatures and water shortage and rationing, had significant impacts on ARV non-adherence and further research is warranted.
As climate variability increases in Southern Africa’s semi-arid and arid countries that are also the home to four countries with the highest HIV prevalence in the world, strategies to minimize the impacts of higher ambient temperatures and water scarcity and rationing on ARV non-adherence are needed. The strategies within the current WASH and Treat All Programs should align to ensure safe water and hygiene practices that promote ARV adherence in rural and peri-urban villages. In Botswana, the WUC could accommodate water rationing schedules with HIV clinics, to optimize services, while also adding water storage-reservoirs and improving the electrical infrastructure, including generators for air conditioners and fans in waiting areas. Within HIV clinics, water-reservoirs could also provide for toilet accommodations and showers to provide for the basic needs of rural people attending the clinic. The efforts to integrate WASH and Treat All Program should acknowledge the lessons learned on existing integration initiatives from other sectors, such as those between HIV and nutrition. Finally, all key stakeholders such as government ministries (health, water) and the community should have a voice when WASH/Treat All Program integration initiatives are drawn. PEFRAR country-programs should continue to investigate the barriers of ART adherence, to eliminate new HIV infections and achieve the goal of having a free HIV generation by the target year of 2030.
The authors have not declared any conflict of interests.
REFERENCES
|
Aibibula W, Cox J, Hamelin AM, McLinden T, Klein MB, Brassard P (2017). Association between food insecurity and HIV viral suppression: a systematic review and meta-analysis. AIDS and Behavior 21(3):754-765.
Crossref
|
|
|
|
Aketch CO, Ngure K, Kiplangat JM (2016). The effects of water quality and sanitation on immunocompromised: A case study of people living with HIV/AIDS in Kibera slum, Kenya. East African Medical Journal 93(2):72-81.
|
|
|
|
|
Atanga PN, Ndetan HT, Fon PN, Meriki HD, Muffih TP, Achidi EA, Kroidl A (2018). Using a composite adherence tool to assess ART response and risk factors of poor adherence in pregnant and breastfeeding HIV-positive Cameroonian women at 6 and 12 months after initiating option B+. BMC Pregnancy and Childbirth 18(1):418.
Crossref
|
|
|
|
|
Bermudez LG, Jennings L, Ssewamala FM, Nabunya P, Mellins C, McKay M (2016). Equity in adherence to antiretroviral therapy among economically vulnerable adolescents living with HIV in Uganda. AIDS Care 28(sup2):83-91.
Crossref
|
|
|
|
|
Botswana environment statistics (BES) (2017). Botswana environment statistics. Climate Digest. Statistics Botswana, Gaborone, Botswana.
|
|
|
|
|
Botswana environment statistics (BES) (2020). Botswana environment statistics. Climate Digest. Statistics Botswana, Gaborone, Botswana.
|
|
|
|
|
Botswana Census (2011). Botswana population and housing census. Botswana Statistics. Department of Printing Public Service: Gaborone, Botswana 211:4.
|
|
|
|
|
Botswana stigma index survey report (2014). National AIDS Coordinating Agency, Gaborone, Botswana.
|
|
|
|
|
Botswana Technology Centre (BTC) (2006). Botswana Technology Centre. Feasibility study on rainwater harvesting. Proceedings of the Workshop on Project. Botswana Technology Centre, Gaborone, Botswana, March 2006, 8-9.
|
|
|
|
|
Braun V, Clarke V (2006). Using thematic analysis in psychology. Qualitative Research in Psychology 3(2):77-101.
Crossref
|
|
|
|
|
Cronk R, Bartram J (2018). Environmental conditions in health care facilities in low-and middle-income countries: coverage and inequalities. International Journal of Hygiene and Environmental Health 221(3):409-422.
Crossref
|
|
|
|
|
Department of Water Affairs (DWA) (2005). Department of Water Affairs. National water master plan review (vol 2), Government of Botswana, Gaborone.
|
|
|
|
|
Drimie S, Mullins D (2002). HIV/AIDS and land: case studies from Kenya, Lesotho, and South Africa. Report for Southern Africa Regional Office of the Food and Agricultural Organization (FAO), Human Sciences Research Council, Pretoria, South Africa.
Crossref
|
|
|
|
|
Electrical Generation and Distribution (EGD) (2018). Electrical Generation and Distribution. Statistics Botswana. Stats Brief, Quarter, Gaborone, Botswana.
|
|
|
|
|
Environmental Systems Research Institute (ESRI) (2020). ArcMap v. 10.6. Redlands California.
|
|
|
|
|
Faturiyele I, Karletsos D, Ntene-Sealiete K, Musekiwa A, Khabo M, Mariti M, Mahasha P, Zulu T, Pisa PT (2018) Access to HIV care and treatment for migrants between Lesotho and South Africa: a mixed methods study. BMC Public Health 18(1):668.
Crossref
|
|
|
|
|
Fewtrell L, Kaufmann RB, Kay D, Enanoria W, Haller L, Colford Jr JM (2005). Water, sanitation, and hygiene interventions to reduce diarrhoea in less developed countries: A systematic review and meta-analysis. The Lancet Infectious Diseases 5(1):42-52.
Crossref
|
|
|
|
|
Frega R, Duffy F, Rawat R, Grede N (2010). Food insecurity in the context of HIV/AIDS: A framework for a new era of programming. Food and Nutrition Bulletin 31(4_suppl4): S292-S312.
Crossref
|
|
|
|
|
Galvin M, Masombuka LN (2020). Failed intentions? Meeting the water needs of people living with HIV in South Africa. Water SA 46(2):242-251.
Crossref
|
|
|
|
|
Gondo R, Kolawole OD (2019). Sustainable water resources management: Issues and principles of water governance in the Okavango Delta, Botswana. International Journal of Rural Management 15(2):198-217.
Crossref
|
|
|
|
|
Haftu D, Gebremichael G, Ajema D, Gedamu G, Agedew E (2017). Water, sanitation and hygiene practice associated factors among HIV infected people in Arba Minch town, Southern Ethiopia. Journal of Water and Health 15(4):615-625.
Crossref
|
|
|
|
|
Hong SY, Fanelli TJ, Jonas A, Gweshe J, Tjituka F, Sheehan H MB, Wanke C, Terrin N, Jordan MR, Tang AM (2014). Household food insecurity associated with antiretroviral therapy adherence among HIV-infected patients in Windhoek, Namibia. Journal of Acquired Immune Deficiency Syndromes 67(4):e115.
Crossref
|
|
|
|
|
Hsu JW, Pencharz PB, Macallan D, Tomkins A (2005). Macronutrients and HIV/AIDS: A review of current evidence. Durban, South Africa: World Health Organization.
|
|
|
|
|
IBM Corp (2017). IBM SPSS Statistics for Windows, Version 25.0. Armonk, New York.
|
|
|
|
|
International Panel of Climate Change (IPCC) (2007). Climate Change 2007: Synthesis Report. Contribution of Working Groups I, II and III to the Fourth Assessment Report of the Intergovernmental Panel on Climage Change (IPCC). Geneva Switzerland:104.
|
|
|
|
|
Liu H, Golin CE, Miller LG, Hays RD, Beck CK, Sanandaji S, Wenger NS (2001). A comparison study of multiple measures of adherence to HIV protease inhibitors. Annals of Internal Medicine 134(10):968-977.
Crossref
|
|
|
|
|
Makaudze EM (2016). Measuring willingness-to-pay for water and sanitation by people living with HIV and AIDs in South Africa. Journal of Water, Sanitation and Hygiene for Development 6(1):161-169.
Crossref
|
|
|
|
|
Mannheimer SB, Mukherjee R, Hirschhorn LR, Dougherty J, Celano SA, Ciccarone D, Botsko M (2006). The CASE adherence index: A novel method for measuring adherence to antiretroviral therapy. AIDS Care 18(7):853-861.
Crossref
|
|
|
|
|
Marriott NG, Schilling MW, Gravani RB (2018). Food contamination sources. In Principles of Food Sanitation. Springer Nature:83-91.
Crossref
|
|
|
|
|
Mbereko A, Scott D, Chimbari JM (2016). The relationship between HIV and AIDS and water scarcity in Nyamakate resettlements land, north-central Zimbabwe. African Journal of AIDS Research 15(4):349-357.
Crossref
|
|
|
|
|
Mills JE, Cumming O (2016). The impact of water, sanitation and hygiene on key health and social outcomes. Available at:
View
|
|
|
|
|
Ministry of Minerals, Energy & Water Resources (MMWER) (2013). Ministry of Minerals, Energy & Water Resources. Botswana Integrated Water Resources Management & Water Efficiency Plan. Gaborone, Botswana.
|
|
|
|
|
Ministry of Minerals, Energy & Water Resources (MMWER) (2015). Botswana Water Policy. Gaborone, Botswana.
|
|
|
|
|
Ministry of Health and Wellness Botswana (MoHW) (2016). Handbook of the Botswana 2016 integrated HIV clinical care guidelines. Republic of Botswana, Gaborone, Botswana.
|
|
|
|
|
Munyaneza F, Ntaganira J, Nyirazinyoye L, Birru E, Nisingizwe MP, Gupta N, Hedt-Gauthier BL (2018). Community-based accompaniment and the impact of distance for HIV patients newly initiated on antiretroviral therapy: early outcomes and clinic visit adherence in rural Rwanda. AIDS and Behavior 22(1):77-85.
Crossref
|
|
|
|
|
Ngwenya BN, Mosepele K (2007). HIV/AIDS, artisanal fishing and food security in the Okavango Delta, Botswana. Physics and Chemistry of the Earth Parts A/B/C 32(15-18): 1339-1349.
Crossref
|
|
|
|
|
Nkongo D, Chonya C (2009). Access to water and sanitation for people living with HIV and AIDS: an exploratory study. Water Aid and AMREF.
|
|
|
|
|
National Oceanic and Atmospheric Administration (NOAA) (2020). Global Observing Systems Information Center (GOSIC) National Oceanic and Atmospheric Administration (NOAA) National Centers for Environmental Information.
|
|
|
|
|
Obi CL, Onabolu B, Momba MNB, Igumbor JO, Ramalivahna J, Bessong PO, Mulaudzi TB (2006). The interesting cross-paths of HIV/AIDS and water in Southern Africa with special reference to South Africa. Water SA 32(3):323-343.
Crossref
|
|
|
|
|
Pednekar PP, Agh T, Malmenäs M, Raval AD, Bennett BM, Borah BJ, Turcu-Stiolica A (2019). Methods for measuring multiple medication adherence: a systematic review-report of the ISPOR medication adherence and persistence special interest group. Value In Health 22(2):139-156.
Crossref
|
|
|
|
|
President's Emergency Plan for AIDS Relief (PEPFAR) (2019). Botswana Country Operational Plan 2019: Strategic Direction Summary. Gaborone, Botswana.
|
|
|
|
|
Rawat R, Faust E, Maluccio JA, Kadiyala S (2014). The impact of a food assistance program on nutritional status, disease progression, and food security among people living with HIV in Uganda. Journal of Acquired Immune Deficiency Syndromes 66(1):e15-e22.
Crossref
|
|
|
|
|
Rural Development Council (RDC) (2017). Rural Development Council. Drought and household food security outlook for 2016/17. Ministry of Local Government and Rural Development. Gaborone, Botswana.
|
|
|
|
|
SAS Institute, Inc (2020). Cary, North Carolina.
|
|
|
|
|
Sekantsi LP, Timuno S (2017). Electricity Consumption In Botswana: The Role Of Financial Development, Industrialisation And Urbanization. Review of Economic and Business Studies, Alexandru Ioan Cuza University, Faculty of Economics and Business Administration 19:75-102.
Crossref
|
|
|
|
|
Selabe M, Minyoi MK (2018). Perceived barriers to rainwater harvesting as a source of water supply in Botswana: case of Gaborone city. Botswana Journal of Technology 23(1):86-96.
|
|
|
|
|
Singer AW, Weiser SD, McCoy SI (2015). Does food insecurity undermine adherence to antiretroviral therapy? A systematic review. AIDS and Behavior 19(8):1510-1526.
Crossref
|
|
|
|
|
United Nations AIDS (UNAIDS) (2019). Joint United Nations Programme on HIV/AIDS (UNAIDS), Data and AIDS Epidemic update. Geneva, Switzerland.
|
|
|
|
|
United Nations AIDS (UNAIDS) (2020). Global Estimates. Available at:
View
|
|
|
|
|
United Nations Development Programme (UNDP) (2012). Botswana Water Sector Policy Brief, Gaborone, Botswana.
|
|
|
|
|
United Nations International Children's Emergency Fund (UNICEF) (2008). Why improved sanitation is important for children. Available at:
View
|
|
|
|
|
United States Agency International Development (USAID) (2011). Programming Water, Sanitation and Hygiene (WASH) Activities in U.S. Government Country Operational Plans (COPs).
|
|
|
|
|
United States Agency International Development (USAID) (2014). HIV, integrated approach to, sanitation water, and hygiene in southern africa. "a gap and needs assessment."
|
|
|
|
|
Water Utilities Corporation (WUC). (2016). Water Rationing Schedule of March 2016, Sedibeng House, Gaborone, Botswana.
|
|
|
|
|
Weiser SD, Palar K, Frongillo EA, Tsai AC, Kumbakumba E, DePee S, Bangsberg DR (2014). Longitudinal assessment of associations
|
|
|
|
|
between food insecurity, antiretroviral adherence and HIV treatment outcomes in rural Uganda. AIDS 28(1):115.
Crossref
|
|
|
|
|
Yallew WW, Terefe MW, Herchline TE, Sharma HR, Bitew BD, Kifle MW, Adane MM (2012). Assessment of water, sanitation, and hygiene practice and associated factors among people living with HIV/AIDS home based care services in Gondar city, Ethiopia. BMC Public Health 12(1):1057.
Crossref
|
|
|
|
|
Yates T, Lantagne D, Mintz E, Quick R (2015). The impact of water, sanitation, and hygiene interventions on the health and well-being of people living with HIV: A systematic review. Journal of Acquired Immune Deficiency Syndromes 68:S318-S330.
Crossref
|
|
|
|
|
Young S, Wheeler AC, McCoy SI, Weiser SD (2014). A review of the role of food insecurity in adherence to care and treatment among adult and pediatric populations living with HIV and AIDS. AIDS and Behavior 18(5):505-515.
Crossref
|
|