ABSTRACT
A cross sectional study was conducted between January and May 2015 in and around Kombolcha town, Eastern Amhara, to identify methicillin resistant Staphylococcus aureus and their resistance to antimicrobials and also to identify risk factors associated with the occurrence of dairy cow mastitis. A total of 150 dairy cows were included during the study period. A total of 600 quarters were examined to detect clinical and subclinical mastitis by physical examinations of udder and milk and California mastitis test, respectively. The prevalence of mastitis was 56%. Age, milking hygiene, parity, and floor system had significant (p<0.05) effect on the prevalence of mastitis. S. aureus was isolated at a rate of 11 (73.3%) and 29 (42%) in clinical and subclinical mastitis, respectively. The result showed the occurrence of S. aureus in clinical mastitis was found to be significantly higher than subclinical mastitis (p=0.028). The present study showed that S. aureus was resistant to penicillin G (100%), amoxicillin (100%), cefoxitin (42.7%) and tetracycline (77.4%) however all the isolates were found to be totally (100%) susceptible to the gentamycin. 45.3% of the isolates were found to be multidrug resistant. Antimicrobial resistance is due to repeated therapeutic use of drugs. Regular antimicrobial sensitivity testing helps to select effective antibiotics that ultimately reduce the development of resistance towards commonly used antibiotics. In conclusion, Mastitis caused by S. aureus is one of the major problems of dairy cows in milk production in the study area.
Key words: Antimicrobials, Mastitic cows, Methicillin-resistant Staphylococcus aureus, prevalence, risk factors.
Mastitis, inflammation of the mammary gland, is a costly production disease affecting the dairy cattle industry worldwide. Mastitis may be caused by either infectious or non-infectious agents. Infectious mastitis results from bacterial, mycotic or algal pathogens. Non-infectious mastitis is the result of injury, chilling, bruising or rough or improper milking. But it is almost due to the effect of infection by bacteria or mycotic pathogens. A total of about 140 microbial species, subspecies and serovars have been isolated from the bovine mammary gland. Pathogens causing mastitis in cattle are divided into major pathogens (those that cause clinical mastitis) and minor pathogens (those that normally cause subclinical mastitis and less frequently clinical mastitis (Firaol et al., 2013).
Staphylococcus aureus is a versatile pathogen of humans and animals that causes a wide variety of the disease (Abebe et al., 2013). The bacterium is a colonizer of the skin and mucosae from which it can invade multiple organs. In livestock S. aureus is an important cause of mastitis, skin and soft tissue infections (SSTI) and to lesser extent infections of the locomotory system. Surgical site infections (SSI) in which S. aureus is isolated have been increasingly reported in small companion animals and horses (Normanno et al., 2007).
In recent years, there has been increased concern about antibiotic resistant strains of S. aureus. Development of resistance has been attributed to the extensive therapeutic use of antimicrobials or to their administration as growth promoters in food animal production (Normanno et al., 2007). Isolates of S. aureus are frequently resistant to methicillin and essentially all other β-lactam antibiotics. The resistance to methicillin in staphylococci is mediated by the mecA gene that encodes a modified penicillin-binding protein (PBP), the PBP2a or 2’, which shows reduced affinity to penicillins, such as methicillin and oxacillin and for all other beta-lactam antibiotics. An organism with this type of resistance is referred to as methicillin-resistant S. aureus (MRSA). The mecA gene resides on a staphylococcal chromosomal cassette (SCCmec) (Kwon et al., 2006).
MRSA was initially reported as a nosocomial pathogen in human hospitals (hospital-associated MRSA) and was isolated from patients with compromised immune systems undergoing medical procedures. MRSA accounts for 30 to 40% of all hospital-acquired infections and for 40 to 70% of S. aureus infections in intensive care units (Gordon and Lowy, 2008). In the 1990s, a major change in the epidemiology of MRSA has been observed, with the appearance of cases affecting people with no epidemiological connection to hospitals; strains that cause such infections are referred to as community-acquired or community associated MRSA (EFSA, 2009).
Until recently, such strains were susceptible to many antibiotics other than β-lactams; however, resistance seems to be increasing, and multiple antibiotic resistant strains have started to emerge (Otter and French, 2010). There is now increasing concern about the public health impact of MRSA associated with food producing animals, because MRSA and, consequently, their resistance genes can spread from animals to humans by direct contact or through the food chain (Kluytmans, 2010). MRSA strains have been isolated in many countries from cows’ or small ruminants’ milk and various dairy products (Ünal et al., 2012).
Milk-secreting tissues and various ducts throughout the udder can be damaged by bacterial toxins, and sometimes permanent damage to the udder occurs. Severe acute cases can be fatal, but even in cows that recover there may be consequences for the rest of the lactation and subsequent lactations. The illness is in most respects a very complex disease, affected by a variety of factors: it can be present in a herd subclinically, where few, if any, symptoms are present in most cows. Practices such as close attention to milking hygiene, the culling of chronically-infected cows, good housing management and effective dairy cattle nutrition to promote good cow health are essential in helping to control herd mastitis levels (
Ricardo, 2011).
There are also some studies on MRSA in some part of Ethiopia such as in Hawasa (Daka et al., 2012), in Adama (Abera et al., 2013), in and around Addis Ababa (Abebe et al., 2013). Concerning the study area south wollo, in and around kombolcha, MRSA is not studied. Knowledge of MRSA is necessary to make decisions regarding antibiotic treatment and prerequisite for establishing control strategies in the area.
Therefore, this study was designed with the following objectives:
1) Isolation and identification of methicillin resistant S. aureus (MRSA) from mastitic cow’s milk.
2) Determining the occurrence of bovine mastitis in the selected dairy cows in the study area
3) Assessment of potential risk factors associated with the disease
4) Determining the antimicrobial resistance patterns of the isolates to the selected antibiotic discs.
Study area
The study was conducted at Kombolcha Town, which is found in South Wollo Administrative Zone of Amhara National Regional State in North Eastern Ethiopia. The study area is located 376 km north of Addis Ababa with 11°084'49''°N latitude and 0.39°737' 46''°E longitude at an altitude of 1840 meter above sea level (masl) (Figure 1). Based on the central statistical agency 2005, kombolcha town has an estimated total human population of 68,766 of which 36,102 are males and 32,664 females. The woreda has an estimated area of 8.66 km2, which gives kombolcha a density of 7940.60 people per square kilometer. Animal population includes 22,455 cattle, 9,537 sheep and 15,310 goats.
The kombolcha town experiences a bi-modal rainfall, the short rainy season occurs usually from March 15 to May. The minimum and maximum mean annual rainfall in and around kombolcha ranges from 750 to 900 mm. The average minimum and maximum daily temperature during short and long rains are 23.9 and 11.7°C, respectively and the relative humidity of the area varies from 23.9 to 79% (Kombolcha Town Agricultural Office).
Study design
A cross sectional type of study was conducted from January 2015 to May 2015 to isolate and identify methicillin resistance S. aureus.
Study population
The study animals were dairy cattle in and around Kombolcha town. Four breeds of cattle (Holstein-Friesian, Jersey, between the breeds and local (Zebu)) were included in the study.
Sample size
Purposive sampling technique was applied on all available dairy cows in the study area. A total of 150 dairy cows from 18 selected dairy farms in and around kombolcha were selected conveniently based on the availability of dairy cows.
Study methodology
Questionnaire survey
A questionnaire was developed and all information relating to the study objectives was recorded. Data were collected on potential risk factors for the occurrence of mastitis in dairy cows based on observation and by interviewing the farm owners or handlers. The animal level factors such as herd size, presence of teat lesion, teat blindness, body condition, parity, lactation stage, breed and age difference was recorded. The farm level factors such as housing types, farm hygiene, previous history of treatment of mastitis, barn floor status, type of milking method, use of towels, milking sequences and hygiene was recorded. Udder and milk abnormalities (injuries, blindness, tick infestation, swelling, milk clots, abnormal secretion, etc.) were also recorded.
Clinical examination of the udder
Udders of the selected dairy cows were examined by visual inspection and palpation for the presence of any lesion, pain, heat and swelling. In addition, milk from each quarter was withdrawn and checked for any change in color and consistency.
California mastitis test (CMT)
The California mastitis test (CMT) was conducted to diagnose the presence of subclinical mastitis and it was carried out according to standard procedures. A squirt of milk from each quarter of the udder was placed in each of four CMT paddle and an equal amount of the reagent, approximately 4-5 drop was added. A gentle circular motion was applied in a horizontal plane. Positive samples show gel formation within a few seconds. The result was scored based on the gel formation and categorized as negative if there was no gel formation and record as 0 (zero), or positive if there was gel formation ranging from trace (T) to +3. If at least one quarter was positive by the CMT then the cow was considered as positive (Quinn et al., 1994).
Sampling method
Strict aseptic procedure was followed when collecting milk samples in order to prevent contamination with microorganisms present on the skin udder and teats, on the hands of samplers and on the barn environment. Teat ends were cleaned and disinfected with ethanol (70%) before sampling. Strict foremilk (first jets) was discharged to reduce the number of contamination of teat canal (Quinn et al., 1999). Sterile test tubes with tight fitting cups were used. The test tube was labeled with permanent marker before sampling. To reduce contamination of teat ends during sample collection, the near teats were sampled first and then followed by the far ones (Quinn et al., 1999).
Milk samples were collected from each of clinically and subclinically mastitic non-blind quarters of the selected cows for bacterial isolation. About 10 ml of milk was aseptically collected from each mastitis positive quarter using sterile test tube. Then, samples were transported in an icebox to Kombolcha Regional Laboratory for microbiological examination. If immediate inoculation is not convenient, samples were kept at 4°C until cultured for isolation.
Laboratory work
Culturing and biochemical tests
A loop full of milk sample was streaked on 5% sheep blood agar and the plates were incubated aerobically at 37°C and examined after 24 h of incubation for growth. The colonies were provisionally identified on the basis of staining reaction with Gram's stain, cellular morphology and hemolytic pattern on blood agar. The representative colonies were sub cultured on nutrient agar and incubated at 37°C for 24 h. The isolated colonies from nutrient agar were exposed to Catalase test, slide or tube coagulase. Coagulase positive colonies were grown on manitol salt agar and S. aureus were isolated for anti-microbial susceptibility testing (Quinn et al., 1994).
Anti-microbial susceptibility testing
The S. aureus isolates were tested for anti-microbial susceptibility by disc diffusion method (Quinn et al., 1999). Drugs like cefoxitin (30 mg), gentamycin (10 mg), erythromycin (15 mg), streptomycin (10 mg), tetracycline (30 mg) ampicillin (10 mg) and penicillin G (10 units) were used for Anti-microbial susceptibility testing. Approximately 3 to 5 colonies isolated from pure culture were transferred into a test tube of 1ml tryptone soya broth (TSB) and incubated at 37°C for 24 h. The turbidity of the suspension was adjusted by adding 9 ml saline water. Muller-Hinton Agar plate was prepared and a sterile cotton swab was dipped into the suspension and swabbed on the surfaces of Muller-Hinton Agar plate. Then, the antibiotic discs were placed on the agar plate using disc dispenser. The plates were read after 24 h of incubation at 37°C under aerobic condition. However cefoxitin discs were incubated at 35°C for 24 h. The isolates were classified in accordance with the guideline of the National Committee for Clinical Laboratory Standards (CLSI, 2006) as susceptible, intermediate or resistance for each antibiotic tested according to the manufacturer’s instructions by measuring the zone of inhibition around the antibiotic disc. Intermediate results were considered resistant (Huber et al., 2011).
Data management and analysis
Collected data was coded and entered to MS Excel spreadsheet and checked for accuracy. After validation, it was transferred and processed using computer software SPSS version 20 for analysis. Pearson’s chi-square tests were used when appropriate to analyze the proportions of categorical data. Odds ratio and 95% CI were computed and the results were considered significant at p<0.05.
Quality assurance
Confidence in the reliability of test results is increased by following adequate quality assurance procedures, and the routine use of control 3503 strains, S. aureus ATCC25923 as a positive control and Escherichia coli ATCC-25922 as a negative control (for culture on MSA) were taken as an important part of quality control for culture and antimicrobial susceptibility test. Thus, quality control microorganisms yielded values within the established ranges, indicating that the test was performed in a satisfactory manner.
Prevalence of mastitis
From the total 150 lactating cows examined during the study period 84 (56%) cows had mastitis, of which 15 (10%) and 69 (46%) showed clinical and subclinical mastitis respectively (Table 1). Out of the 600 quarters examined, the quarter level occurrence of mastitis showed 202 (33.7%); from which 70 (11.7%) were found in front quarters and 132 (22%) in hindquarters. From the total examined quarters (600), 6 quarters were found blind (Tables 2 and 3).
Risk factors associated with mastitis
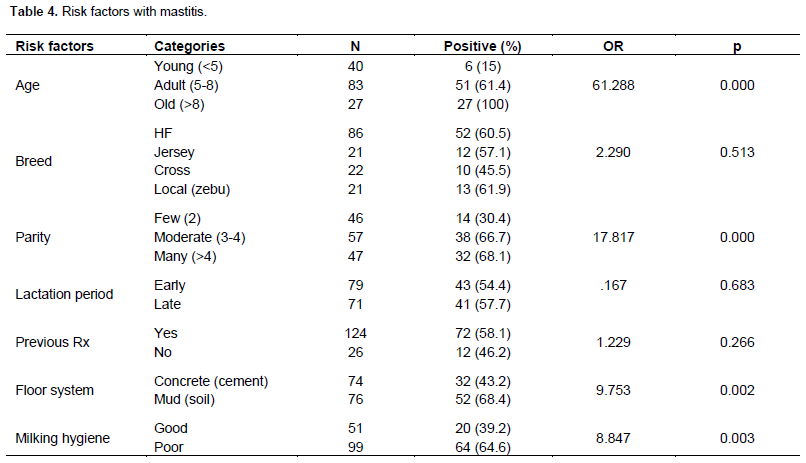
The questionnaire survey and observation data result shows age, parity, milking hygiene and floor are among the potential risk factors which are associated with mastitis (Table 4).
Percentage of S. aureus isolates
Milk samples collected from 84 mastitis positive cows (15 clinical cows and 69 CMT-positive subclinical cows) or from 202 teats were cultured on blood agar. Coagulase positive Staphylococcus species were isolated from 40 cows (53 quarters) from 54 cows (67 quarters) colonies cultured on manitol salt agar. S. aureus was isolated at a rate of 11 (73.3%) and 29 (42%) in clinical and subclinical mastitis, respectively. The isolates of S. aureus in clinical mastitis are found to be significantly higher than subclinical mastitis (Table 5).
Antimicrobial sensitivity test
Antibiotics of veterinary and human health relevance were considered in this study has demonstrated the existence of alarming levels of resistance of S. aureus to commonly used antimicrobial agents in the study area (farms). The present study has demonstrated the existence of alarming levels of resistance of S. aureus to commonly used antimicrobial agents in the study area (farms). From 53 S. aureus grown on muller-hinton agar 25 (47.2%) S. aureus were found to be resistant to cefoxitin which shows the prevalence of MRSA. The resistance pattern of both penicillin G, and amoxacillin and tetracycline is 53 (100%) and 41 (77.4%) respectively (Table 6). Out of the resistance S. aureus isolates, 24 (45.3%) were found to be multidrug resistance against 4 antibiotic discs primarily to penicillin G, ampicillin, cefoxitin, and erythromycin.
Cefoxitin resistance pattern with previous treatment
From a total of 18 (45%) cows, which shows resistance to cefoxitin 14 (77.8%) cows were previously treated. From a total of 22 (55%) cows that shows susceptibility to cefoxitin 17 (77.3%) cows were cefoxitin susceptible without previous treatment (Table 7). Therefore MRSA was found to be associated with previous treatment history of the animal with cefoxitin resistance.
Cefoxitin resistance pattern with age of the animal
From all age groups of animal old age cows were more often cefoxitin resistant S. aureus positive than cows of adult and young aged cows. 8 (80%) of the cows under old age category group are cefoxitin resistant (Table 8).
The study was conducted on small and middle-sized dairy farms in and around kombocha town to determine the prevalence of mastitis and asses the major risk factors associated with MRSA. The result revealed that an overall prevalence of mastitis 84 (56%) and 202 (33.7%) at quarter level. This result agrees with the previous researches conducted by Sori et al. (2005) and Lakew et al. (2009) who reported a prevalence of 52.78% in and around Sebeta and 64.4% in Asella, respectively. This report is also in consistent with the findings of
Radostits et al. (2000) that, in most countries and irrespective of the cause, the prevalence of mastitis was about 50% at cow level and 25% at quarter level.
The current finding of the study is comparably higher than the work of Kerro and Tareke (2003) who recorded 40% in cows and 19% in quarters in Southern Ethiopia. The infection rate in cows was also higher than that of Bitew et al. (2010) and Mulugeta and Wassie (2013), who reported an overall prevalence of 28.8% in Bahir Dar and 29.5% around Wolaita Sodo, respectively. Since mastitis is a complex disease and the difference in results could be due to difference in management system of the farm, difference in drug usage and the geographical locations of the studies.
The prevalence of clinical and subclinical mastitis in the present study is 10 and 46% respectively. In the current study the rate of subclinical mastitis (46%) is higher than that of the clinical mastitis (10%) which in agreement with the reports of Abera et al. (2013) (36.7% subclinical and 10% clinical mastitis) in Adama town. The result of subclinical mastitis (46%) is higher than that of Abera et al. (2013) with a prevalence of 36.7%. In most reports including the present study, clinical mastitis is far lower than subclinical mastitis. This could be attributed to little attention given to subclinical mastitis, as the infected animal shows no obvious clinical symptoms and secrets apparently normal milk. Therefore farmers are not well informed about invisible loss from subclinical mastitis. In Ethiopia, the subclinical forms of mastitis received little attention and efforts have been concentrated on the treatment of clinical cases (Almaw et al., 2008).
The occurrence of mastitis in front quarter and hindquarter was 11.7 and 22% respectively. Mastitis positive cow at quarter level with breed difference was insignificant. This shows that breed difference is not as risk factors for the occurrence of mastitis in hindquarters than front quarters rather than the teat is exposed to contamination. However milking hygienic practice at quarter level mastitis is significant in the hindquarter and has contribution for the occurrence of the infection. Therefore hindquarter is more affected than the front quarter and the current result is comparable with the finding of 19% in quarters by Kerro and Tareke (2003). This is due to the fact that the hindquarters are highly predisposed for contamination with dirt. In addition to this, large amount of milk is produced from hindquarters and as a result the pressure on the teat canal forces the canals to be opened widely which allows entrance of microbes. The observation of blind quarters in this study might be an indication of a serious mastitis problem on the farms and of the absence of culling that should have served to remove a source of mammary pathogens for the cows.
The association between mastitis and age of cows was significant. All of the isolated MRSA were from old age category. Old cows in this study were more susceptible to mastitis infection than young and adult cows. The increasing occurrence of mastitis with increasing age was in agreement with the findings by Kerro and Tareke (2003) who found that, the risk of clinical and subclinical mastitis increase significantly with the advancing age of the cow. This might be due to the increased opportunity of infection with time and the prolonged duration of infection, especially in a herd without mastitis control program (
Radostits et al., 2007).
In this study breed is not significantly influenced on the occurrence of mastitis. In contrast to this study breed has significant influence on the occurrence of mastitis, Almaw et al. (2008) in Gondar town and its surroundings, Sori et al. (2005) in and around Sebeta. Mastitis occurrence among breeds might reflect the differences in management rather than a true genetic difference (
Radostits et al., 2007). The occurrence of mastitis has significant association with parity of the animal. The increased occurrence of mastitis with parity in the current study is in agreement with the previous reports of Mekibib et al. (2010) in Holota town and Haftu et al. (2012) in northern Ethiopia. The association might be due to the increased opportunity of infection with time and the prolonged duration of infection, especially in a herd without mastitis control program (
Radostits et al., 2007).
The observed occurrence of mastitis during early lactation as compared to late lactation stages was insignificant. This study was in contrast to previous reports of Mulugeta and Wassie (2013); Biffa et al. (2005) and Tamirat (2007) showed that higher infection in cows is occurred at early stage of lactation followed by late and medium stages of lactation.
Radostits et al. (2007) suggested that, the mammary gland is more susceptible to new infection during the early and late dry period, which may be due to the absence of udder washing and teat dipping, which in turn may have increased the presence of potential pathogens on the skin of the teat. Moreover, during a dry period due to low dry cow therapy, the pathogens can easily penetrate into the teat canal and multiply; this can be carried over into the post parturient period and ultimately develop into mastitis.
The current study shows the occurrence of mastitis is insignificantly associated with previously treated animals. In contrast to Tacconelli et al. (2008), a causal relationship between the use of antimicrobial drugs and MRSA has been demonstrated in LA-MRSA and often co-resistant to several other antimicrobial agents. The opposite finding of this research may be due to the absence of proper udder washing and teat dipping, increased presence of potential pathogens on the skin of the teat which can easily penetrate into the teat canal and multiply and antibiotic resistance ability of most pathogens.
In this study floor system had a significant influence on the occurrence of mastitis. In agreement with Abera et al. (2013) in Adama town and
Demelash et al. (2005) in southern Ethiopia, the finding of a high prevalence of mastitis in farms with muddy (soil) floors when compared with concrete (cement) floor types. This is due to association with poor sanitation and cows which were maintained in dirty and muddy common barns with bedding materials that favor the proliferation and transmission of mastitis pathogens.
The prevalence of mastitis was significantly associated with milking hygienic practice. Cows at farms with poor milking hygiene standard are severely affected than those with good milking hygiene practices (Mulugeta and Wassie (2013; Lakew et al., 2009; Sori et al., 2005). This might be due to absence of udder washing, milking of cows with common millers using a common udder cloth and milking of subclinical mastitic cows first, which could be vectors of spread especially for contagious mastitis (Radostitis et al., 1994).
In the present study microbiological examination of milk from lactating dairy cows shows the presence of S. aureus. S. aureus isolated at a rate of 73.3 and 42% in clinical and subclinical mastitis infections, respectively is in line with that of Bedada and Hiko (2011), Workineh et al. (2002) and Kerro and Tareke (2003) who reported 39.1%, S. aureus isolates at Assela, 39.2% at Addis Ababa and 40.3% at Southern Ethiopia respectively. This is higher result when it is compared with the findings of Abebe et al. (2013) who reported 15.5% at Addis Ababa. The possible explanation for the variation might be that S. aureus is a contagious pathogen transmitted from one cow to another or individual by contact with animals during unhygienic milking procedures (Rowe, 1999). The isolates of S. aureus in clinical mastitis are significantly higher than subclinical mastitis. This result is due to S. aureus has adapted to survive in the udder and establish chronic and subclinical infections. From there it shed into the milk, which serves as a source of infection for healthy cows during the milking process (Radostitis et al., 1994).
The present study showed the resistance of
S. aureus to penicillin G, amoxicillin, tetracycline, and cefoxitin. This is in accordance with the findings of Abebe et al. (2013) who reported resistance of
S. aureus to penicillin (94%), tetracycline (73.8%) around Addis Ababa. The current finding is in line with the finding of Abera et al. (2013) around Adama who recorded 94.4% to penicillin. The present study has demonstrated the existence of alarming level of resistance of
S. aureus to commonly used antimicrobials (pencillin G and tetracycline including amoxicillin) in the study farms. The results were in accordance with reports from earlier studies in other countries (
Jakee et al., 2008; Edward et al., 2002; Gentilini et al., 2002) suggesting a possible development of resistance from prolonged and indiscriminate usage of some antimicrobials.
Resistance to Penicillin G is a great concern; since this antibiotic represents the main antibiotic groups recommended for Staphylococcal mastitic infection. The regular use of antibiotics for the treatment of cows may result in the spread of resistant strains. Antibiotic resistance is carried on plasmids and transposons that can pass from one Staphylococcal species to another (Hulya et al., 2006). The resistance of S. aureus to penicillin and cefoxitin may be attributed to the production of beta lactamase enzyme that inactivates penicillin and closely related antibiotics. Around 50% of mastitis causing S. aureus strains produces beta-lactamase (Green and Bradely, 2004). Similar suggestion was given by Jaims et al. (2002) that the development of antimicrobial resistance is nearly always as a result of repeated therapeutic and/or indiscriminate use of them.
The resistance of S. aureus isolates to beta-lactams such as penicillin G, cefoxitin, amoxaclin and tetracycline was evident. High percentage of S. aureus was resistant to penicillin G, amoxacilin, tetracycline, cefoxitin, and to some extent streptomycin. In the current study area cefoxitin resistant isolates was isolated from milk and is in line with similar findings. Of All cefoxitin resistant S. aureus were also resistant to penicillin G (100%). Out of the 25 cefoxitin resistant S. aureus isolates, 100% were also resistant to amoxicillin. This is an indicator of MRSA (Daka et al., 2012). In this study, gentamycin is the drug of choice for treating MRSA followed by erythromycin.
CONCLUSION AND RECOMMENDATIONS
The overall prevalence of mastitis in the study area is 56% (10% clinical and 46% subclinical) in cows and 33.7% at quarter level and different risk factors are found to be associated with mastitis. Among these different risk factors, age of the animal, milking hygiene, parity and floor type were critical for the occurrence of mastitis. Mastitis caused by S. aureus is one of the major problems of dairy cows in milk production in the study area. It was found that the majority of the tested isolates were resistant to the various antimicrobial agents especially penicillin G, cefoxitin, tetracycline, amoxicillin and to some extent streptomycin. It was also observed that most proportions of the isolates were susceptible to gentamycin followed by erythromycin. In this study all cefoxitin resistant S. aureus were also resistant to penicillin G. The possible explanations for the high record of most drug resistant S. aureus in dairy farms may be due to the unrestrictive and uncontrolled use of antibiotics in dairy farms.
Based on the above concluding remarks the following recommendation are forwarded:
1) Mechanisms to control the risk factors associated to the disease should be implemented;
2) There should be regular antimicrobial sensitivity testing to select effective antibiotics;
3) Veterinarians should reduce repeated use of drugs to minimize drug resistance development;
4) Awareness should be created among veterinarians, dairy farm owners and dairy workers on the effect of MRSA;
5) S. aureus mastitis control strategy should be implemented and promoted in the study area;
6) Use of gentamycin is better to treat mastitic infected cows in the study area;
7) Application of molecular techniques is mandatory to identify drug resistant gene of MRSA.
The authors have not declared any conflict of interest.
It is our pleasure to thank Addis Ababa university- Thematic research project "Pneumonia, diarrhea and mastitis in food animals: Host immunity, diversity of pathogens and antibiotic resistance, impact on food safety and trade, and intervention strategies" for the provision of all the necessary laboratory ingredients and financial support.
REFERENCES
|
Abebe M, Daniel A, Yimtubezinash W, Genene T (2013). Identification and antimicrobial susceptibility of Staphylococcus aureus isolated from milk samples of dairy cows and nasal swabs of farm workers in selected dairy farms around Addis Ababa, Ethiopia. Afr. J. Microbiol. Res. 7(27):3501-3510.
|
|
|
|
Abera M, Demie B, Aragaw K, Regassa F, Regassa A (2013). Isolation and identification of Staphylococcus aureus from bovine mastitic milk and their drug resistance patterns in Adama town, Ethiopia. J. Vet. Med. Animal Health 2(3):29-34.
|
|
|
|
|
Almaw G, Zerihun A, Asfaw Y (2008). Bovine mastitis and its association with selected risk factors in smallholder dairy farms in and around Bahir Dar, Ethiopia. Trop. Anim Health Prod. 40(6):427-432.
Crossref
|
|
|
|
|
Bedada A, Hiko A (2011). Mastitis and antimicrobial susceptibility test at Asella, Oromia Regional state, Ethiopia. J. Microbiol. Antimicrob. 3(9):228-232.
|
|
|
|
|
Biffa D, Debela E, Beyene F (2005). Prevalence and risk factors of mastitis in lactating dairy cows in southern Ethiopia. Int. J. Appl. Res. Vet. Med. 3(3):189-198.
|
|
|
|
|
Bitew M, Tafere A, Tolosa T (2010). Study on bovine mastitis in dairy farms of Bahir Dar town and its environs. J. Anim. Vet. Adv. 9(23):2912-2917.
Crossref
|
|
|
|
|
Clinical and Laboratory Standards Institute (CLSI) (2006). Investigation and control of vancomycin intermediate and resistant Staphylococcus aureus. A guide book for health departments and infection control personnel, Wayne, PA.
|
|
|
|
|
Daka D, Silassie S, Yihdego D (2012). Antibiotic-resistance Staphylococcus aureus isolated from cow's milk in the Hawassa area, South Ethiopia. Ann. Clin. Microbiol. Antimicrob. 11:26.
Crossref
|
|
|
|
|
Edward M, Anna K, Michal K, Henryka L, Krystyna K (2002). Antimicrobial susceptibility of staphylococci isolated from mastitic cows. Bull. Vet. Inst. Pulawy, pp. 289-294.
|
|
|
|
|
European Food Safety Authority (EFSA) (2009). Assessment of the Public Health significance of methicillin resistant Staphylococcus aureus (MRSA) in animals and foods. Scientific Opinion of the Panel on Biological Hazards. Eur. Food Safe. Authority J. 993:1-73.
|
|
|
|
|
Firaol T, Sefinew A, Awoke T (2013). Aerobic Microorganisms Isolated from Mastitic Bovine Milk and Their Antimicrobial Susceptibility Profiles, Ethiopia. American-Eurasian J. Agric. Environ. Sci. 13(7):920-925.
|
|
|
|
|
Gentilini E, Denamiel G, Betancor A, Rebuelto M, Fermepin M, De Torres R (2002). Antimicrobial susceptibility of coagulase-negative Staphylococci isolated from bovine Mastitis in Argentina. J. Dairy Sci. 85(8):1913-1917.
Crossref
|
|
|
|
|
Gordon J, Lowy D (2008). Pathogenesis of methicillin-resistant Staphylococcus aureus infection. Clin. Infec. Dis. 46:350-359.
Crossref
|
|
|
|
|
Green M, Bradely A (2004). Clinical Forum- Staphylococcus aureus mastitis in cattle. Cattle Pract. 9(4):1-9.
|
|
|
|
|
Haftu R, Taddele H, Gugsa G, Kelayou S (2012). Prevalence, bacterial causes, and antimicrobial susceptibility profile of mastitis isolates from cows in large-scale dairy farms of Northern Ethiopia. Trop. Anim. Health Prod. 44:1765-1771.
Crossref
|
|
|
|
|
Huber H, Giezendanner N, Stephan R, Zweifel C (2011). Genotypes, antibiotic resistance profiles and microarray-based characterization of methicillin-resistant Staphylococcus aureus strains isolated from livestock and veterinarians in Switzerland. Zoo. Pub. Heal. 58(5):343-49
Crossref
|
|
|
|
|
Hulya T, Senay E, Dilek O (2006). Antibiotic resistance of staphylococcus aureus and coagulase-negative staphylococci isolated from bovine mastitis. Bull. Vet. Inst. Pulawy 50(1):41-45.
|
|
|
|
|
Jaims E, Montros L, Renata C (2002). Epidemiology of drug resistance; the case of Staphylococcus aureus and Coagulase negative Staphylococci infections. Salud Publica Mex. 44(2):108-112.
Crossref
|
|
|
|
|
Kerro D, Tareke F (2003). Bovine mastitis in selected areas of Southern Ethiopia. Trop. Anim. Health Prod. 35(3):197-205.
Crossref
|
|
|
|
|
Kluytmans J (2010). Methicillin-resistant Staphylococcus aureus in food products: cause for concern or case for complacency? Clin. Microbiol. Infect. 16(1):11–15.
Crossref
|
|
|
|
|
Kwon N, Park K, Jung W, Youn H, Lee Y, Kim S, Bae W, Lim J, Kim JY, Kim JM, Hong S, Park Y (2006). Characteristics of methicillin resistant Staphylococcus aureus isolated from chicken meat and hospitalized dogs in Korea and their epidemiological relatedness. Vet. Microbiol. 117(2-4):304-312.
Crossref
|
|
|
|
|
Lakew M, Tolesa T, Tigrie W (2009). Prevalence and major bacterial causes of bovine mastitis in Asella, South Eastern Ethiopia. Trop. Anim. Health. Prod. 41(7):1525-1530.
Crossref
|
|
|
|
|
Mekibib B, Furgasa M, Abunna F, Megersa B, Regasa A (2010). Bovine mastitis: prevalence, risk factors and major pathogens in dairy farm of Holeta town, central Ethiopia. Vet. World 3(9):397-403.
Crossref
|
|
|
|
|
Mulugeta Y, Wassie M (2013). Prevalence, risk factors and major bacterial causes of bovine mastitis in and around Wolaita Sodo, Southern Ethiopia. Afr. J. Microbiol. Res. 7(48):5400-5405.
|
|
|
|
|
Normanno G, Corrente M, La Salandra G, Dambrosio A, Quaglia C, Parisi A, Greco G, Bellacicco A, Virgilio S, Celano G (2007). Methicillin-resistant Staphylococcus aureus (MRSA) in foods of animal origin product in Italy. Int. J. Food Microbiol. 117(2):219-222.
Crossref
|
|
|
|
|
Otter A, French L (2010). Molecular epidemiology of community associated meticillin-resistant Staphylococcus aureus in Europe. Lancet Infect. Dis. 10(4):227-239.
Crossref
|
|
|
|
|
Quinn J, Carter E, Markey B, Carter R (1994). Clinical Veterinary Microbiology, Wilfe Publishing, London, pp. 95-101.
|
|
|
|
|
Quinn J, Carter E, Markey B, Carter R (1999). Mastitis. In: Clinical Veterinary Microbiology, Mosby International Limited, London, pp. 327-344.
|
|
|
|
|
Radostitis M, Blood D, Gay C (1994). Veterinary Medicine: A text book of the diseases of cattle, sheep, pigs, goats and horses. 8 th edition, Bailliere Tindall: London, pp. 563-613.
|
|
|
|
|
Rowe J (1999). Milk quality and Mastitis. Small ruminant for mixed practitioner. Western Veterinary Conference, Lasvagas, pp. 152-156.
|
|
|
|
|
Sori H, Zerihun A, Abdicho S (2005). Dairy cattle mastitis in and around Sebeta, Ethiopia. Int. J. Appl. Res. Vet. Med. 3(4):332-338.
|
|
|
|
|
Tacconelli E De Angelis G, Cataldo M, Pozzi E, Cauda R (2008). Does antibiotic exposure increase the risk of methicillin-resistant Staphylococcus aureus (MRSA) isolation? A systematic review and meta-analysis. J. Antimicrob. Chemother. 61(1):26-38.
Crossref
|
|
|
|
|
Tamirat T (2007). Comparison of clinical trials of bovine mastitis with the use of honey. MSc thesis, Addis Ababa University, Ethiopia, pp. 14-30.
|
|
|
|
|
Ünal N, Askar Åž, Macun H, Sakarya F, Altun B, Yıldırım M (2012). Panton-Valentine leukocidin and some exotoxins of Staphylococcus aureus and antimicrobial susceptibility profiles of staphylococci isolated from milks of small ruminants. Trop. Anim. Health Prod. 44:573-579.
Crossref
|
|
|
|
|
Radostits M, Gay C, Hinchcliff K, Constable D (2007). Veterinary medicine: A text book of disease of cattle, horse, sheep, pig and goats.10th edition, London, pp. 673-762.
|
|
|
|
|
Workineh S, Bayleygn M, Mekonnen H, Potgieter L (2002). Prevalence and aetiology of mastitis in cows from two major Ethiopian dairies. Trop. Anim. Health Prod. 34:19-25.
Crossref
|
|