ABSTRACT
The aim of this study was to evaluate the type of microbiota present in root canals with pulp necrosis, with and without periapical lesion. Nineteen patients were selected for the study and 30 root canals were analysed in unirradicular and/or multi-radicular permanent teeth, asymptomatic, with pulp necrosis, with or without periapical lesion, and no communication between root canal and oral cavity. Absorbent paper cones were introduced in the canal for 20 s. Then, the contaminated paper cones were inoculated in a Brain Heart Infusion Agar-BHI culture medium and incubated in an oven for 48 h at 37°C. The data were analysed by non-parametric statistical method of the Cochran-test. The results revealed that there was no statistically significant difference between the amount of Gram-positive and negative bacteria in endodontic infections, indicating that the microbiota of these endodontic infections is mixed.
Key words: Microbiota, lipopolysaccharide, endodontics, root canals, pulp necrosis.
Treatment of infections in the root canal space of the teeth is a challenge in endodontics. One of the aims of endodontic treatment is the complete elimination of existing microorganisms, hence, the importance of chemical means of disinfection has aroused to accomplish complete sterilization of canal and to improve the treatment prognosis (Reddy et al., 2017). For this reason, the knowledge on endodontic microbiota, its organization and its distribution within the root canal system, is of vital importance in understanding the infectious process and the establishment of therapeutic strategies that aims to eradicate the infection. Since some symptoms are associated with the presence of specific bacteria, if they are not completely eliminated from the root canals, they may perpetuate a pathological condition that causes treatment failure (Pajari et al., 1993; Lopes and Siqueira-Júnior, 2004).
However, microorganisms that cause endodontic infections are generally of low virulence. Their pathogenesis and survival are influenced by the release of lipopolysaccharide (LPS), bacterial toxins and synthesis of enzymes (Nair, 2004). The presence of bacteria within root canals contributes to the beginning of pulp necrosis and the possible involvement of the periapical region of the teeth (Dubei, 2016). These lesions are associated with a mixed microbiota, which consist of aerobic, anaerobic, Gram positive and negative microorganisms (Kakehashi et al., 1965; Cheung et al., 2001).
The aim of this study was to analyse the type of microbiota present inside the necrotic root canals with or without periapical lesions. The microbiota was identified through the coloured Gram method and its morphological classification.
Sample selection
Nineteen patients were selected, regardless of age and sex. The research was performed at the College of Odontology, of the Federal University of Alagoas (UFAL), after the project approval by the Ethics Committee of Research of the same unit (No. 007418/2009-35). For this study, 30 root canals were analysed, taking into account, the sampling error, approximately 0.05%, with permanent teeth single-rooted or multi-rooted, asymptomatic, with pulp necrosis, with or without periapical lesion, and not presenting communication between root canal and the oral cavity. Only one exclusion criteria that the patients did not use any antibiotics for a minimum period of three months (Weiger et al., 1995).
Clinical procedures
After signing the consent term, a clinical file of the patient was filled up and an evaluation of the patient’s general health was performed. For diagnosis, radiographs were examined and pulp sensitivity to cold was tested.
Prior to intervention, the patient made a mouthwash with 10 ml of 0.12% chlorhexidine (Periogard ®, Colgate - Palmolive Industry and Trade LTDA, São Paulo, Brazil). After that, the carious tissue was removed. After isolating the tooth, the working station (tooth, clamp, rubber dam and arch) was cleansed with NaOCl 2.5% (Brilux, Pernambuco, Brazil), followed by a surgery to access the pulp chamber.
Collection of samples
Two to four sterile absorbent paper cones (Dentsply Industry and Commerce Ltda., Petrópolis, Brazil) were introduced into the canal for 20 s. In cases where the canal was dry, it was moistened with sterile distilled water (Laborclin Laboratory Products Ltda., Paraná, Brazil) to ensure a viable sample. Then, the contaminated paper cones were inoculated in a Brain Heart Infusion (BHI) Agar (Interlab Scientific Products Distributor S / A, São Paulo, Brazil) culture medium and incubated in an oven for 48 h at 37°C.
Laboratory analysis
The analyses were done in the Laboratory of Biochemistry and Physiology in the Department of Antibiotics, at Federal University of Pernambuco (UFPE).
The Gram methodology was used according to the methodology of Winn et al. (2005). A thin scrub was prepared and the material was left to air dry. After this step, the material was passed 3 to 4 times by a flame of a Bunsen burner to be fixed on a lamina, so that it will not be detached during the colouring process. Then, the material was covered with ethanol for 4 min. Next, it was put on a holder and its surface was covered with crystal violet solution for 1 min, washed with distilled water and covered with Gram iodide solution for 1 min. Subsequently, it was washed again with distilled water. Then, acetone was dropped on the material until the purple colour disappeared, within 10 s, washed with distilled water. The scrub surface was covered with safranin for 1 min and washed with distilled water. After this step, the material was placed in an upright position on a holder and left to dry. The stained lamina was examined through an optic microscope with objective lens, 100x, using a drop of immersion oil. Gram-positive bacteria were coloured in dark blue and Gram-negative, in pink-red. Optical microscope (Nikon E200) observation of the lamina for morphological classification of microorganisms was performed with an objective lens, 100x.
Statistical analysis
The data were analyzed by non-parametric statistical method of Cochran-test, using the software Assistat, Beta version 7.0; the data do not follow a normal distribution (p<0.05).
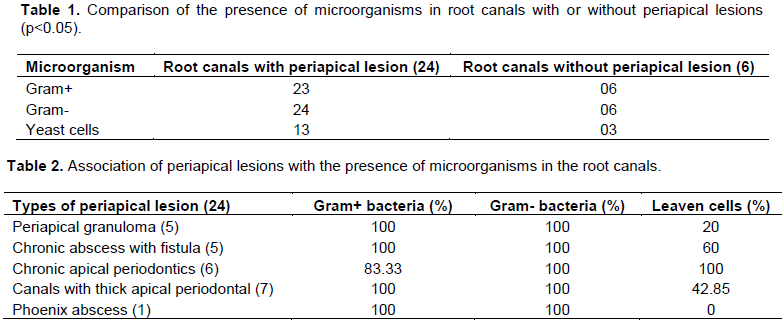
Thirty root canals with pulp necrosis were analysed, from which 24 presented periapical lesions. All the root canals with pulp necrosis and periapical lesions examined showed Gram-negative bacilli (100%), whereas in the canals of 23 teeth, it was possible to find cocci and Gram-positive bacilli (95.8%). In 13 teeth canals with pulp necrosis and periapical lesions, yeast infections were observed (54.1%) as described in Table 1. In the 6 cases evaluated with pulp necrosis without periapical lesions, there was a balance in the presence of Gram-positive and negative microorganisms (100%). However, yeast cells were found in three of these six root canals (50%, p <0.05) evaluated (Table 1).
For 24 teeth studied with periapical lesions, five cases of granuloma were diagnosed through radiographs and all of them presented Gram-negative and positive microorganisms (100%). Furthermore, one canal also had yeast cells (20%). In the five cases of chronic abscess with fistula, A balance of Gram-positive and negative bacteria was also observed in all the root canals (100%) and in 3 out of 5 canals, leavens cells were found (60%). Studying the six cases of chronic apical periodontitis, all of them presented Gram-positive microorganisms and yeast cells (100%), and in 5 out of the 6 canals, Gram-negative bacteria (83.33%) were found. Moreover, seven canals were diagnosed with thick apical periodontal, all these canals presented Gram-positive and negative microorganisms (100%), and in 3 canals, yeast cells (42.85%) were found. In this research, one canal with phoenix abscess was diagnosed. In this canal, Gram-negative and positive microorganisms (100%) were found. However, yeast cells were not present (Table 2).
Due to its complexity, the endodontic microbiota has been extensively studied through macroscopic and microscopic observations, and also through the classification of the microorganisms by means of Gram colour and evaluation of the presence of yeasts, aerobic microorganisms, strict anaerobes and facultative bacteria, which supports the claim that endodontic infections are polymicrobial in nature (Jacinto et al., 2003; Pazelli et al., 2003; Pinheiro et al., 2003; Gomes et al., 2004; Vianna et al., 2006). In this research, the presence of a mixed microbiota colonizing the root canals with pulp necrosis was observed presenting aerobic, anaerobic, Gram positive, Gram negative microorganisms, Cocci, Bacillus and yeasts. In previous researches (Jacinto et al., 2003; Gomes et al., 2004; Pourhajibagher et al., 2017), there was a prevalence of Gram-negative microorganisms in the primary endodontic infections, both symptomatic and asymptomatic, which is in line with the data presented here. In the current study, there was no significant difference (p> 0.05) in the presence of Gram-positive and negative microorganisms in the root canals with pulp necrosis, as reported by Nobrega et al. (2016) but in the study of Vianna et al. (2006) and Sakko et al. (2016), the prevalence of Gram-positive microorganisms was found. Other researches showed that the microbiota of untreated teeth is different from that in which failure in endodontic treatment occurred. In the case of retreatment of root canals, there is a prevalence of Gram-positive microorganisms. These findings indicate that the microbiota differs from primary to secondary endodontic infections (Gomes et al., 2004; Silva et al., 2006; Sousa, 2000).
In the analysis of teeth with periapical abscess, Siqueira and Rôças (2013) and Sousa (2000) found a predominance of Gram-positive anaerobic bacteria in most of the root canals observed, although less frequent facultative anaerobic bacteria had also been found, demonstrating the presence of a mixed microbiota in the infected root canals associated with periapical abscesses. In the current research, a predominance of strict anaerobic bacteria was also verified, but there are no major Gram-positive or negative bacteria. However, Saini et al. (2004) observed the predominance of aerobic microorganisms in the abscesses. In another study, Penãrrocha et al. (2007) evaluated microbiologically and histopathologically, 30 cases of periapical lesions. A total of 137 bacterial strains, including Fusobacterium nucleatum (Bacillus, Gram-negative, Anaerobic), Gram-negative anaerobic bacilli, Peptostreptococcus sp. (Cocci, Gram positive, Anaerobic), Streptococcus mitis Anaerobic facultative) and Gram-positive non-spore anaerobic bacilli were, in descending order, the most commonly isolated bacteria from the lesions. Histopathological analysis revealed the prevalence of periapical granuloma. However, in this study, the presence of Gram positive and negative bacteria in granulomatous lesions was observed.
On the other hand, study on the composition of the bacterial flora of infected root canals with apical periodontitis was carried out (Jacinto et al., 2003; Gomes et al., 2004). The presence of a mixed microbiota with the prevalence of Gram-negative anaerobic bacteria was observed, which corroborates the results of the current study study.
Gram-negative bacteria were found in all root canals of the chronic apical periodontitis affected teeth. Gram-positive bacteria was also found in 5 of the 6 evaluated teeth, corroborating with the findings of Foschi et al. (2005) and Vengerfeldt et al. (2014), which demonstrated the presence of Gram-positive coccus E. faecalis, suggesting that these species play critical roles in endodontics pathology.
Pinheiro et al. (2003) and Pourhajibagher et al. (2017) examined teeth with failure in endodontic treatment and in the presence of periapical lesions, and observed Candida albicans. In the present study, the existence of C. albicans was observed in both canals, with and without periapical lesions. Therefore, fungi may play a significant role in the pathogenesis of periapical diseases, as they have the ability to adapt to a variety of environmental conditions, adhere to different surfaces, produce hydrolytic enzymes, form biofilms and modulate hosts immunity (Siqueira et al., 2004). In this way, Gomes et al., (2008) analyzed teeth with pulp necrosis (primary infection) associated with the periapical lesion and reported the presence of C. albicans. Lana et al. (2001) studied channels without periapical lesion and indicated the presence of fungi. These data confirmed the presence of fungi both in channels with necrotic pulp associated with periapical lesion or not.
The result of this research showed a predominance of Gram negative microorganisms, in root canals with periapical lesion in comparison with Gram positive microorganisms and yeast.
In root canals without periapical lesions, there was a balance between the presence of Gram positive and negative microorganism; however, presenting a lower amount of yeast. In all the canals analysed with periapical lesion only in those affected by chronic apical periodontitis, there was a balance regarding the presence of Gram positive, negative and yeast bacteria.
The results obtained demonstrated that the microbiota of the root canals studied is mixed with a predominance of Gram negative bacteria.
The authors have not declared any conflict of interests.
REFERENCES
|
Cheung GS, Ho MW (2001). Microbial flora of root canal-treated teeth associated with asymptomatic periapical radiolucent lesions. Oral Microbiology and Immunology 16(6):332-337.
Crossref
|
|
|
|
Dubei S (2016). Comparative antimicrobial efficacy of herbal alternatives (Emblica officinalis, Psidium guajava), MTAD, and 2.5% sodium hypochlorite against Enterococcus faecalis: An in vitro study. Journal of Oral Biology and Craniofacial Research 6(1): 46-49.
Crossref
|
|
|
|
|
Foschi F, Cavrini F, Montebugnoli L, Stashenko P, Sambri V, Prati C (2005). Detection of bacteria in endodontic samples by polymerase chain reaction assays and association with defined clinical signs in Italian patients. Oral Microbiology and Immunology 20(5):289-295.
Crossref
|
|
|
|
|
Gomes BP1, Pinheiro ET, Gadê-Neto CR, Sousa EL, Ferraz CC, Zaia AA, Teixeira FB, Souza-Filho FJ (2004). Microbiological examination of infected dental root canals. Oral Microbiology and Immunology 19(2):71-76.
Crossref
|
|
|
|
|
Gomes BPFA, Pinheiro ET, Jacinto RC, Zaia AA, Ferraz CCR, Souza-Filho FJ (2008). Microbial analysis of canals of root-filled teeth with periapical lesions using polymerase chain reaction. Journal of Endodontics 34 (5): 537-540.
Crossref
|
|
|
|
|
Jacinto RC, Gomes BP, Ferraz CC, Zaia AA, Filho FJ (2003). Microbiological analysis of infected root canals from symptomatic and asymptomatic teeth with periapical periodontitis and the antimicrobial susceptibility of some isolated anaerobic bacteria. Oral Microbiology and Immunology 18(5):285–92.
Crossref
|
|
|
|
|
Kakehashi S, Stanley HR, Fitzgerald RJ (1965). The effects of surgical exposure of dental pulps in germ free and conventional laboratory rats. Oral Surgery, Oral Medicine, Oral Pathology, and Oral Radiology 20(3): 340-349.
Crossref
|
|
|
|
|
Lana MA, Ribeiro-Sobrinho AP, Ste hling R, Garcia GD, Silva BK, Hamdan JS, Nicoli JR, Carvalho MA, Farias LM (2001). Microorganisms isolated from root canals presenting necrotic pulp and their drug susceptibility in vitro. Oral Microbiology and Immunology 16(2):100-105.
Crossref
|
|
|
|
|
Lopes HP, Siqueira-Júnior JF (2004). Endodontics, Biology and Technique, 2th edition. pp. 25-27.
|
|
|
|
|
Nair PNR (2004). Pathogenesis of apical periodontitis and the causes of endodontic failures. Critical Reviews in Oral Biology and Medicine 15(6):348-381.
Crossref
|
|
|
|
|
Nóbrega LM, Montagner F, Ribeiro AC, Mayer MA, Gomes BP (2016). Bacterial diversity of symptomatic primary endodontic infection by clonal analysis. Brazilian Oral Research 30(1):e103:1-9.
|
|
|
|
|
Pajari U, Ahola R, Bäckman T, Hietala EL, Tjäderhane L, Larmas M (1993). Evaluation of Gram's Method of staining for prognosis of root canal treatment in nonvital dental pulps. Oral Surgery, Oral Medicine, Oral Pathology, and Oral Radiology 76(1): 91-96.
Crossref
|
|
|
|
|
Pazelli C, Freitas AC, Ito IY, Souza-Gugelmin MCM, Medeiros AS, Nelson-Filho P (2003). Prevalence de microorganisms in root canals of human deciduous teeth with necrotic pulp and chronic periapical lesions. Pesquisa Odontologica Brasileira 174(4): 367-371.
Crossref
|
|
|
|
|
Penãrrocha M, Marti E, Garcia B, Gay C (2007). Relationship of periapical lesion radiologic size, apical resection, and retrograde filling with the prognosis of periapical surgery. Journal of Oral and Maxillofacial Surgery 65(8):1526-1529.
Crossref
|
|
|
|
|
Pinheiro ET, Gomes BPFA, Ferraz CCR, Sousa ELR, Teixeira FB, Souza Filho FJ (2003). Microorganisms from canals of root-filled teeth with periapical lesions. International Endodontic Journal 36(1):1-11.
Crossref
|
|
|
|
|
Pourhajibagher M, Ghorbanzadeh R, Bahador A (2017). Culture-dependent approaches to explore the prevalence of root canal pathogens from endodontic infections. Brazilian Oral Research 31:1-7
Crossref
|
|
|
|
|
Reddy GA, Sridevi E, Sai Sankar AJ, Pranitha K, Pratap Gowd MJ, Vinay C (2017). Endodontic treatment of chronically infected primary teeth using triple antibiotic paste: An in vivo study. Journal of Conservative Dentistry 20(6):405-410.
Crossref
|
|
|
|
|
Saini S, Naveen G, Aparna L, Griwan MS (2004). Surgical infections: a microbiological study. Brazilian Journal of Infectious Diseases 8(2):118-125.
Crossref
|
|
|
|
|
Sakko M, Tjäderhane L, Rautemaa-Richardson R (2016). Microbiology of root canal infections. Primary Dental Journal 5(2):84-89.
|
|
|
|
|
Silva LAB, Nelson-Filho P, Faria G, Souza-Gugelmin MCM, Ito IY (2006). Bacterial profile in primary teeth with necrotic pulp and periapical lesions. Brazilian Dental Journal 172(2):144-148.
Crossref
|
|
|
|
|
Siqueira JF, Rôças IN (2013). Microbiology and treatment of acute apical abscesses. Clinical Microbiology Reviews, 26(2):255-273.
Crossref
|
|
|
|
|
Siqueira JF, Sen BH (2004). Fungi in endodontic infections. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology, 97(5):632-641.
Crossref
|
|
|
|
|
Vengerfeldt V, Špilka K, Saag M, Preem JK, Oopkaup K, Truu J, Mändar R (2014). Highly diverse microbiota in dental root canals in cases of apical periodontitis (data of illumina sequencing). Journal of Endodontics 40(11):1778-1783.
Crossref
|
|
|
|
|
Vianna ME, Conrads G, Gomes BP, Horz HP (2006). Identification and quantification of archaea involved in primary endodontic infections. Journal of Clinical Microbiology 44(4): 1274–1282.
Crossref
|
|
|
|
|
Weiger R, Manncke B, Werner H, Löst C (1995). Microbial flora of sinus tracts and root canals of non-vital teeth. Endodontics and Dental Traumatology 11(1):15-19.
Crossref
|
|