ABSTRACT
Comparison of heavy metal levels in Oreochromis niloticus and Chrysichthys nigrodigitatus caught in Ogun River (Nigeria) was carried out to reveal the potential health risks associated with fish consumption from the river. Concentrations of copper, lead and cadmium were determined in the gills, eyes, bone, flesh, liver and stomach of the fish species. Ten fish of each species were obtained commercially and analyzed for the metals using Atomic Absorption Spectrophotometer. Data obtained were subjected to one-way analysis of variance (ANOVA) at 5% significant level. The highest concentrations of Cu were observed in the gills of O. niloticus and the eyes followed by liver of C. nigrodigitatus, while the lowest concentrations were detected in the eyes of O. niloticus and bone of C. nigrodigitatus. Lead concentrations were higher in the liver of O. niloticus and gills of C. nigrodigitatus, while the lowest concentrations were detected in stomach of O. niloticus and C. nigrodigitatus respectively. Also, in proximate analysis, composition of dry matter, crude protein, crude fat, ash and carbohydrate contents recorded high in C. nigrodigitatus and O. niloticus and significantly differs. All the metals studied exceeded standard permissible limit for human consumption. Therefore, monitoring of the river is highly recommended, while adequate information, education and training should be strengthened to control further pollution.
Key words: Freshwater fish, Africa, contamination, Nile tilapia, catfish.
The contamination of freshwater bodies with a wide range of pollutants has become a great concern over the last few decades. Heavy metals are natural trace elements of the aquatic environment, but their levels have increased due to industrial, mining, domestic and agricultural activities (Kalay and Canli, 2000). Owing to their toxicity and cumulative effect, discharge of heavy metals into the aquatic environments can alter the diversity of aquatic species and ecosystems (Berga, 2006). Aquatic organisms (such as fish and shell fish) accumulate heavy metals concentrations many times higher than what is present in water or sediment (Olaifa et al., 2004; George et al., 2013). At low concentration, some heavy metals such as iron, manganese, copper, cobalt and zinc, are essential in human body for enzymatic activities and many other biological processes which also become toxic at high concentrations (Bryan, 1976). Several researches carried out on fish have shown that, heavy metals may have toxic effects by changing their physiological activities and biochemical composition in tissue (Larsson et al., 1985; Nemesok and Huphes, 1988; George et al., 2013).
In Nigeria, all water bodies serve as sources of exploitation for industrial, domestic, agricultural and urban uses. This has made many water bodies to be greatly affected by these intensive anthropogenic practices. Whereas, it is important to monitor heavy metals in aquatic environments (water, sediment and biota) as their consequences of their pollution can be hazardous to man through his food (Khaled, 2004). Oreochromis niloticus and Chrysichthys nigrodigitatus are two important species for aquaculture in Nigeria.
Therefore, the aim of this study investigated the levels of heavy metals in two commercially important fish species (O. niloticus and C. nigrodigitatus) obtained from Ogun River.
Study area
Ogun River (8.68333°N and 3.46667°E) takes its source from Igaran hills near Shaki in Oyo State at an elevation of about 530 m above the sea level and flows directly southwards through Ogun State to Lagos State, over a distance of 480 Km before it discharge into the Lagos Lagoon (Ayoade et al., 2004) (Figure 1).
Metal analysis
Ten fish of each species, C. nigrodigitatus (22 ± 2 cm total length and 27 ± 2 g) and O. niloticus (19 ± 2 cm total length and 27 ± 2 g) were bought from the local fishermen at fish landing in the study area in June 2014. All the fish samples were carried to the laboratory, dissected to remove bone, stomach, gill, eye, liver and flesh and oven dried at 150°C. Each dried tissue/organ was homogenized using Association of Official Analytical Chemist (2005) standard procedure.
Two grams of each dry sample were weighed, transferred into polyethylene tubes; 10 ml of freshly prepared nitric acid (HNO3) were added to the sample and left overnight at room temperature. Then, the samples were digested and put in a water bath set at boiling water temperature (100°C) with the contents boiled for about
2 h until all the samples dissolved. The digests were allowed to cool, filtered, transferred to 25 ml volumetric flasks and made up to mark with 1% nitric acid (AOAC, 2005).
The digests were kept in plastic bottles and the heavy metal concentrations were determined using an Atomic Absorption Spectrophotometer (Bulk Scientific VGB 211 Model). Working calibration standards for copper, lead and cadmium were prepared by serial dilutions of concentrated stock solutions in accordance to manufacturer’s specification and instruction (Merck, Germany). The actual concentrations of each metal were calculated using the formula (Mansour and Sidky, 2002):
Actual concentration of metal in sample = ppmR× dilution factor
Where: ppmR= AAS Reading of digest and Dilution Factor = Volume of digest used ⁄ Weight of sample digested.
Proximate analysis
Proximate analysis of the experimental fish was carried out using the standard procedures of AOAC (2005). Crude protein was quantified by standard micro-kjehdahl nitrogen method using a kjehdahl digestion apparatus and behr 1 steam distillation apparatus. Crude fat was determined by subjecting the samples to continuous extraction with soxhlet apparatus, which is quantified by expressing the difference in weight as a percentage of the original sample weight. The Ash content in each sample was determined as the weight of the residue after 5 g of the sample has been ashed at 550°C in a muffle furnace overnight. Moisture content was determined by drying the samples to a constant weight at 100 to 102°C for 16 h in a draft air oven, the loss in weight was reported as moisture. Crude fibre was determined using 2 g from each sample into fibre flask and 100 ml of NH2SO4 was added.
Statistical analysis
The data were subjected to statistical analysis using statistical package for social sciences (SPSS). A one - way analysis of variance (ANOVA) was carried out, followed by Scheffe post hoc comparisons for the source of statistically significant differences in accordance to Canli and Atli (2003) at P < 0.05.
Heavy metals
Cadmium concentrations in all the tissues and organs of O. niloticus and C. nigrodigitatus were not significantly different (P > 0.05) to each other (Table 1). The highest concentrations of Cd were detected in the stomachs (75.00 ± 14.43 mg/kg and 83.33 ± 28.48 mg/kg) of O. niloticus and of C. nigrodigitatus respectively, while the lowest concentrations were present in the liver (22.00 ± 6.93 mg/kg) of O. niloticus and gills (56.67 ± 17.64 mg/kg) of C. nigrodigitatus (Table 1).
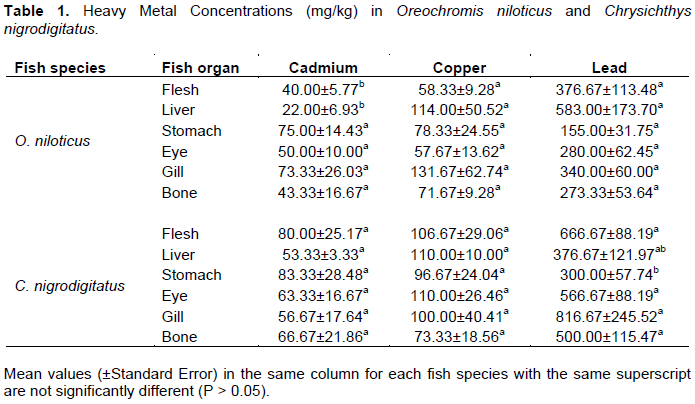
The concentrations of Cd were significantly higher (P < 0.05) in the stomach (75.00 ± 14.43 mg/kg) than that observed in the gills (73.33 ± 26.03 mg/kg); eyes (50.00 ± 10.00 mg/kg); bones (43.33 ± 16.67 mg/kg); flesh (40.00 ± 5.77 mg/kg); liver (22.00 ± 6.93 mg/kg) for O. niloticus and significantly higher in the stomach (83.33 ± 28.48 mg/kg); flesh (80.00 ± 25.17 mg/kg); bone (66.67 ± 21.86 mg/kg); eyes (63.33 ± 16.67 mg/kg); gills (56.67 ± 17.64 mg/kg); liver (53.33 ± 3.33 mg/kg) for C. nigrodigitatus (Table 1).
On the other hand, the copper (Cu) was significantly higher in the gills (131.67 ± 62.74 mg/kg) than that observed in the liver (114.00 ± 50.52 mg/kg); stomach (78.33 ± 24.55 mg/kg); bone (71.67 ± 9.28 mg/kg); flesh (58.33 ± 9.28 mg/kg); eyes (57.67 ± 13.62 mg/kg) for O. niloticus and higher in the eyes (110.00 ± 26.46 mg/kg); liver (110.00 ± 10.00 mg/kg); flesh (106.67 ± 29.06 mg/kg); gills (100.00 ± 40.41 mg/kg); stomach (96.67 ± 24.04 mg/kg); bone (73.33 ± 18.56 mg/kg) for C. nigrodigitatus (Table 1). The highest concentrations of Cu were observed in the gills (131.67 ± 62.74 mg/kg) of O. niloticus and the eyes (110.00 ± 26.46 mg/kg) of C. nigrodigitatus, while the lowest concentrations were detected in the eyes (57.67 ± 13.62 mg/kg) of O. niloticus and bone (73.33 ± 18.56 mg/kg) of C. nigrodigitatus (Table 1).
The concentrations of Pb were higher in the liver (583.00 ± 173.70 mg/kg) than that found in other organs: flesh (376.67 ± 113.48 mg/kg); gills (340.00 ± 60.00 mg/kg); eyes (280.00 ± 62.45 mg/kg); bone (273.33 ± 53.64 mg/kg); stomach (155.00 ± 31.75 mg/kg) for O. niloticus and gills (816.67 ± 245.52 mg/kg) flesh (666.67 ± 88.19 mg/kg); eyes (566.67 ± 88.19 mg/kg) bone (500.00 ± 115.47 mg/kg); liver (376.67 ± 121.97 mg/kg); stomach (300.00 ± 57.74 mg/kg) for C. nigrodigitatus (Table 1).
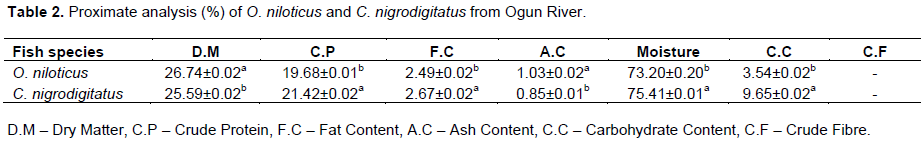
Proximate analysis
Percentages of dry matter, crude protein, fat content, moisture, carbohydrate content and ash content recorded in O. niloticus and C. nigrodigitatus were significantly different (P < 0.05) to each other. The crude fibre content in both fish species was undetected throughout the sampling period (Table 2). The ash contents (1.03 ± 0.02% and 0.85 ± 0.01%) were found to be lowest and the moisture contents (73.20 ± 0.20% and 75.41 ± 0.01%) were highest in both O. niloticus and C. nigrodigitatus respectively. The percentage of proximate composition in both O. niloticus and C. nigrodigitatus was in descending order of ash content < fat content < carbohydrate content < crude protein < dry matter < Moisture concurrently (Table 2).
Cadmium is a non-essential trace metal that is potentially toxic to most fish and wildlife particularly freshwater organism (Fafioye and Ogunsanwo, 2006; George et al., 2013). The high concentrations of Cd above 0.1 mg/kg threshold were considered to be harmful to fish and other fish predators (FAO, 1983). The concentrations of Cd in flesh and bone of O. niloticus and C. nigrodigitatus were lower than what was found in previous findings (El-Nemr, 2003; Mwashot, 2003; Van Aardt and Erdman, 2004; Khaled, 2004).
Cadmium level in gills was higher than other tissues and organs in O. niloticus and C. nigrodigitatus compared to the value of liver which was in line with some previous researches (Canli and Kalay, 1998; Khaled, 2004; Fafioye and Ogunsanwo, 2007). The effects of acute Cd concentration are high blood pressure, kidney damage, destruction of testicular tissue as well as destruction of red blood cells (Gupta and Mathur, 1983). Sources of Cd in human bodies were through food consumption. Severe toxic symptoms resulting from Cd ingestion are reported between 19 to 26 mg/kg, while fatal ingestion produces shock and acute renal failure which occur from ingestion exceeding 350 mg/kg (Sivaperumal et al., 2007). Copper is an essential part of several enzymes that is necessary for haemoglobin synthesis, but can cause harm at high concentrations and lead to serious health risk (Telisman et al., 2000). Concentrations of Cu in both the fish species of Ogun River were far above permissible limits for Cu in fish (30.0 mg/kg) as proposed for human consumption by FAO (1983). Copper is present in the water surface and ground water due to the extensive use of pesticides sprays containing copper compounds for agricultural purposes. It is an essential element in human metabolism, but can cause anemia, bone disorders and liver damage at excessive levels which depends upon the water hardness and pH (Taha, 2004).
In this study, the level of Pb in O. niloticus and C. nigrodigitatus was higher in the gills than in the bones and eyes corroborating the studies of Metcalfe et al. (1999). The sub-lethal concentrations effects of lead include delayed embryonic development, suppressed reproduction, increased mucous formation, neurological problems and kidney dysfunction (Rompala et al., 2004; Fafioye et al., 2008). Oladimeji and Offem (2009) noticed in O. niloticus and C. nigrodigitatus, that the gills had high cumulative potential of Pb as Lead nitrate. The mean concentrations of Pb found in this study were above the maximum acceptable limit 0.5 mg/ kg for human consumption (FAO, 1983; WHO, 2004). Meanwhile, high level of Pb content in this study might be strongly related to discharge of industrial wastes, oil leakage, grease and antifouling paints which are serious lead pollution sources into the river body. Statistical comparisons revealed that the metals concentration in O. niloticus were not significantly different except in flesh and liver, while no difference was observed in C. nigrodigitatus except in the liver and stomach which differs to each other.
The main site of accumulation in aquatic organisms is the kidney and liver, other notable site are the gills, bone and exoskeleton. Although, fish muscle (flesh) is the most important part used for human consumption, bone and liver may also be consumed to some extent. Target organs such as liver, eye and gill have tendency to accumulate heavy metals in high values as shown in many fish species in different areas, which O. niloticus and C. nigrodigitatus from Ogun River also concord (Abdel-Moniem, et al., 1994; Romeo et al., 1999; Yilmaz, 2003; 2005; George et al., 2013).
Heavy metals concentrations in inner parts of the flesh used in this present study were not in line with Yilmaz (2003) findings which indicated that concentrations of heavy metals were higher in flesh samples than other parts studied. The reason for high metal concentrations in the flesh could be due to the metal complexion with the mucus that is impossible to be removed completely from the tissue before the analysis.
The levels of heavy metals in fish vary in various species and in various aquatic environments (Canli and Atli, 2003; George et al., 2013). The trace elements considered in this study showed some trends of accumulation in the fish which indicates the importance of using fish as a bio-indicator of aquatic pollution. Results of the trace metal analyses showed that Cd, Cu and Pb residues were detected in all the fish species. Different fish organs had varied concentrations of residues; the order of accumulation of trace metals in the organs of the fishes was largely gills followed by eyes and lastly, by bones.
Levels of residues in the studied fish were above the recommended maximum residues limits (MRLs), suggesting that these one are unsafe for human consumption. Highest metal concentrations were found in C. nigrodigitatus which may be related to the differences in ecological needs, swimming behaviors and the metabolic activities among different fish species. Also, the differences in concentration of these metals in the fish species might suggest the food and the feeding habit of the fish species.
The differences in patterns of these contaminants in the liver and flesh might be a reflection in the differences of metabolism of the contaminants, the composition of lipids and/or the degree of blood perfusion in the various tissues as documented by Sunday et al. (2013). All the metals studied exceeded standard FAO (1983) and WHO (2004) permissible limit for human consumption which might be due to the increase in some anthropogenic activities around the river water.
Generally, fish are low in fat, but an excellence source of protein (Abdullahi, 2001). The ash content was low in Ogun River fish species than those reported in Effiong and Mohammed (2008) findings, with lowest value (0.85%) in C. nigrodigitatus and highest (1.03%) in O. niloticus. Percentage of fat content in the proximate composition is a reflection that the fish contains essential fatty acid needed for prevention of cardiovascular disorder.
Concentration of fish fat obtained indicates high level of bio-accumulation of trace metals, meanwhile the fat contents in C. nigrodigitatus (2.49 ± 0.02%) was higher than O. niloticus (2.67 ± 0.02%) which affirm the rate of metal accumulation in the former to be higher than the later (Table 2). However, findings from this study revealed slight differences in the nutritional compositions of the fish from other previous studies. The proximate analysis of these fish values obtained from this study would help the consumer in choosing fish based on their nutritional values. The crude protein content of C. nigrodigitatus is higher than that of O. niloticus.
Every industry should have its own effluents treatment plant, where proper municipal and domestic effluents treatments would be carried out before draining into the river bodies. Strict legislations to control indiscriminate use of pesticides and/or using standard quality pesticides should be enforced. Various conservation strategies should be employed with adequate educational programmes, training and public awareness on trace metal safety and management.
The authors have not declared any conflict of interests.
REFERENCES
|
Abdel-Moniem M, Khaled A, Iskander M (1994). A study on levels of some heavy metals in El-Mex bay, Alexandria, Egypt. In: Proceeding of the fourth conference on Environmental Protection Must, Alexandria, Egypt. Pp. 155-174.
|
|
|
|
Abdullahi SA (2001). Investigation of Nutritional status of Chrysichthys nigrodigitatus and Auchenoglanis occidentals' family Bagridae. J. Aid J. Arid Zone Fish. 1:39-50.
|
|
|
|
|
AOAC (2005). Official methods of analysis. Association of Official Analytical Chemists 15th Edition, Washington D.C. pp. 11-14.
|
|
|
|
|
Ayoade AA, Sowunmi AA, Nwachukwu HI (2004). Gill asymmetry in Labeo ogunensis from Ogun River, South-west Nigeria. Rev. Biol. Trop. 52(1):171-175.
Crossref
|
|
|
|
|
Berga L (2006). Dams and Reservoire, societies and environment in the 21st century (Eds). Proceedings of the international symposium on Dam in the societies of the 21st Century, 22nd International congress on large Dams (ICOLD), Barcelona, Spain. P 34.
|
|
|
|
|
Bryan GW (1976). Some effects of heavy metal tolerance in aquatic organisms. In: Lockwood A.P.M. (Eds) Effects of pollutants on aquatic organisms. Cambridge University Press, England. P 7.
|
|
|
|
|
Canli M, Atli G (2003). The relationships between heavy metal levels and the size of six Mediterranean fish species. Environ. Pollut. 121:129-136.
Crossref
|
|
|
|
|
Canli M, Kalay M (1998). Levels of heavy metals in tissues of Cyprinus carpio, Barbus capito and Chondrostoma regium from Seyhan River, Turkey. Turk. J. Zool. 22:149-157.
|
|
|
|
|
Effiong BN, Mohammed I (2008). Effect of seasonal variation on the nutrients composition in selected fish species in Lake Kanji, Nigeria. Nat. Sci.1-5.
|
|
|
|
|
El-Nemr A (2003). Concentrations of certain heavy metals in imported frozen fish in Eygpt. Eygpt. J. Aquat. Biol. Fish. 7:139-154.
|
|
|
|
|
Fafioye OO, Ogunsanwo BM (2006). The comparative toxicities of cadmium, copper and lead to Macrobranchium rosenbergii and Penaeus monodon post lavae. Afr. J. Agric. Res. 2(1):31-35.
|
|
|
|
|
Fafioye OO, Ogunsanwo BM (2007). Carbohydrate reserves and metal accumulation of the Nile Tilapia, Oreochromis niloticus after treatment with heavy metals. Ife J. Sci. 9(2):151-158.
|
|
|
|
|
Fafioye OO, Adeogun AO, Omoniyi IT (2008). Lethal limits and respiration in the cichlid fishes, Tilapia zillii, Sarotherodon galilaeus, S. melanotheron and Orechromis noliticus exposed to effluent from Chemistry Department Laboratories. J. Appl. Sci. Environ. Manage. 12(1):63-66.
|
|
|
|
|
FAO (1983). Manual of Methods in Aquatic Environmental Research, Analyses of metals and organochlorines in fish. FAO Fisheries Technical Paper, Rome. P 212.
|
|
|
|
|
George U, Asuquo F, Idung J, Andem A (2013). Bioaccumulation of heavy metals in three fresh water fishes caught from Cross River system. Eur. J. Exp. Biol. 3(3):576-582.
|
|
|
|
|
Gupta BN, Mathur AK (1983). Toxicity of heavy metals. Ind. J. Med. Sci. 37:236-240.
|
|
|
|
|
Kalay M, Canli M (2000). Elimination of essential (Cu, Zn) and nonessential (Cd, Pb) metals from tissue of a freshwater fish Tilapia zillii following an uptake protocol. Turk. J. Zool. 24:429-436.
|
|
|
|
|
Khaled A (2004). Heavy metal concentrations in certain tissues of five commercially important fishes from El-Mex Bay, Al-Exandria, Egypt. pp. 1-11.
|
|
|
|
|
Larsson A, Haux C, Sjobeck M (1985). Fish physiology and metal pollution: Results and experiences from laboratory and field studies. Ecotox. Environ. Safety 9:250-281.
Crossref
|
|
|
|
|
Mansour SA, Sidky MM (2002). Heavy metals contaminating water and fish from Fayoum Governorate, Egypt. Food Chemistry 78:15-22.
Crossref
|
|
|
|
|
Metcalfe C, Metcalfe T, Ray S, Paterson G, Koenig B (1999). Polychlorinated Biphenyls and Organochlorine Compounds in Brain, Liver and Muscle of Beluga Whales (Delphinapterusleucas) from the Arctic and St. Lawrence Estuary. Mar. Environ. Res. 47:1-15.
Crossref
|
|
|
|
|
Mwashot BM (2003). Levels of cadmium and lead in water, sediments selected fish species in Mombasa, Kenya. Western Ind. Ocean J. Mar. Sci. 2(1):25-34.
|
|
|
|
|
Nemesok JG, Huphes ZGM (1988). The effects of copper sulphate on some biochemical parameters of rainbow trout. Environ. Pollut. 49:77-85.
Crossref
|
|
|
|
|
Oladimeji AA, Offem BO (2009). Metals and organic compounds in fish of the Ogun River. Water, Air and soil Pollution 44:191-201.
Crossref
|
|
|
|
|
Olaifa FE, Olaifa AK, Adelaja AA, Owolabi AG (2004). Heavy metal contamination of Clarias gariepinus from a lake and Fish farm in Ibadan, Nigeria. Afr. J. Biomed. Res. 7:145-148.
|
|
|
|
|
Romeo M, Siau Y, Sidoumou Z, Gnassia Barelli M (1999). Heavy metal distribution in different fish species from the Mauritania coast. Sci. Total Environ. 232:169-175.
Crossref
|
|
|
|
|
Rompala JM, Rutosky FW, Putnam DJ (2004). Concentrations of environmental contaminants from selected waters in Pennsylvania.U.S. Fish Wildl. Serv. Rep., State College, Pennsylvania. P 102.
|
|
|
|
|
Sivaperumal P, Sankar TV, Viswanathan PG (2007). Heavy metal concentrations in fish, shellfish and fish product from internal markets of India vis-a-vis international standards. Food Chemistry 102:612-618.
Crossref
|
|
|
|
|
Sunday AD, Aminu AY, Augustina DO (2013). Elemental analysis of flesh, bones and gills of Oreochromis niloticus consumed in Nigeria for improvement of nutrition and health. Int. J. Sci. Res. Environ. Sci. 1(6):122-133.
Crossref
|
|
|
|
|
Taha AA (2004). Pollution sources and related environmental impacts in the new communities, Southeast Nile Delta, Egypt. Emirat. J. Eng. Res. 19:1-44.
|
|
|
|
|
Telisman S, Cvitkovic P, Jurasovic J, Pizent A, Gavella M, Rocic B (2000). Semen quality and reproductive endocrine function in relation to biomarkers of lead, cadmium, zinc, and copper in men. Environ. Health Perspect, 108(1):45-53.
Crossref
|
|
|
|
|
Van Aardt WJ, Erdman R (2004). Heavy metals (Cd, Pb, Cu, Zn) in mudfish and sediments from three hard-water dams of the Mooi river catchment, South Afr. Water 30:211-218.
Crossref
|
|
|
|
|
WHO (2004). Health Criteria and Other Supporting Information in Guideline for Drinking Water Quality (3rd Eds), World Health Organization, Geneva. pp. 301-388.
|
|
|
|
|
Yilmaz AB (2003). Levels of heavy metals (Fe, Cu, Ni, Cr, Pb and Zn) in tissue of Mugil cephalus and Trachurus mediteraneus from Iskenderun bay, Turkey. Environ. Res. 92:277-281.
Crossref
|
|
|
|
|
Yilmaz AB (2005). Comparison of heavy metal levels of Grey Mullet (Mugil cephalus L.) and sea bream (Sparus aurata L.) caught in Iskendrun Bay (Turkey). Turk. J. Vet. Anim. Sci. 29:257-262.
|
|