ABSTRACT
Nature of gene action and genetic parameters for disease resistance are important attributes in developing resistant cultivars. This provides the sustainable, economically justifiable and environmentally friendly means of controlling plant diseases. In this study 6 x 6 full diallel cross involving genetically divergent maize inbred lines was performed with the aim of developing resistant cultivars against Maize Lethal Necrosis (MLN) disease under MLN disease hot spot areas in Mlangarini, Ngaramtoni and Kiru six in the Northern Zone of Tanzania during 2015 cropping season. The experimental materials consisted of thirty single cross hybrids, six parents and two local checks. The experiment was laid down in a randomized complete block design (RCBD) with three replications per location. The general combing ability (GCA) and specific combining ability (SCA) effects were significantly different for MLND response among genotypes across all locations. The combined analysis revealed that GCA was highly significant at (P≤0.001) than SCA in all locations with mean squares of (5.551***), (1.61***) and (4.527***) for Mlangarini, Kiru six and Ngaramtoni respectively. The GCA: SCA ratios were 1.894, 1.726 and 1.403 for Mlangarini, Kiru six and Ngaramtoni respectively. The implication of GCA/SCA ratio of more than a unity proves that GCA is significant in all locations where this study was conducted. The results also revealed the presence of both additive and non-additive genetic effects, with the former more pronounced than the later. This implies that developing composite variety will be the better option in combating the disease. However the best cross was observed between CML 144 X CML444 with mean square -0.10, -0.45* and -0.18* for Mlangarini, Kiru six and Ngaramtoni respectively.
Key words: Diallel cross, general combing ability (GCA), heritability, Maize Lethal Necrosis disease (MLND), specific combining ability (SCA), Zea mays.
The popularity of maize in Africa has been increasing to the extent of replacing traditional crops like sorghum and millet. (DeVries and Toenniessen, 2001). An estimate of 90% of the maize produced in Africa is consumed as food (Katinila et al., 1998). Despite the importance of maize as the main staple crop, average yields in farmers’ fields are relatively low averaging 1.2 metric tons per hectare compared to the estimated potential yields of 4–5 metric tons per hectare (WEMA, 2010). While farmers are keen on increasing maize productivity, their efforts are hampered by a wide range of constraints such as pest and diseases, low soil fertility and unpredictable weather. Currently the outbreak (MLN) disease has been a serious threat for maize production in Tanzania and other East African countries (Wangai et al., 2012). According to the survey conducted by CIMMYT in 2012, potential yield loss of more than 60% was reported in the affected areas. Infection rate and damage can be very high seriously affecting yields and sometimes causing complete crop loss (Wangai et al., 2012). The disease is caused by the co-infection of Maize Chlorotic Mottle Virus (MCMV) and any potyvirus group such as Sugar Cane Mosaic Virus (SCMV).
Biologically maize is the natural host for more than 50 viruses and an experimental host for about 30 more (Lapierre and Signoret, 2004), but only some cause diseases that seriously affect yield (Ali and Yan, 2012; Redinbaugh and Pratt, 2009). Among the most aggressive are the members of the Potyviridae and Maize Chlorotic mottle virus (MCMV), which form the devastating complex known as maize lethal necrosis virus (Uyemoto et al., 1980; Wangai et al., 2012). Although plants have evolved passive and active defense mechanisms that are responsible for the suppression of virus multiplication and spread, such mechanisms require interaction of plant and viral factors to confer plant resistance or susceptibility (Gomez et al., 2009). Identifying the loci conferring resistance to virus diseases, offers an approach to develop genetically resistant lines that are able to reduce the yield losses caused by diseases. Estimation of genetic effects and variances in a population is of great importance to plant breeders in making decision concerning the type of breeding programme to be used and in selecting breeding materials that will show the greatest success against various stresses.
Therefore, in this study full diallel cross was used in order to gather important genetic information of the parental materials to combat maize lethal necrosis virus. This technique has been extensively used and hailed by plant breeders as a long over-due methodology for rationalizing the genetic study of continuous variation (Jawahar, 2006). The strength of diallel technique is that, additional information such as reciprocal effects, maternal and paternal effects and allelic distribution can be obtained quite in early generation such as F1, thus useful to define breeding strategy without losing much time. Moreover, diallel design is a useful tool of obtaining combining ability of the parents used in the cross. Combining ability has a prime importance in plant breeding since it provides information for the selection of parents and also provides information regarding the nature and magnitude of involved gene action (Griffing, 1956). The knowledge of genetic structure and mode of inheritance of different characters help breeders to employ suitable breeding methodology for their improvement (Kiani et al., 2007). However little is known about MLN disease and there are limited information about the viruses causing the disease. Although Sugar Cane Mosaic virus was common in East Africa and well known in sugar cane crops, Maize Chlorotic Mottle Virus is new virus in most part of the region.
The experimental materials used in this study were thirty single cross developed from six maize inbred lines (CKH 10767, CKH 114272, CML312, CML444, CML503 and CML144) and 2 local checks (SeedCo527 and Selian H308). The six inbred lines were selected based on their genetic diversity of the disease response. While CML144 was known to be resistant, CML503 was the highly susceptible material. The rest lines are moderately resistant to the disease.
The breeding nursery was established at Kiru six secondary school gardens under irrigation system during 2014 off season for developing single cross hybrids. Crossings were performed in 6 x 6 full diallel fashion according to Griffings (1956) Design 1 model 1 using six heterotically divergent parents. In order to increase genetic variation, resistant, moderately resistant and susceptible inbreds were used in the ratio of 3:2:1. Both ear and tassel bagging were done prior to flowering in order to avoid unintended cross and self-pollination. In the breeding nursery sowing dates were adjusted to facilitate nicking in flowering so that sufficient crosses can be made.
Evaluation
trials were laid
down in a randomized complete block design (RCBD) with three replications. Each entry was planted in one row plot of 5 m long; inter and intra row spacing was kept to 0.75 m by 0.30 m respectively. Three spreader rows of maize inbred line CML 503 were planted as border rows as the highly susceptible check. Pesticides were not applied to allow movement of insect vectors in the field
. The F
1’s hybrids were evaluated for general and specific combining ability for disease resistance in Mlangarini, Kirusix and Ngaramtoni in the Northern zone of Tanzania. These are the locations currently having high MLN disease occurrences. Parental lines were concurrently evaluated together with the developed hybrids in order to study their genetic information on disease scores, days to 50% flowering and grain weight from ten plants randomly selected in each row. These plants were tagged three weeks after emergence. Disease severity scores were rated on scale of 1-5 according to Shekhar and Kumar (2012) as follows: 1= Resistant (No Symptoms), 2=Moderately Resistant, 3= Moderately Susceptible, 4= Susceptible, 5= Highly Susceptible (plant dead completely). The disease scores were recorded three weeks after emergence (3WAE), six weeks after emergence (6WAE), nine weeks after emergence (9WAE) and twelve weeks after emergence (12WAE). Data collected were normalized using square root transformation and subjected to analysis of variance (ANOVA) using Window stat version 9.2 software developed by international crops research institute for the semi-arid tropics (ICRISAT), Hyderabad, India. The main focus in the analysis was given only to data relating MLN disease response. The statistical model used was;
Yij= µ+gi+gj+sij+É›ij,
where: Yij is the mean value of the combination (i ≠ j) or parental (i = j); µ is the general mean; gi, gj are the effects of the general combining ability of ith and jth parent respectively; Sij is the specific combining ability effect for the crosses. For this model to be adequate the following assumption must be considered. Firstly, it is assumed that the trait to be studied is not under the influence of the multiple allelism. Secondly there should be no maternal effects, while the genotypes studied should be a regular diploid in nature. Another assumption is that there is no epistasis and each gene assorts independent of the other.
The Griffings fixed model was used since the parental materials were chosen from the population based on their known traits in order to study both GCA and SCA focusing on MLN disease response. While the GCA effects can aid breeders to exploit existing variability in breeding materials to choose genotypes having desirable attributes and to distinguish relatedness among the breeding materials (Ai-Zhi et al., 2012; Matta and Viana, 2003; Sprague and Tatum, 1942); the SCA is the manifestation of non-additive component of genetic variance and associated with interaction effects, which may be due to dominance and epistatic component of genetic variation that are non-fixable in nature. Such non-fixable components are potential parameters for heterosis breeding which is very much useful in maize and other crops where commercial exploitation of heterosis is required.
The heterotic effects of F1 are normally estimated as percentages over mid-parent and better parent using the following formula:
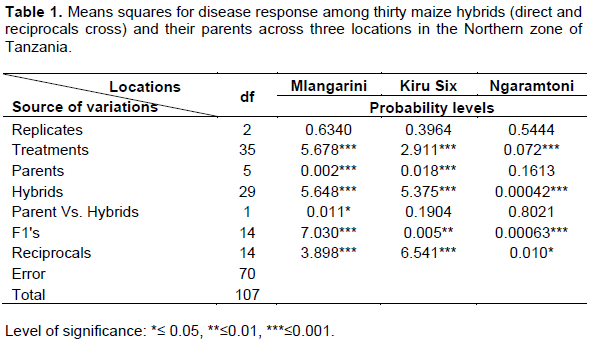
Analysis of variance showed highly significant differences among genotypes for MLN response indicating the presence of sufficient genetic variation among treatments (Table 1). On the other hand there are highly significant differences for disease resistance among parents (P≤0.001) at Mlangarini and Kiru six but not significant in Ngaramtoni.
Reciprocal effects were significant in Mlangarini and Kiru six at (P≤0.001) indicating the presence of maternal effects in controlling the disease reaction among the studied genotypes (Table 1).
Both GCA and SCA were determined in respect to disease response against maize lethal necrosis virus in the study areas. However, GCA was found to be significant at P≤0.001 than SCA (Table 2).

The parents respond differently to MLN based on their genetic background. Generally CML 144, CML 503 and CML 444 were the best combiners. However only CML 144 showed highly negative GCA which imply that it is the best combiner for disease resistance whereas CML 503 and CML 444 were found to be susceptible lines with positive and highly significant GCA effects (Table 3). Moreover, only one cross (CML 144 x CML444) showed the promising results with SCA effects of (-0.45*) in Kirusix. Other cross with significant positive SCA effect were CML312 x CML444 (0.48*) and (0.73**) in Mlangarini and Ngaramtoni respectively. For the selection purposes therefore only the crosses with significant negative values are desirable for disease resistance, while the significant positive values indicate the susceptibility. In this case CML312 x CML444 will not be a useful cross (Table 4).
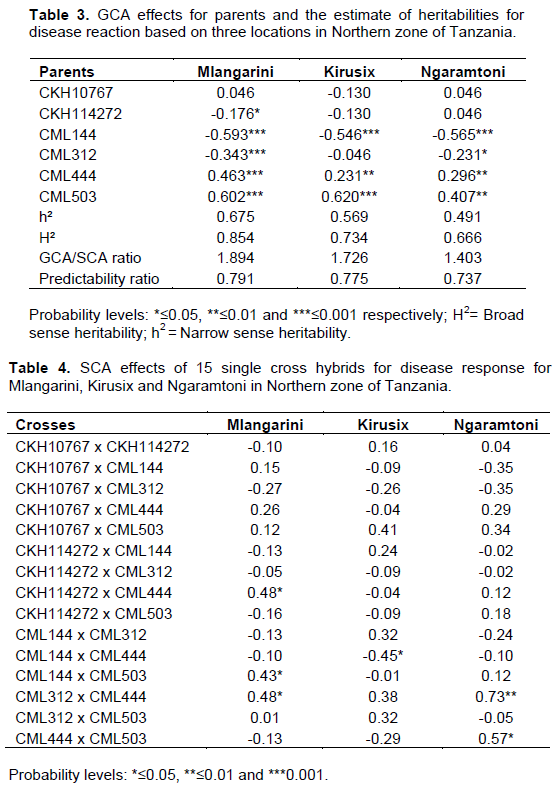
The estimate of narrow sense heritability (h2) was moderate, ranging from 0.491 to 0.675 than the broad sense heritability (H2) which ranged from 0.666 to 0.854 for the MLN. This result was in agreement with that reported by Mahmoud et al. (1990), that the low narrow sense heritability indicates that environmental factors had pronounced effects for the disease response.
On the other hand the predictability ratio ranged from 0.737 to 0.791. According to Patel et al. (2014) the predictability ratio approaching unity indicates the
preponderance of additive genetic effects (Table 3).
Estimates of heterosis among mid (MP) and better (BP) parents
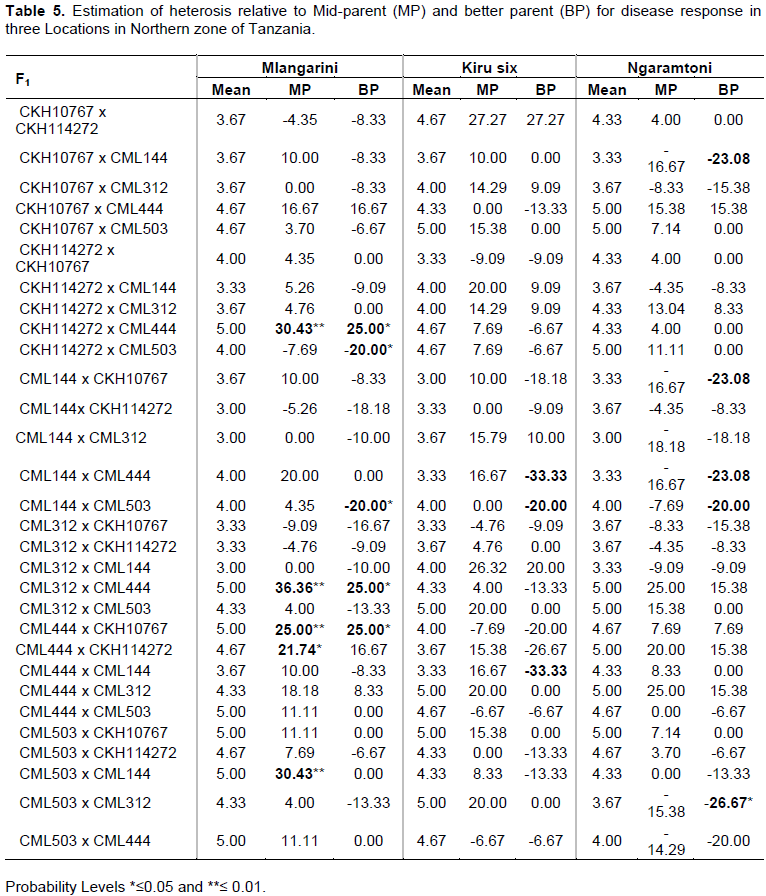
Heterosis is the deviation in performance among homozygous parents and their resulting off-springs. Significant differences were observed among 30 F1 hybrids for disease response. The heterosis for the yield would have been very important information in this study. However it was not possible to obtain adequate data due to excessive missing variables on yield since most of the genotypes dead before maturity thus only heterosis for disease reaction is reported. Since disease is undesirable phenomenon therefore heterosis in the negative direction will be favoured in the selection of the best crosses. The exceptional crosses both positive and negative are indicated in bold (Table 5). The best cross combinations are therefore those showed significant negative values. These includes CKH 114272 X CML 503 (-20*), CML 144 X CML 503 (-20*) in Mlangarini and CML 503 x CML 312 (-26.67) in Ngaramtoni. The poor crosses are the ones showed highly significant positive values. For example CML312 x CML444 (36.36**) and CML503 x CML144 (30.43**) are among the poor crosses for disease resistance.
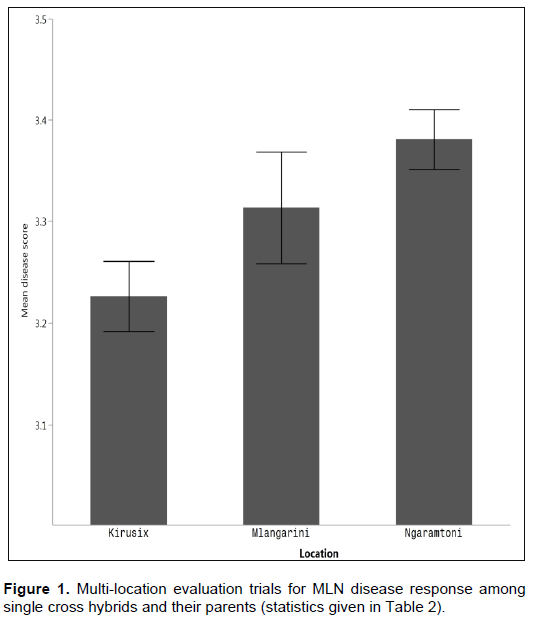
In respect to the experimental findings it is evident that both additive and non-additive gene effects were important with predominance of additive gene effects in inheritance of resistance trait. On the basis of general combining ability the most promising parents identified were CML144, CML312 and CKH114272. For SCA effects for disease resistance, the promising cross was CML 144 x CML444. The highly significant GCA principally imply the preponderance of additive genetic effects, therefore breeding strategy for controlling the disease is mainly to focus on developing composite or synthetic cultivars. As indicated in (Table 1), there is no significant difference among parents in Ngaramtoni location, implying that no parents showed significant resistance to MLN virus. This revealed that, there is a favourable environment for the disease progression than other locations where the study was also conducted (Figure 1). Derera et al. (2007) also found that disease development was highly affected by the environment indicating that incidence and severity may differ between locations and seasons, and between seasons within location.
The authors have not declared any conflict of interests.
REFERENCES
|
Ai-Zhi LV, Zhang H, Zhang ZX, Tao YS, Bing YU, Zheng YL (2012). Conversion of the statistical combining ability into a genetic concept. J. Integr. Agric. 11(1):43-52.
Crossref
|
|
|
|
Ali F, Yan J (2012). Disease Resistance in Maize and the Role of Molecular Breeding in Defending against Global Threat. J. Integr. Plant Biol. 54:134-151.
Crossref
|
|
|
|
|
DeVries J, Toenniessen G (2001). Securing, the Harvest: Biotechnology, Breeding and Seed Systems for African Crops. The Cromwell Press, Trowbridge, Wiltshire, UK.
|
|
|
|
|
Derera J, Tongoona P, Vivek B, VanRij N, Laing M (2007). Gene action determining Phaeosphaerian leaf spot disease resistance in Experimental maize hybrids; African Centre for Crop improvement; University of Kwazulunatal. S. Afr. J. Plant Soil. 24:138-143.
Crossref
|
|
|
|
|
Gomez P, Rodriguez-Hernandez AM, Moury B, Aranda MA (2009). Genetic resistance for the sustainable control of plant virus diseases: Breeding, mechanisms and durability. J. Plant Pathol. 125:1-22.
Crossref
|
|
|
|
|
Griffing B (1956). Concept of general and specific combining ability in relation to diallel crossing systems. Aust. J. Biol. Sci. 9:463-493.
Crossref
|
|
|
|
|
Jawahar RS (2006). Statistical and Biometrical Techniques in Plant Breeding; New International (P) Ltd, Publishers, New Delhi.
|
|
|
|
|
Katinila N, Moshi A,Verkuij H, Mwangi W, Anandajagayasekerum P (1998). Adoption of Maize Technologies in Southern Tanzania.International Maize and Wheat Improvement Center (CIMMYT) Mexico. The United Republic of Tanzania and the Southern Centre for Cooperation in Agricultural Research (SACCAR).
|
|
|
|
|
Kiani G, Nematzadeh S, Alisha O (2007). Combining ability in cotton cultivars for Agronomic traits. Int. J. Agric. Biol. 9:74-79.
|
|
|
|
|
Lapierre H, Signoret P (2004). Viruses and virus diseases of poaceae (gramineae).Paris: INRA Editions.
|
|
|
|
|
Mahmoud I, Rashed M, Fahmy E, Abo D (1990). Heterosis, Combining ability and type of gene actions in a 6x6 diallel of maize. The Proceedings of the third conference of Agricultural Development Research, held on 22-24 December, 1990; Cairo, Egypt.
|
|
|
|
|
Matt FP, Viana JM (2003). Analysis of general and specific combining abilities of popcorn populations including selfed parents. J. Genet. Mol. Biol. 26(4):465-471.
Crossref
|
|
|
|
|
Patel S, Patel M, Pateland B, Patel J (2014). Combining ability and gene action for grain yield and agronomic traits in pearl millet restorer lines; Elect. J. Plant Breed. 5: 394-401, Gujarat, India.
|
|
|
|
|
Redinbaugh MG, Pratt RC (2009).Virus Resistance. In: Bennetzen JL, Hake SC, (eds). Handbook of maize: It's Biology. Springer, New York, pp. 251-268.
Crossref
|
|
|
|
|
Shekhar M, Kumar S (2012). 2nd eds. Inoculation Methods and Disease Rating; Scales for Maize Diseases. Directorate of Maize Research, Indian Council of Agricultural Research, New Delhi.
|
|
|
|
|
Sprague GF, Tatum RL (1942). General vs Specific combining ability in single cross of corn. J. Am. Soc. Agron. 34:923-932.
Crossref
|
|
|
|
|
Uyemoto J, Bockelman D, Claflin L (1980). Severe Outbreak of Corn Lethal Necrosis Disease in Kansas. Plant Dis. J. 64:99-100.
Crossref
|
|
|
|
|
Wangai AW, Redinbaugh MG, Kinyua ZM, Miano DW, Leley PK, Kasina M, Mahuku G, Scheets K, Jeffers D (2012). First report of Maize chlorotic mottle virus and Maize Lethal Necrosis in Kenya. Plant Dis. 96:1582-1582.
Crossref
|
|
|
|
|
Water Efficient Maize for Africa (WEMA) project policy brief (2010). Mitigating the impact of drought in Tanzania.
|
|