Full Length Research Paper
ABSTRACT
Plant extracts are very cost effective and eco-friendly, thus, can be an economic and efficient alternative for the large-scale synthesis of nanoparticles. The preparation of stable, uniform silver nanoparticles by reduction of silver ions with Emblica officinalis, Terminalia catappa and Eucalyptus hybrida extract is reported in the present paper. It is a simple process of global research interest for obtaining silver nanoparticles in least amount of time. These nanoparticles were characterized with UV-Vis spectroscopy, X-ray diffraction (XRD), transmission electron microscopy (TEM) and energy diffraction X- ray (EDX analysis which revealed that the silver nanoparticles are polydisperse and of different morphologies ranging from 20 to 80 nm in size. XRD results reveal that these nanostructures exhibit a face-centered cubic crystal structure. The UV/Vis spectra absorption peak confirms their production. Pioneering of reliable and eco-friendly process for synthesis of metallic nanoparticles biologically is an important step in the field of application of nanobiotechnology. Thus, these silver nanoparticles (Ag-NPs) may prove as a better candidate for drugs and can potentially eliminate the problem of chemical agents because of their biogenic nature. The indiscriminate use of antibiotics has fuelled the development of drug resistance at an alarming rate. To overpower this burning problem, the Ag-NPs may prove to be a universal solution.
Key words: Nanobiotechnology, silver nanoparticles (Ag-NPs), Emblica officinalis, Terminalia catappa and Eucalyptus hybrida.
INTRODUCTION
MATERIALS AND METHODS
RESULTS AND DISCUSSION
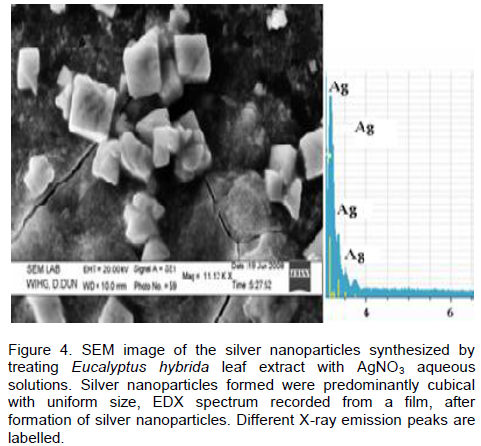
CONCLUSION
In the present study, plant extracts were studied for the synthesis of Ag-NPs. We have reported a simple biological process for synthesizing Ag-NPs. The FTIR analysis confirms capping over Ag-NPs. These capped Ag-NPs will help to enhance the stability of Ag-NPs in a colloidal solution by preventing aggregation of particles. Because they are capped by biomolecules and they may serve as a better candidate for the drug delivery systems. However, synthesis of nanoparticles using plant extracts can potentially eliminate the problem of chemical agents, which may have adverse effects in its application, thus making nanoparticles more biocompatible.
CONFLICT OF INTERESTS
The authors did not declare any conflict of interest.
ABBREVIATIONS
XRD, X-Ray diffraction; Ag-NPs, silver nanoparticles; NT, nanotechnology; TEM, transmission electron microscopy; EDX, energy diffraction X- ray; FFT, fast Fourier transform; FTIR, Fourier transform infrared spectroscopy; SAED, selected area electron diffraction.
REFERENCES
|
Catauro M, Raucci MG, De Gaetano FD, Marotta A (2004). Antibacterial and bioactive silver-containing Na2O CaO 2SiO2 glass prepared by sol-gel method. J. Mater. Sci. Mater. Med. 15(7):831-837. |
|
|
Chanda S (2014). Silver nanoparticles (medicinal plants mediated): a new generation of antimicrobials to combat microbial pathogens – a review. In: Mendez-Vilas, A. (Ed.), Microbial Pathogens and Strategies for Combating Them: Science Technology and Education. FORMATEX Research Center, Badajoz, Spain. pp. 1314-1323. |
|
|
Chen JC, Lin ZH, Ma XX (2003). Evidence of the production of silver nanoparticles via pretreatment of Phoma sp. 3Æ2883 with silver nitrate. Lett . Appl. Microbiol. 37:105-108. |
|
|
Crabtree JH, Burchette RJ, Siddiqi RA, Huen IT, Handott LL, Fishman A (2003). The efficacy of silver-ion implanted catheters in reducing peritoneal dialysis-related infections. Perit. Dial. Int. 23(4):368-374. |
|
|
Dibrov P, Dzioba J, Gosink KK, Hase CC (2002). Chemiosmotic mechanism of antimicrobial activity of Ag (+) in Vibrio cholera. Antimicrob. Agents Chemother. 46: 2668-2670. |
|
|
Duran N, Alves OL, De Souza GIH, Esposito E, Marcato PD (2007). Antibacterial effect of silver nanoparticles by fungal process on textile fabrics and their effluent treatment. J. Biomed. Nanotechnol. 3:203-208. |
|
|
Gade AK, Bonde PP, Ingle AP, Marcato PD, Duran N, Rai MK (2008). Exploitation of Aspergillus niger for fabrication of silver nanoparticles. J. Biobased Mater. Bioenergy 2:243-247. |
|
|
Gajbhiye M, Kesharwani J, Ingle A, Gade A, Rai M(2009). Fungus-mediated synthesis of silver nanoparticles and their activity against pathogenic fungi in combination with fluconazole. Nanomed. NBM 5:382-386. |
|
|
Gonzalez AL, Noguezm C (2007). Influence of Morphology on the Optical Properties of Metal Nanoparticles. J. Comput. Theor. Nano- sci. 4 (2): 231-238. |
|
|
Gross M, Winnacker MA, Wellmann PJ (2007). Electrical, Optical and Morphological Properties of Nanoparticle Indium-Tin-Oxide Layers. Thin Solid Films 515 (24): 8567-8572. |
|
|
Hamouda T, Baker JR (2000). Antimicrobial mechanism of action of surfactant lipid preparations in enteric Gram-negative bacilli. J. Appl. Microbiol. 89:397-403. |
|
|
Hemali P, Pooja M, Sumitra C (2014). Green synthesis of silver nanoparticles from marigold flower and its synergistic antimicrobial potential. Arabian J. Chem. (In Press). |
|
|
Huang J, Chen C, He N, Hong J, Lu Y, Qingbiao L, Shao W, Sun D et al. (2007). Biosynthesis of silver and gold nanoparticles by novel sundried Cinnamomum camphora leaf. Nanotechnology 18:105-106. |
|
|
Ingle A, Gade A, Pierrat S, Sonnichsen C, Rai M (2008). Mycosynthesis of silver nanoparticles using the fungus Fusarium acuminatum and its activity against some human pathogenic bacteria. Curr. Nanosci. 4:141–144. |
|
|
Jones SA, Bowler PG, Walker M, Parsons D (2004). Controlling wound bioburden with a novel silver-containing Hydrofiber dressing. Wound Repair Regen. 12 (3):288-294. |
|
|
Kim JY, Kim M, Kim HM, Joo J, Choi JH (2003). Electrical and Optical Studies of Organic Light Emitting Devices Using SWCNTs-Polymer Nanocomposites. Opt. Mater. 21 (1-3):147-151. |
|
|
Kim KJ, Sung WS, Suh BK, Moon SK, Choi JS, Kim JG, Lee DG (2009). Antifungal activity and mode of action of silver nano-particles on Candida albicans. Biometals 9(22):235-242. |
|
|
Mihail CR (2003). Nanotechnology: convergence with modern biology and medicine. Curr. Opin. Biotechnol. 14:337-346. |
|
|
Niraimathi KL, Sudha V, Lavanya R, Brindha P (2013). Biosynthesis of silver nanoparticles using Alternanthera sessilis (Linn.) extract and their antimicrobial, antioxidant activities. Colloids Surf. B Biointerfaces 102:288-291. |
|
|
Parak WJ, Gerion D, Pellegrino T, Zanchet D, Micheel C, Williams SC, Boudreau R, Le Gros MA, Larabell CA, Alivisatos AP (2003). Biological Applications of Colloidal Nanocrystals. Nanotechnology 14(7):15-27. |
|
|
Rafiuddin ZZ (2013). Bio-conjugated silver nanoparticles from Ocimum sanctum and role of cetyltrimethyl ammonium bromide. Colloids Surf. B Biointerfaces 108:90-94. |
|
|
Sadowski Z, Maliszewska IH, Grochowalska B, Polowczyk I, Kozlecki T (2008). Synthesis of silver nanoparticles using microorganisms. Mater. Sci. Pol. 26:419-425. |
|
|
Sastry M, Ahmad A, Khan MI, Kumar R (2003). Biosynthesis of metal nanoparticles using fungi and actinomycetes. Curr. Sci. 85: 162–170. |
|
|
Schultz DA (2003). Plasmon Resonant Particles for Biological Detection. Curr. Opin. Biotechnol. 14(1):13-22. |
|
|
Silver S, Phung LT (1996). Bacterial heavy metal resistance: new surprises. Annu. Rev. Microbiol. 50:753- 89. |
|
|
Smith AM, Duan H, Rhyner MN, Ruan G, Nie S (2006). A Systematic Examination of Surface Coatings on the Optical and Chemical Properties of Semiconductor Quantum Dots. Phys. Chem. Chem. Phys. 8(33):3895-3903. |
|
|
Stevanovic MM, Skapin SD, Bracko I, Milenkovic M, Petkovic J, Filipic M (2012). Poly (lactide-co-glycolide)/silver nanoparticles: synthesis, characterization, antimicrobial activity, cytotoxicity assessment and ROS-inducing potential. Polymer 53:2818-2828. |
|
|
Thakur M, Pandey S, Mewada A, Shah R, Oza G, Sharon M (2013). Understanding the stability of silver nanoparticles biofabricated using Acacia arabica (Babool gum) and its hostile effect on microorganisms. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 109: 344–347. |
|
|
Thirunavokkarasu M, Balaji U, Behera S, Panda PK, Mishra BK (2013). Biosynthesis of silver nanoparticles from extract of Desmodium gangeticum (L.) DC. and its biomedical potential. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 116:424-427. |
|
|
Vivek R, Thangam R, Muthuchelian K, Gunasekaran P, Kaveri K, Kannan S (2012). Green biosynthesis of silver nanoparticles from Annona squamosa leaf extract and its in vitro cytotoxic effect on MCF-7 cells. Process Biochem. 47:2405-2410. |
|
|
Wang HY, Li YF, Hua CZ (2007). Detection of ferulic acid based on the plasmon resonance light scattering of silver nanoparticles. Special Issue on China Japan-Korea Environmental Analysis, Talanta 72(5):1698-1703. |
|
|
Wei GH, Zhou Z, Liu Z. (2005). A Simple Method for the Preparation of Ultrahigh Sensitivity Surface Enhanced Raman Scattering (SERS) Active Substrate. Appl. Surf. Sci. 240(1-4): 260-267. |
|
|
Yamanaka M, Hara K, Kudo J (2005). Bactericidal actions of a silver ion solution on Escherichia coli, studied by energy-filtering transmission electron microscopy and proteomic analysis. Appl. Environ. Microbiol. 71:7589-7593. |
|
|
Zhao G, Stevens Jr SE (1998). Multiple parameters for the comprehensive evaluation of the susceptibility of Escherichia coli to the silver ion. Biometals 11:27-32. |
|
Copyright © 2024 Author(s) retain the copyright of this article.
This article is published under the terms of the Creative Commons Attribution License 4.0