ABSTRACT
This study assessed handling practices and microbiological contamination of salted sun-dried Nile Perch, Lates niloticus, commonly known as Kayabo in Tanzania. The processors of Kayabo were small-scale processors located at Kanyama and Mwaloni, Mwanza. Mixed methods (observations, face-to-face interviews, and microbiological sampling) were used to assess handling practices of processors and microbiological quality of Kayabo. Hierarchical cluster analysis on handling practices of Kayabo processors produced three distinct clusters of 15 (Cluster I), 10 (Cluster II), and 5 (Cluster III) processors. In general, the majority of processors had inadequate training on appropriate food handling, lack of hygienically designed facilities (building and cooling), poor sanitation, and the use of low-quality raw materials (rejects from industrial processing units). Assessment of microbiological quality of Kayabo indicated a significant (P<0.05) variation in Total Viable Counts, TVC (2.08 - 8.68 Log CFU/25 g), and Staphylococcus aureus (<1-5.49 log CFU/25 g) among the clusters. About 80% (12/15) and 42% (5/12) of the processors in 'Cluster I' exceeded the legal limits for TVC (5 Log CFU/25 g) and S. aureus (3 Log CFU/25 g), respectively. Strikingly, Escherichia coli and Listeria monocytogenes were below the quantification limits. Intervention measures for improvement on the raw materials, structure, and infrastructure, training, and adoption of good practices across the value chain are urgently needed to ensure the quality and safety of Kayabo.
Key words: Fish, salted sundried nile perch, handling practices, microbiological quality, Kayabo, Lake Victoria.
The Tanzanian fishery sector is among the leading (154.5 Million USD) exporting sectors in the country. It is the source of income and contributes to 1.3% of Gross Domestic Product, GDP (LVFO, 2009; URT, 2013; FAO, 2014). The sector employs more than 400,000 full-time artisanal fishermen and more than 4 million people in various fisheries-related activities (URT, 2013). About 30% of the total animal protein requirements in the country come from fish (URT, 2013). Lake Victoria contributes to 85% of the total export of fish and fish products (URT, 2013). Although the Lake has more than 290 fish species, only Nile perch (Lates niloticus), Tilapia (Oreochromis niloticus), and Sardines (Rastrineobola argentea) are of commercial importance (Abila, 2000; Akande and Diei-Ouadi, 2010; Kabahenda et al., 2009). The Nile perch is the only species from Lake Victoria industrially processed for the export market, whereas other species are processed solely for the domestic market (Kussaga et al., 2014; Medard et al., 2019).
Although fish processing companies for the export market enjoy strict control and monitoring of the food safety hazards, fish processing units for the domestic market like Kayabo processors are barely controlled (Kussaga et al., 2014; Baniga et al., 2019). Processors for the domestic market are small-scale with limited knowledge of proper fish handling and inadequately designed facilities to ensure the quality and safety of the products (Baniga et al., 2019). Processors use traditional processing techniques, particularly curing and sun-drying. When fish is salted and dried, it can have a shelf life of more than a year. Kayabo processing is mainly done by women and involves removal of guts, swim-bladder, and scales, before salting (Medard et al., 2019). Even though such traditional processing techniques are cheap and easy to operate, they expose the products to various food safety hazards.
Fish contains 70-84% water, 15-24% protein, 0.1-2.2% fat, and 1-2% minerals (Ghaly et al., 2010). Fish is an excellent dietary source of highly unsaturated fatty acid (UFA) and polyunsaturated fatty acid (PUFA), especially the omega-3 fatty acids, Eicosapentaenoic acid (EPA) and Docosahexaenoic acid (DHA) (El-Sherif and Abd El-Ghafour, 2016). Unless properly preserved or processed, fish is a highly perishable product that could be rendered unfit for human consumption within 12 h of capture under tropical conditions (Ghaly et al., 2010). Immediately after a fish dies, several physiological and microbial processes occur (Davies, 2009; Jinadasa, 2014). The quality and safety of fish is affected by capture method, processing, and handing conditions along the chain (Quang, 2005; Amos, 2007; Akande and Diei-Ouadi, 2010). However, the fish processing value chain for the domestic market is not well regulated.
Although the raw materials for the domestic-market oriented fish processors are tilapia and sardines, the supply is not yet sufficient and of low quality (as there is no cooling and hygienic handling practiced by most of the artisanal fisheries). Consequently, the small-scale Kayabo processors purchase rejects (undersize or low quality fish and fish frames) from the export-market oriented fish processing companies (Kabahenda et al., 2009), which focus on Nile Perch processing. The use of low quality raw materials associated with inadequate handling and processing conditions could result in more food safety problems.
Various studies have been conducted to assess the quality and safety of fish and fish products for the export market (Kussaga et al., 2014, 2017; Baniga et al., 2019); however, there is very restricted information on the quality and safety of fish or fish products like Kayabo for the domestic market. The status of premises and buildings of Kayabo processors has never been evaluated, limiting the opportunities for improvement. Moreover, it is not yet known to what extent consumers of processed fish by the domestic market-oriented units are exposed to various food safety hazards. Therefore, the purpose of this study was to investigate the handling practices and microbiological quality of Kayabo along the product value chain. The information generated from this study will be beneficial to consumers, small-scale processors, researchers, food control/competent authorities, and policymakers to set strategies for improving fish processing and handling practices to reduce food safety risks along the fish value chain.
Characteristics of Kayabo processors involved in the present study
The present study was conducted in two municipalities of Mwanza City, namely: Nyamagana and Ilemela, where Kanyama and Mwaloni processing sites are located, respectively. Thirty Kayabo processors, 18 being micro-scale (<10 employees) and 12 small-scale (10-49 employees) participated in this study. These processors produced whole salted sun-dried Nile perch (commonly known as Kayabo), salted sun-dried Nile perch heads (also known as Mapanki), salted sun-dried Nile perch trims (chips), and sun-dried Nile perch bones for fish meal production. Face-to-face interviews, observation, and sampling for microbiological assessments were carried out. However, Kayabo was the only product sampled and analysed for microbiological hazards as it was the main product processed.
Assessment of hygienic practices
A structured questionnaire and observation checklist were used to assess processors' knowledge of manufacturing and hygienic practices (transportation, processing conditions, packaging, and storage of raw materials and products). The questionnaire contained both open and closed-ended questions. The questions were on location and building/equipment layout, source of water, raw materials, sanitation, and personal hygiene, control by food control authorities, packaging, and demographic information of the respondents. The study assessed thirty Kayabo processing companies; the interviewees were either operators or owners.
Assessment of microbiological contamination
Selection of microbiological parameters
Five microbiological parameters including indicators of general process hygiene (Total Viable Counts, TVC), fecal hygiene (total coliforms and Escherichia coli), personnel hygiene (Staphylococcus aureus), and food safety (Listeria monocytogenes) were selected and analyzed (Jacxsens et al., 2009; Kussaga et al., 2017). Testing for TVC aimed at assessing the quality of raw materials, effectiveness of the handling procedures and hygiene conditions during processing, sanitary conditions of equipment and utensils, and time/temperature profile during storage and distribution. The natural habitat for E. coli is in the intestines of human and vertebrate animals (Berthe et al., 2013). Contamination of food with E. coli implies a risk that one or more enteric pathogens may have gained access to the food. Total coliforms indicate a higher probability that organisms of fecal origin are present and hence fecal contamination. The natural reservoirs for S.aureus are human skin, hair, and superficial mucous membranes (nose), the presence of large numbers indicates a possibility of enterotoxin production or fault in sanitary or production practices. The natural habitat of L. monocytogenes is in environments like soil, water, and fecal matter. Its presence in food is an indication of poor sanitation and animal control (FAO / CDR, 2013).
Sample collection and analysis
Systematic random sampling was used to take samples (whole Kayabo) from 30 processors. Each sample was inserted in a labelled sterile bag and then placed into a sterile cool box maintained at <5°C and transported in that condition to the accredited National Fish Quality Control Laboratory (NFQCL) for analysis. The samples that were not analysed on the same day of sampling were frozen at -18°C until analysed. Enumerations of TVC, total coliforms, E. coli, S. aureus were performed according to ISO 4833:2003 (ISO, 2003a), ISO 4831:2006 (ISO, 2006), ISO 7251: 2005 (ISO, 2005), ISO 6888 – 3: 2003 (ISO, 2003b), respectively. Detection of L. monocytogenes was as per ISO 11290-1:1996/Amd 1: 2004 (ISO, 2004) methods. The preparation of analytical reagents (diluents) and media was according to the manufacturers' instruction(s) and the specific test method requirements.
The preparation of analytical samples was in accordance with US-FDA Bacteriological Analytical Manual (2001). Weighing of 25 g Kayabo samples was by an analytical balance, Adventurer TM PRO OHAUS made in China. Then 225 ml buffered peptone water (BPW) was added to each sample and blended for 1 min to homogenize the mixture by using a stomacher (Seward STOMACHER R 3500 Lab System). Serial dilutions were aseptically prepared; 1 ml was taken from each dilution and plated on Plate count Agar (PCA, for TVC), Violet Red Bile Agar (VRBA for E.coli), and Baird Parker Agar (BPA, for S. aureus). The MPN was used for analysis of total coliforms. The incubation conditions were 30 ± 1°C for 72 ± 2 h for TVCs, 44 ± 1°C for 24 + 2 h for E.coli, 37 ± 1°C for 48 ± 2 h for S. aureus and 37 ± 1°C for 48 ± 2 h for total coliforms. For the detection of L. monocytogenes; 225 ml of Half Fraser broth for the primary enrichment were added to 25 g of each Kayabo sample and the mixture blended by using the stomacher for 1 min. Then, the homogenate was incubated at 30 ± 1°C for 24 ± 2 h.
Interpretation of microbiological results
Tanzanian standards (TZS), East African Standards (EAS), and Codex Alimentarius Commission (CAC) standards were used to interpret the results. The criteria used to interpret microbiological results are indicated in Table 1.
Statistical data analysis
A hierarchical cluster analysis with the furthest neighbour method and squared Euclidean distance (using SPSS Version 16.0 for Windows, SPSS Inc., Chicago, IL, USA) was used to analyse the hygienic performance data. A non-parametric test (Kruskal Wallis Non-Parametric H test) was performed to determine the differences among the clusters on the handling practices. Analysis of microbiological data was by R statistical package (R Development Core Team Version 3.0, Vienna, Austria), whereby One-way ANOVA test was carried out to determine whether there are differences among the factors. Means were separated by Duncan Multiple Range Test (DMRT). The significance level was set at P<0.05.
Hygienic performance of Kayabo processors
Characteristics of Kayabo processors
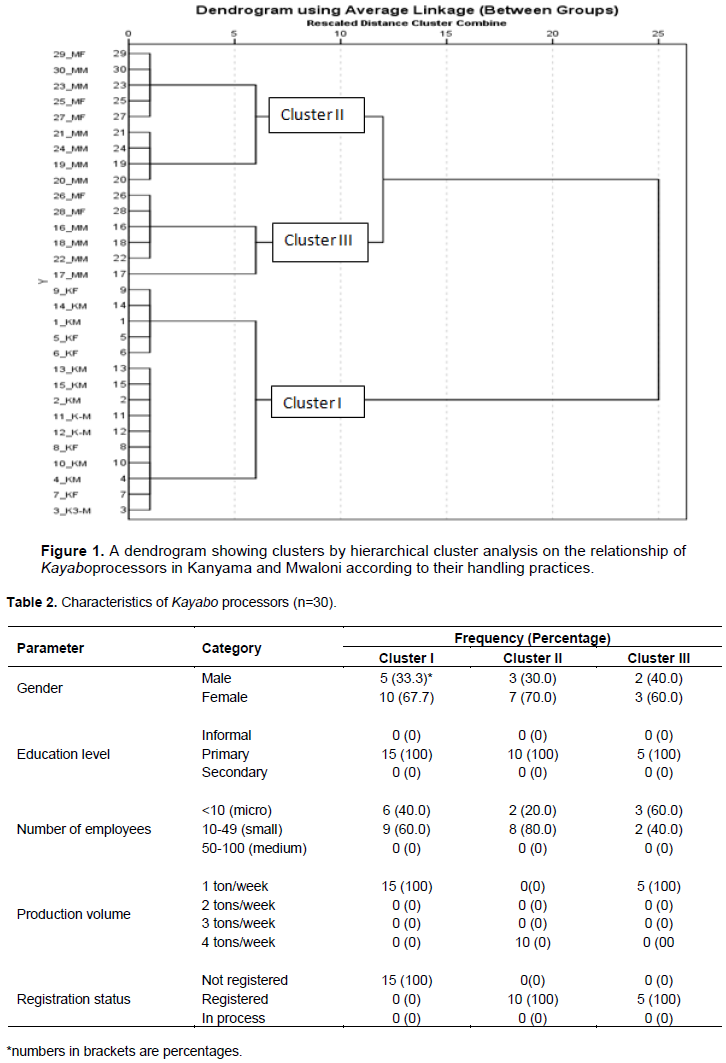
The hierarchical cluster analysis (average linkage) produced a dendrogram (Figure 1) showing three clusters. Cluster I had 15 processors from Kanyama, while cluster II composed of 10 processors and cluster III had 5 processors from Mwaloni. However, the processors (n=15) from Kanyama (Cluster I) were predominantly micro-scale, which were neither registered by the then Tanzania Food and Drugs Authority (TFDA), Municipal Council, nor by the Fisheries Department of the Ministry of Livestock and Fisheries (Table 2).
The majority (20/30 = 66.67%) of Kayabo processors were women. These processors were micro- (36.67%) and small- (63.33%) scale with a weekly processing capacity of 1-4 tons. Previous studies found that domestic-oriented fish processing companies are mainly artisanal and of small-scale (Kussaga et al., 2013; Akintola and Fakoya, 2017) and dominated by women carrying out fish smoking, salting, and drying (Kweka et al., 2006; Akande and Diei-Ouadi, 2010). The current study revealed that all processors (100%) had primary level education; consequently, none of them had attended any training on food hygiene and processing (Table 2). This situation could lead to inadequate processing and handling of Kayabo. Likewise, other studies reported low levels of formal education among the fishers and processors (Omwega et al., 2006; Olale et al., 2010; Ibrahim et al., 2011; Akintola and Fakoya, 2017). It is, however, reported that trained personnel could use appropriate fish handling practices in value chain processes (Davies, 2009; Akande and Diei-Ouadi, 2010), which might reduce post-harvest losses and improve the quality of the products. All processors in clusters I and III had a weekly processing capacity of 1 ton, whereas those in Cluster II could process 4 tons per week (Table 2). Processors in Cluster I operated in unregistered premises with poorly designed facilities. They did operate at an informal level without any control/monitoring from the food safety control authorities or health officers. Similar studies in Nigeria reported inadequate contact of processors with the extension agents, which deny them access to improved processing techniques (Ibrahim et al., 2011). Products from such premises are often of low quality and unsafe due to possible contamination with food safety hazards (FAO/ CDR, 2013).
Site and building layout
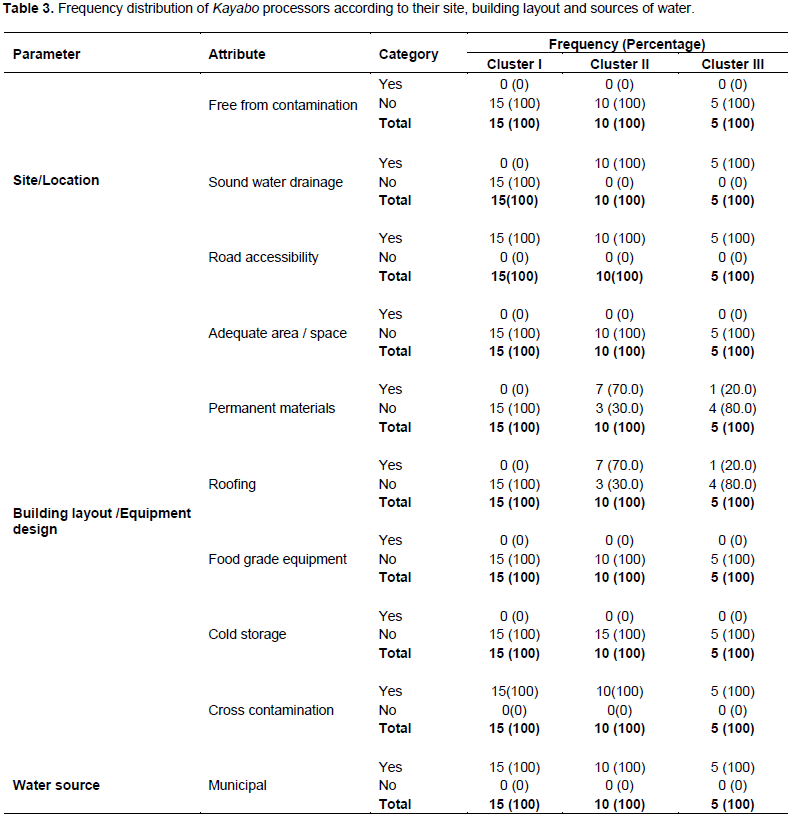
Table 3 shows the site and building layout of Kayabo processors. It shows that all processors operated in inadequately designed premises without good drainage systems. Particularly, 'Cluster I' processors had an un-fenced processing premise to control access of pests, animals, and unauthorized people. Animals were roaming around the processing premise. Although processors in clusters II and III had fenced premises, there was no control of food vendors (including fruits and vegetables) into the site. Animals and fruits and vegetables could be vehicles of microbiological hazards that could subsequently cross-contaminate the premises and products.
Moreover, the majority of processors in cluster I (100%), cluster III (80%), and cluster II (30%) had buildings not constructed with permanent materials. The buildings were makeshifts; constructed with wooden materials and thatched with plastic sheets or dried grasses without ceilings. The roofs could leak during rains to spoil or contaminate the products. Unfortunately, all processors used inadequately designed equipment and had no cold storage facilities. However, all processors used potable water from the Municipal supply (Table 3). Lack of adequate knowledge on proper food handling accompanied by poor design and layout of the premises (no separation between dirty and clean processes) could lead to cross-contamination. Previous studies reported inadequate facilities and the use of traditional tools and practices for fish processing (Ibrahim et al., 2011; Akintola and Fakoya, 2017). Such ill-practices are linked to low quality and unsafe products.
Sanitation and personnel hygiene
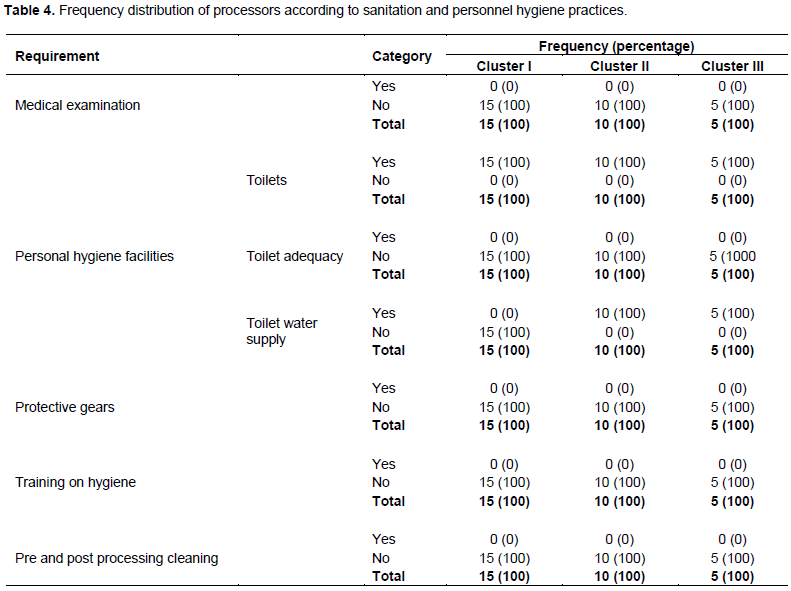
Table 4 shows the sanitation of premises and equipment and personal hygiene. It revealed that personnel were not subjected to any medical examination as required by the Tanzanian food laws and regulations. Tanzanian food law requires that all food handlers check their health when employed and after every six months (TFDCA, 2003). In addition, it prohibits personnel suffering from a septic sore, diarrhoea, chronic cough or septic sore throat, typhoid, paratyphoid fever, any Salmonella infection, dysentery, or any staphylococcal infection to handle food (TFDCA, 2003).
Moreover, all processors had no hygienically designed toilets with hand-washing facilities. Although cluster I processors had toilets, there were pit latrines located several meters away from the processing building and without water supply. Besides, food handlers had never received any training on food processing and good hygienic (GHP) and manufacturing (GMP) practices. Eventually, none of the personnel had put on personal protective gears and uniforms to minimize (cross) contamination. Such unhygienic conditions could result in product contamination with both pathogenic and spoilage microorganisms. Previous studies also observed inadequate conditions by artisanal and small-scale fish processors (Ibrahim et al., 2011; Akintola and Fakoya, 2017). The majority of food-borne outbreaks occur as a result of the failure of the food preparation procedures to adhere to good hygienic practices (FAO/WHO, 2013). Fish is a perishable product that needs good hygienic practices along the value chain to control microbial contamination and spoilage (Okonkwo et al., 1993; Nguyen et al., 2007). Inadequate cleaning and sanitation of processing equipment like containers, knives, and contact surfaces is a potential source of bacterial contamination in fish processing operations (Reij and Aantrekker, 2004; FAO/WHO, 2009) and could result into spoilage.
Source of raw materials, processing and packaging of final products
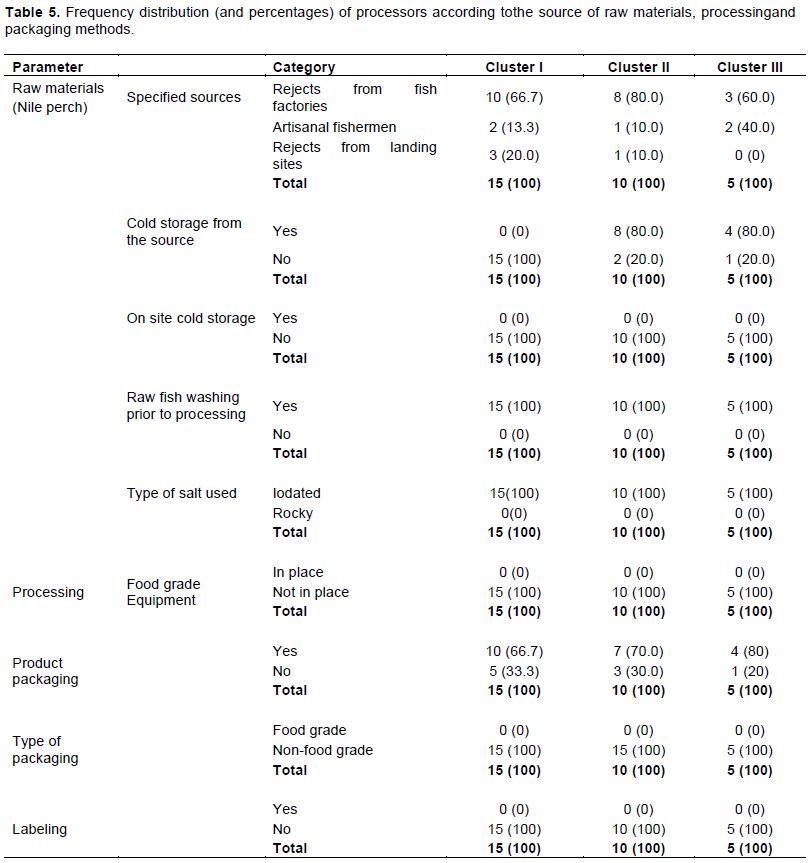
The majority of Kayabo processors (80% in cluster II, 67% in Cluster I, and 60% in Cluster III) obtained their raw materials (Lates niloticus) as rejects from large fishprocessing factories, for the export market (Table 5). While raw materials and products are properly handled in such factories, rejects are not hygienically handled or chilled. Although perishable raw materials like fish would require chill storage (between -1 and 4°C) to minimize the growth of micro-organisms and spoilage (Berkel et al., 2004; Ghaly et al., 2010), none of the processors had cold storage facilities. This could result in the proliferation of microorganisms and toxins formation. However, washing of Nile perch prior to processing was done by all processors and fish curing was done by using iodized salt. Rough food contact surfaces of drying racks (often made of wooden materials) are difficult to clean and sanitize; they are harbourage areas of pests, pathogenic and spoilage micro-organisms.
Generally, Kayabo was packaged in bulk (25-30 kg). Bulky packaging was carried out with non-food grade materials such as re-used, non-cleaned, and sanitized polyethylene bags. The packaging was carried out in a very unhygienic and rudimentary condition. After filling the polythene bags by hand, the products were compressed by using bare feet. These ill-practices could expose the products to various microbiological hazards (FAO/WHO, 2009; FAO/CDR, 2013). To ensure the quality and safety of Kayabo, proper, and hygienic packaging practices are the pre-requisites (FAO/WHO, 2009).
Assessment of microbiological contamination
The TVC were quantified in all clusters with levels of contamination ranging from 2.08 - 8.68 Log CFU/25 g (Table 6). While cluster I had 48% (12/15) of the processors exceeding the maximum allowable limit (5 Log CFU/25 g) for TVC; none of the processors from cluster II and cluster III exceeded the legal limit. This current study reported high TVC than a study in salted sun-dried Nile perch products which observed 2.9-4.5 log CFU/g (Baniga et al., 2019). Staphylococcus aureus counts ranging from <1 to 5.49 Log CFU/25 g were quantified from 47% (14/30) of processors; however, majority (12/14) were from Cluster I (Table 6). About 42% (5/12) of processors from Cluster I exceeded the allowable limit (3 Log CFU/25 g) for S. aureus. Although total coliforms were occasionally quantified in Cluster I (2/15 processors) and Cluster II (1/10 processors), they were within the legal limits. Unexpectedly, pathogens were either below the quantification (<1 Log CFU/25 g for Escherichia coli) or not detected (Absent, P/25 g for L. monocytogenes) throughout the study.
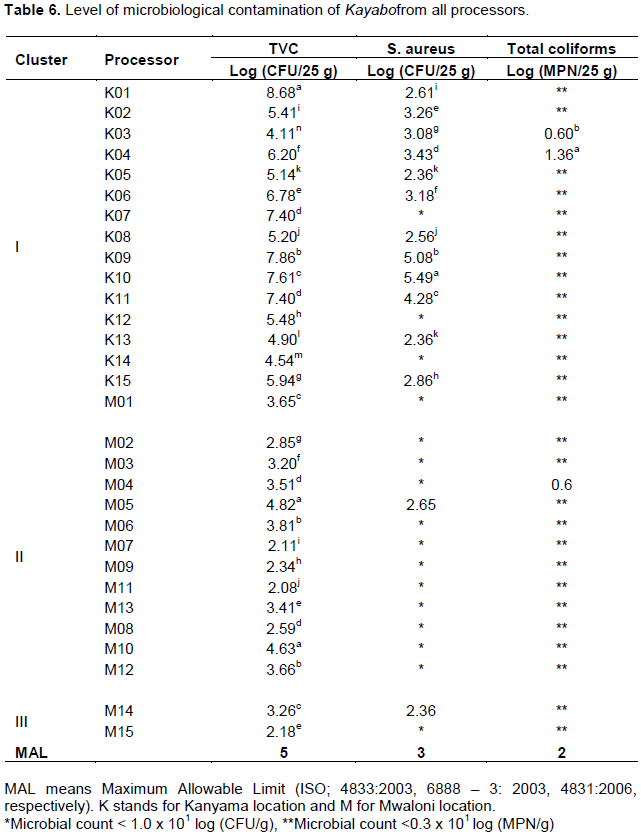
The counts beyond the acceptable limits for TVC (5.14 - 8.68 Log CFU/25 g) and S. aureus (3.08- 5.49 Log CFU/25 g) of the analysed samples from the majorityof processors in Cluster I are due to the observed unhygienic handling conditions of raw materials and finished products, poor sanitation and personal hygiene, use of non-food grade equipment (wooden materials as drying racks which are difficult to clean and sanitize hence harbouring microorganisms) and inadequate packaging conditions. According to Norman (1999) and Kussaga et al. (2017), possible causes of food contamination by enteric microorganisms (such as, Enterobacteriaceae and E. coli) are inadequate procedures of handling, packaging, storage, sanitation, and personal hygiene.
Despite the observed poor hygienic conditions, all Kayabo samples tested for total coliforms, E. coli and L. monocytogenes complied with the legal limits. This could be due to the effect of salt (Sodium chloride) on water activity (aw) as well as moisture content of Kayabo, which inhibits the growth and proliferation of pathogenic microorganisms. A similar study by Wijnker et al. (2006) on antimicrobial properties of salt (NaCl) in fish, fishery products, and natural sheep casings at different water activity levels found that microbiological (E. coli, Salmonella typhimurium, L. monocytogenes, S. aureus and E. coli O157: H7) activities stopped when water activity of 0.89 was reached. The inhibitory effects of sucrose and sodium chloride on S. aureus are primarily related to lower water activity (Dave and Ghaly, 2011). Sodium chloride has more inhibitory effects than glycerol in inhibiting the growth of spoilage microorganisms. Salting decreases water activity and has an inhibitory effect on pathogenic bacteria (Teklemariam et al., 2015).
The TVC variation was significantly different (P<0.05) among the clusters. Although coliforms and S. aureus were observed in all companies among the clusters, the variations were not significantly different. Post Hoc (Bonferroni) test indicated that a significant difference (P<0.05) in TVC between Clusters I and II and Clusters I and III (Table 7). As compared with other clusters, 'cluster I' had significantly high contamination with TVC (6.17 Log CFU/25 g) and S. aureus (3.38 Log CFU/25 g). The level of TVC and S. aureus was beyond the respective legal limits. Microbiological assessment results correspond with the prevailing conditions as cluster I had an un-fenced processing site to control pests, animals, and unauthorized people, as well as animals, could access the premises.
Kayabo processing is dominated by women, often without the knowledge of proper food handling and operating in very unhygienic conditions. The raw materials are of inadequate quality, lack of food-grade processing equipment, and poor packaging and storage conditions. Although the processors receive inadequate handled raw materials (as rejects) from fish processing companies accompanied by poor processing and storage conditions, some processors had products with less microbiological contamination. This could be attributed to the use of the curing process, which inhibits growth and possibly kill micro-organisms. If these processors are trained on best handling practices and receive quality raw materials, they could process products of good microbial quality. Moreover, fish companies for the domestic market did not receive the same level of control as for the export. Therefore, similar level of control in the domestic oriented companies is highly recommended.
The authors have not declared any conflict of interests.
The authors are thankful to the Tanzania Food and Drugs Authority (TFDA) for sponsoring this study.
REFERENCES
|
Abila RO (2000). Development of the Lake Victoria Fishery: A Boon or Bane for Food Security? Socio-economics of the Lake Victoria Fisheries Project Technical Report No. 7. IUCN-EARO, Nairobi 28 p.
|
|
|
|
Akande G, Diei-Ouadi Y (2010). Post-harvest Losses in Small-scale Fisheries: Case Studies in Five Sub-Saharan African Countries. Fisheries and Aquaculture Technical Paper. No. 550. FAO, Rome 72 p.
|
|
|
|
|
Akintola SL, Fakoya KA (2017). Small-scale fisheries in the context of traditional post-harvest practice and the quest for food and nutritional security in Nigeria. Agriculture and Food Security 6(1):34.
Crossref
|
|
|
|
|
Amos T (2007). Production and productivity of crustaceans in Nigeria. Journal of Social Science 15:229-233.
Crossref
|
|
|
|
|
Baniga Z, Dalsgaard A, Kusiluka LJM, Mdegela RH (2019). Microbial quality of Nile perch (Lates niloticus) and physico-chemical properties of salted sun-dried products sold at regional markets, Tanzania. African Journal of Microbiology Research 13(7):128-133.
Crossref
|
|
|
|
|
Berkel BM, van Boogaard B, Heijnen C (2004). Preservation of Fish and Meat. Digigrafi, Wageningen, the Netherlands P 86.
|
|
|
|
|
Berthe T, Ratajczak M, Clermont O, Denamur E, Petit F (2013). Evidence for coexistence of distinct Escherichia coli populations in various aquatic environments and their survival in estuary water. Appled Environmental Microbiology 79(15):4684-4693.
Crossref
|
|
|
|
|
Dave D, Ghaly AE (2011). Meat spoilage mechanisms and preservation techniques: A critical review. AmericanJouranl of Agriculture and Biological Sciences 6(4):486-510.
Crossref
|
|
|
|
|
Davies RM (2009). Traditional and improved fish processing technologies in Bayelsa State, Nigeria. European Journal of Scientific Research 26(4):539-548.
|
|
|
|
|
El-Sherif SAEH, Abd El-Ghafour S (2016). Investigation of the quality properties and nutritional values of four fish species from lake qaroun, Egyptiona International Journalof Chemical and Technical Research 9(4):16-26.
|
|
|
|
|
FAO (2014). Present and future markets for fish and fish products from small-scale fisheries - case studies from Asia, Africa and Latin America. FAO Report (Vol. 1033).
|
|
|
|
|
FAO/Corporate Document Repository, FAO/CDR (2013). Assurance of seafood quality Fisheries and Aquaculture Department.
|
|
|
|
|
FAO/WHO (2009). Code of practice for fish and fishery products. Rome, Italy. Available at:
View
|
|
|
|
|
Ghaly AE, Dave D, Budge S, Brooks MS (2010). Fish spoilage mechanisms and preservation techniques. American Journal of Applied Science 7(7):859-877.
Crossref
|
|
|
|
|
Ibrahim HI, Kigbu AA, Mohammed R (2011). Women's experiences in small scale fish processing in Lake Feferuwa fishing community, Nasarawa State, Nigeria. Livestock Research for Rural Development 23(3):1-8.
|
|
|
|
|
ISO (2003a). Microbiology of food and animal feeding stuffs - Horizontal method for the enumeration of microorganisms - Colony-count technique at 30 0C. 9 p. Available at:
View
|
|
|
|
|
ISO (2003b). Microbiology of food and animal feeding stuffs - Horizontal method for the enumeration of coagulase - positive staphylococci (Staphylococcus aureus and other species) - Part 1: Technique using Baird-Parkeragar medium- Amendment 1: Inclusion of precision data 11 p.
|
|
|
|
|
ISO (2004). Microbiology of food and animal feeding stuffs - Horizontal method for the detection and enumeration of Listeria monocytogenes - Part 1: Detection method 16 p.
|
|
|
|
|
ISO (2005). Microbiology of food and animal feeding stuffs- Horizontal Method for the detection and enumeration of presumptive E. coli - Most probable number technique. Available at:
View
|
|
|
|
|
ISO (2006). Microbiology of food and animal feeding stuffs - Horizontal Method for the detection and enumeration of coliforms - Most probable number technique. Available at:
View
|
|
|
|
|
Jacxsens L, Kussaga J, Luning PA, Van der Spiegel M, Uyttendaele M (2009). A microbial assessment scheme to support microbial performance measurements of food safety management systems. International Journal of Food Microbiology 134:113-125.
Crossref
|
|
|
|
|
Jinadasa BKKK (2014). Determination of Quality of Marine Fishes Based on Total Volatile Base Nitrogen test (TVB-N). Natural Science 12(5):106-111.
|
|
|
|
|
Kabahenda MK, Omony P, Hüsken SM (2009). Post-harvest handling of low-value fish products and threats to nutritional quality: a review of practices in the Lake Victoria region. Available at:
View
|
|
|
|
|
Kussaga JB, Jacxsens L, Luning PA, Tiisekwa BPM (2013). Diagnosis of food safety management systems performance in food processing sectors for export and domestic markets. African Journal of Food Science and Technology 4(10):240-250.
|
|
|
|
|
Kussaga JB, Luning PA, Tiisekwa BPM, Jacxsens L (2014). Challenges in Performance of Food Safety Management Systems: a case of Fish Processing Companies in Tanzania. Journal of Food Protection, 77(4):621-630.
Crossref
|
|
|
|
|
Kussaga JB, Luning PA, Tiisekwa BPM, Jacxsens L (2017). Microbiological performance of HACCP-based food safety management systems: a case of Nile perch processing company. African Journal of Food Science 11(7):200-214.
Crossref
|
|
|
|
|
Kweka J, Musa J, Kabelwa G (2006). The Linkages Between Trade, Development And Poverty Reduction: The case study of fisheries sub-sector in Tanzania. Economic and Social Resarch Foundation pp. 1-19.
|
|
|
|
|
LVFO (2009). The fisheries management plan for Lake Victoria 2009-2014.
|
|
|
|
|
Medard M, van Dijk H, Hebinck P (2019). Competing for kayabo: gendered struggles for fish and livelihood on the shore of Lake Victoria. Maritime Studies 18:321-333.
Crossref
|
|
|
|
|
Nguyen TL, Dalsgaard A, Phung DC, Mara D (2007). Microbiological quality of fish grown in wastewater-fed and non-wastewater-fed fish ponds in Hanoi, Vietnam: influence of hygiene practices in local retail markets. Journal of Water Health 5(2):209-218.
Crossref
|
|
|
|
|
Norman GM (1999). Principles of Food Sanitation; Fourth Edition. Aspen Publishers, Inc. 253 p.
|
|
|
|
|
Okonkwo TM, Obanu ZA, Oludusin AO (1993). The safety traditionally smoked fish products with respect to Staphylococcus aureus, Escherichia coli and Aspergillus flavus. Proceeding of FAO expert Consultation on Fish Technology in Africa, Accra, Ghana, 22-25 October, 1991. pp. 210-227.
|
|
|
|
|
Olale E, Henson S, Cranfield J (2010). Determinants of income diversification among fishing communities in Western Kenya. Selected paper prepared for presentation at the Agricultural and applied economics association 2010. Joint Annual Meeting, Denver, Colorado 31 p.
|
|
|
|
|
Omwega RN, Abila R, Lwenya C (2006). Fishing and poverty levels around Lake Victoria (Kenya). Proceedings of the 11th World Lakes Conference, Lake Victoria pp. 193-199.
|
|
|
|
|
Quang NH (2005). Guidelines for Handling and Preservation of Fresh Fish for Further Processing in Vietnam. United Nations Universities Fisheries Programme 57 p.
|
|
|
|
|
Reij MW, Aantrekker ED (2004). Recontamination as a source of pathogens in processed foods. International Journal of Food Microbiology 91(1):1-11.
Crossref
|
|
|
|
|
Teklemariam AD, Tessema F, Abayneh T (2015). Review on evaluation of safety of fish and fish products. International Journal of Fisheries and Aquatic Studies 3(2):111-117.
|
|
|
|
|
TFDCA (2003). Tanzania Food, Drugs and Cosmetics Act. Available at:
View
|
|
|
|
|
United Republic of Tanzania (URT) (2013). Fisheries Sector Development Programme: Fisheries sector Development Programme. Livestock and Fisheries Development pp. 25-27.
|
|
|
|
|
US-FDA BAM (2001). Manual: Bacteriological Analytical Manual. Available at:
View
|
|
|
|
|
Wijnker JJ, Koop G, Lipman LJA (2006). Antimicrobial properties of salt (NaCl) used for the preservation of natural casings. Food Microbiology 23:657-662.
Crossref
|
|