ABSTRACT
Guava and jackfruit are popular fruits in East Africa. With consideration of the high post-harvest losses of these two fruits and only seasonal availability, this study aimed to produce nutrient rich fruit-based snacks to decrease this problem and make use of surplus fruits during on-season. Given the nutritional situation in East Africa, these products were also developed to have a high content of desired nutrients. Next to either guava or jackfruit also mango, different nuts and lemon juice was partly added. Processing methods included cooking and drying, which are suitable for local households and small processing groups. Chemical analyses were implemented to determine nutrient contents before cooking, after cooking and after drying. Major results included that bars with guava and lemon juice contained the highest content of ascorbic acid, 81.19 and 48.18 mg/100 g FM before and after cooking, respectively; jackfruit-based samples without lemon juice after drying contained more phenolic content than guava-based samples; fruit bars with lemon juice had higher acidity; samples of guava contained more β-carotene than jackfruit. In conclusion, the fruit-nut-bars can provide a good option to process surplus fruits and provide essential nutrients to the local population in East Africa.
Key words: Guava, jackfruit, fruit-nut-bars, East Africa.
Guava (Psidium guajava) and jackfruit (Artocarpus heterophyllus) are naturalized in tropical and subtropical areas around the world (Flores et al., 2015). Both of them have high contents of several nutrients that are essential to human beings, such as ascorbic acid and potassium (Lukmanji et al., 2008). However, in East Africa, these fruits have usually high post-harvest-losses which decrease the benefit of local people (Omayio et al., 2019). At the same time, lacking nutrients in the diet, especially micronutrients, also known as hidden hunger, is a major problem for many people in sub-Saharan Africa (Tulchinsky and Varavikova, 2009). On the other hand, also the overconsumption of certain nutrients is of a problem and, for example, diabetes is also widespread among African people (IDF, 2019). The share of adults aged 20-79 years with diabetes is 3.9% of the total population in the IDF Africa Region with an increasing trend (FAO et al., 2020).
There are different types of guava products, such as jam, jelly and juice (Leite et al., 2006). There are also studies that investigated the nutrient content of guava together with one other fruit such as orange (Srivastava et al., 2019), papaya (Bisen and Ruchi, 2020) and mango (Sucheta et al., 2017). Products made from the ripe jackfruit already investigated are jackfruit powder (Ahiduzzaman, 2016), jackfruit chips (Yi et al., 2016), jelly, syrup and jams (Mondal, 2013).
This study also focuses on processing options in order to extend the shelf-life of guava and jackfruit which were selected as target fruits by the overall project. One aim is, however, to increase different kind of nutrients by mixing the target fruits with a combination of other fruits and nuts. The form of products in this study was chosen to be fruit-nut-bars, which can be easily carried and should be accepted, especially by children and adolescents similar to dried-fruit-slices that are already available in East Africa (Omayio et al. 2019). However, the nutrient content of fruit-nut-bars which combine guava or jackfruit with other fruits and nuts still lack studies in East Africa.
Given that diabetes is on the rise in East Africa (IDF, 2019), sugar content in this study is considered in particular and there is no extra sugar added to the products. In addition, with the consideration of chemical residuals, there were also no extra chemical preservatives, which, however, can lead to a problem of food safety and reduced long-time storage. Ascorbic acid and β-carotene as well as mineral content inside the fresh fruits were also considered in addition to the degree of color change after drying.
Cooking was chosen because it is a popular food processing method in East Africa and it helps to reduce microbiological contamination (Njoroge et al., 2015). Grinding was applied to make the fruit-nut-mixture to be as uniform as possible. Drying with oven was used to decrease the water content in samples, because bacteria would spoil foods with high moisture (Ponte et al., 1993). If solar drying is not possible, electric power is needed for the oven, which creates some costs and environmental issues (Bieber et al., 2018).
The objectives of this study were as follows:
1) To develop healthy fruit-nut-bars with high essential nutrient content, low sugar content and with potential for a long shelf life.
2) To develop fruit-nut-bars with crispy texture and high acceptability in East Africa.
This study was carried out in the framework of a large study on “Fruit and vegetables for all seasons” (FruVaSe) with partners in East Africa and Germany.
Although the FruVaSe project and most of its components take place in East Africa, the fruit-nut-bar development and analysis took place at the University of Goettingen in Germany. The fresh guavas and jackfruits required for product development and testing were bought from a supermarket (KIM) in Hannover, Germany, and an online shop (Tropenkost) in Frankfurt, respectively. The origin country of both fruits was Thailand. Because of limited availability, in this study, only the white-fleshed types of guava could be studied. Mangoes and bananas were bought from a local supermarket (Rewe) in Goettingen, Germany, origin country was Spain. Fruits were stored at 4ºC before being processed.
At the beginning, cashew nuts were decided to be used, because cashew trees also grow in East Africa like Tanzania and Kenya (McLaughlin et al., 2018). It is also a good choice for adding more minerals and using local materials. However, in Uganda, cashew nuts are mainly grown in northern and eastern Uganda; most of them have to be bought from other countries (Wanyama et al., 2017). Consequently, some bars were made by using peanuts to replace cashew nuts. Both nuts were bought in local supermarkets in Goettingen and the origin was Egypt for peanuts and Vietnam for cashew nuts.
In order to increase the flavor, desiccated coconut (Renuka Agri Organics Ltd, origin country Sri Lanka) was also added. According to the results of Okafor and Ugwu (2014), when compared with snacks without coconut, those that contained coconut gained higher acceptability in a study in Nigeria.
Preparation of fruit-nut-bars
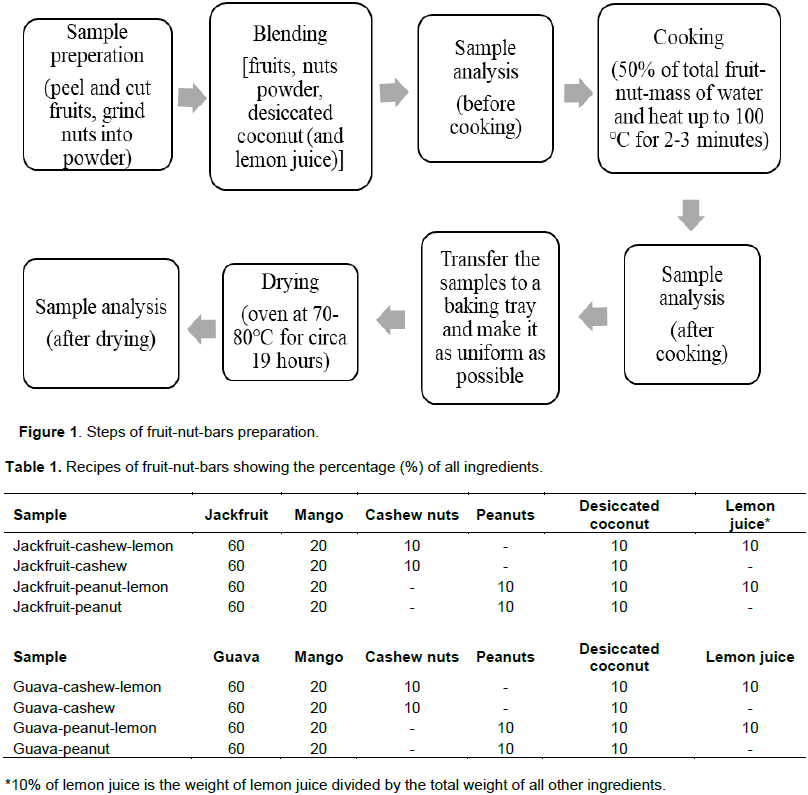
The routine of making fruit-nut-bars can be divided into preparing, blending, cooking and drying. During the process of cooking, 50% of total fruit-nut-mass of water was added, in order to prevent the samples from sticking to the pot. Specific processes are shown in Figure 1. The final products of this study can be divided into guava-bar with mango or jackfruit-bar with mango; the choice of nuts can be divided into peanuts and cashew nuts; lemon juice is another alternative. Consequently, there are eight final different recipes (Table 1 and Figure 2).
The eight final recipes were chosen after the sensory test. The concentration of lemon juice increased after the sensory test in these final recipes; banana, which was an ingredient in the first set of recipes, was not chosen to be an ingredient of the final recipes; peanuts were added as an alternative of cashew nuts.
Sensory test
Eleven participants from Uganda, Kenya and Tanzania tasted six different fruit-nut-bars with different ingredients (Table 2) at the beginning of this study in order to refine the recipes. This sensory test also included a questionnaire (Appendix Table 1). The questionnaires were given to each participant before the samples were served. Each sample was put in a bowl and served separately to each person. Recipes were not revealed until all questionnaires were filled.
Instrumental methods
Water content and total soluble solids
To determine the water content, about 10 g of the samples were placed in a petri dish and the total weight was determined, and then dried for 19 hours at 60ºC and 4 h at 105ºC. Afterwards the total weight was determined again, and the water content was calculated (Rutter and Slatyer, 1968).
The method of ºBrix was used to determine the content of total soluble solids (Ranganna, 1976). 1 g of the sample after drying were weighed for three replicates, 9 ml water was added afterwards. The samples were mixed (Reamix 2789 Vortex Mixer, MTC, Hamburg) and shaken (shaker Swip, Edmund Bühler) for 1 h, centrifuged for 5000 rpm for 5 min (Centrifuge 5804 R, Eppendorf, Hamburg), then Pasteur pipettes were used to place few drops of the supernatant on a refractometer (handheld refractometer, A. Krüss Optronic GmbH, Hamburg) to obtain the value of °Brix.
Mineral content
The mineral content was determined as described by Koch et al. (2019) with slight modification. In brief, about 100 mg of milled samples were weighed in a Teflon vessel and digested with 4 ml of 65% (v/v) nitric acid and 2 ml of 30% (v/v) hydrogen peroxide in a microwave (Ethos 660; MWT AG, Heerbrugg, Switzerland) for 75 min at 200ºC and 40 bar. Afterwards, the samples were filled up to 25 ml with distilled water. Before samples were measured, 2 ml solution of samples were taken out and 8 ml distilled water was added for dilution. The concentrations of macro- and micronutrients were measured with inductively coupled plasma optical emission spectrometry (Vista-PRO CCD Simultaneous ICP-OES; Varian Inc., Palo Alto, CA).
Titratable acidity and ascorbic acid
Titratable acidity content was determined by titration method (0.1N NaOH solution) according Kanski et al. (2020) with modification.
About 0.2 g of the milled samples after drying was weighed into a beaker and 20 ml water was added together with a magnet. The beaker was put on a magnetic stirrer for 15 min. The solution was titrated with a solution of 0.1 N NaOH to pH 8.1 (pH-titrator Titro line 96, SCHOTT AG, Mainz).
The ascorbic acid content was determined according to Sonntag et al. (2020) with modifications. 5 g of samples and 20 ml of meta-phosphoric acid were mixed with Ultra-Turrax (T18 digital Ultra Turrax, IKA, Staufen) for 2 min. The pulp was transferred into a measuring cylinder, filled up to 50 ml with pure water and filtrated; thereafter, 10 ml of the filtrate was transferred into the Erlenmeyer flask (2-3 times for each sample). Finally, samples were titrated against 2,6-Dichlorophenolindophenol (DIP) solution until a light pink end point was reached and could persist for 15 s, then the used amount of ml of DIP solution could be recorded.
Total phenolic content
The total phenolic content was determined by using Folin and Ciocalteu’s phenol reagent (Folin-C reagent) and photometrical determination (Singleton and Rossi, 1965). About 0.25 g of each sample powder was weighed and 5 ml 80% Ethanol was added in a centrifugation tube. The tube was vortexed and then centrifuged at 5000 rpm for 10 min (Centrifuge 5804 R, Eppendorf, Hamburg). The supernatant was transferred into a 10 ml graduated flask, and then the extraction was repeated. The supernatants were combined together and filled up to 10 ml with 80% Ethanol and were frozen in a szintilation vessel prior analysis. Before measurement, the supernatants were taken out at room temperature for 1 h.
For measurements, the samples were prepared as follows: water 2.4 ml, NaOH 1 ml, sample solution 500 μl and Folin reagent 100 μl. Afterwards they were measured immediately at 735.8 nm with a photometer (HP 8453, Hewlett-Packard, Waldbronn) and the results were expressed in mg gallic acid equivalents (GAE).
β-carotene
The β-carotene content was determined according to Schex et al. (2018). As the samples after drying were difficult to be grinded into powder, only samples before and after cooking were tested. 100 mg of lyophilized powdered samples were placed in a 2 ml reaction tube together with 600 μl Methanol (MeOH)/ Tetrahydrofuran (THF) (1:1, v/v). The solution was mixed in a thermomixer (ThermoMixer C, Eppendorf AG, Hamburg) for 10 min at 1400 rpm and centrifuged at 8000 rpm for 5 min (Centrifuge 5804 R, Eppendorf, Hamburg). Afterwards 500 μl supernatant was transferred into another 2 ml reaction tube. 500 μl MeOH/THF (1:1, v/v) was added in the old tube and the extraction was repeated twice. The supernatants (1500 μl) were combined together and evaporated to dryness in a rotational vacuum concentrator (RVC 2-25 CDplus, Martin Christ Gefriertrocknungsanlagen GmbH, Osterode am Harz) for almost 5 h at 20°C. Then the samples were frozen at -80°C until measurement. 250 μl Methyl tert-butyl ether (MTBE) were added to the samples to re-dissolve the residue and vortexed (Mini Vortexer, Heathrow Scientific, and Vernon Hills). Then, 250 μl MeOH were added and vortexed. Afterwards, the samples were dissolved by using a thermomixer (ThermonMixer C, Eppendorf AG, Hamburg) for 5 min at 800 rpm. Then the solution was filtered through syringe-filter with pore size of 0.2 μm and almost 400 μl filtered solution was transferred into a HPLC vial with insert and measured by HPLC (System “Prominence”, Schimadzu). The instrument settings for measurement are shown in Table 3 (Schex et al., 2018).

Color
Color was determined using the machine MINOLTA Chroma Meter - CR310 (Konica Minolta, Inc., Marunouchi, Japan) for samples before and after cooking and by MINOLTA - CR400 (Konica Minolta, Inc., Marunouchi, Japan) for samples after drying to obtain L (brightness), a*(variation from green to red) and b* (variation from blue to yellow) values (Itten, 1997).
Textural profile analysis
The texture properties determined were the hardness and crispness of the fruit-nut-bars. They were measured in terms of the maximum peak force and number of peaks during the first compression cycle with the texture analyzer (‘Stable Micro Systems’, Winopal, Germany) according to Yadav and Bhatnagar (2017) with the following settings:
Pre-Test Speed: 2 mm/s; Test Speed: 1 mm/sec; Post-Test Speed: 10 mm/s; Force: 10 g; Trigger Force: 10 g; Probe: P/5; 5 mm Dia Cylinder (Figure 3).
Depending on the thickness and breakage of fruit-nut-bars, the parameter of distance was set differently. It was the peak distance reached by the probe, that was deep enough to completely penetrate the bars, but did not touch the platform on which the samples were placed.
Data analysis
Data analysis was mainly carried out with Excel, where the mean values were calculated, the standard deviation as well as the formation of bar graphs was undertaken. The significant difference (POSTHOC Tukey HSD) was calculated using PSPP version GNU pspp 1.4.1-g79ad47.
The following results are for eight final fruit-nut-bars, the recipes are shown in Table 1. The names of these fruit-nut-bars show the varying ingredients (such as Guava- Cashew-Lemon). Ingredients, which were present in all eight fruit-nut-bars, namely mango and desiccated coconut, will not be mentioned explicitly.
Sensory test
Eleven participants that were project partners from Kenya, Tanzania and Uganda took part in a sensory test by tasting six different preliminary fruit-nut-bars and answering a questionnaire. Average values of each attribute were calculated (Figure 4). As the radar graph shows, Guava-Mango and Jackfruit-Mango-Lemon gained the highest overall points; desiccated coconut was an ingredient in both recipes. Jackfruit-Mango-Lemon showed almost in all attributes the highest points except aroma. As for color and consistency, Guava-Banana and Guava-Banana-Lemon achieved the least points, respectively. For the other three attributes (mouthfeel, taste and aroma), Jackfruit-Peanut-Lemon (without desiccated coconut) showed the least points.
Water content, total soluble solids, titratable acidity and color
Table 4 shows the results of water content, total titratable acidity and color of guava- and jackfruit-based snacks after drying. Table 5 shows the average contents of total soluble solids of guava- and jackfruit-based snacks before and after cooking, and after drying. Only one test of water content after drying was done, so there were no average results or standard deviations. Almost all samples showed water content less than 10% because of drying. Jackfruit-based samples showed higher results of total soluble solids than guava-based samples. Guava-Peanut-Lemon bars had the lowest content among eight samples. Except for Jackfruit-Cashew-Lemon samples, other snacks with lemon juice all showed higher values of titratable acidity. For color test, the data of ‘a’ (variation from green to red) were very low for after drying and so for final products only the data of ‘L’ (brightness) and ‘b’ (variation from blue to yellow) were recorded. In terms of brightness, the bottom side of products showed higher values than the top side (brighter); jackfruit-based samples were significantly brighter than guava-based samples (P<0.05). Within samples with the same basic fruit (guava or jackfruit), there was no significant brightness difference (except the bottom side of Jackfruit-Cashew samples). For the top side of jackfruit-based samples, samples with lemon juice were brighter and had more yellowness, which was also the same tendency for the bottom side of all samples.
Ascorbic acid, total phenolic content and β-carotene
Figure 5 and 6 show the results of ascorbic acid, namely the average contents in two stages, before and after cooking. As a result of drying, the water content in the samples was very low, so it was not possible to determine ascorbic acid content in them. The cooking process resulted in a significant decrease in ascorbic acid content, with guava-based samples showing higher values than jackfruit samples.
Figures 7 and 8 illustrate the average results of total phenolic content of guava- and jackfruit-based samples in three stages (before cooking, after cooking and after drying). Lemon juice led to a result of higher values of total phenolic content for all samples before cooking. In jackfruit-based samples with lemon juice total phenolic content decreased from before cooking to after drying, while samples without lemon juice showed the opposite results.
As in pre-test, β-carotene contents of dried samples were hardly measurable with HPLC, for final tests, only samples before and after cooking were measured (
Table 6). Guava-based snacks showed higher values than jackfruit, Guava-Cashew samples before cooking and Guava-Peanut samples after cooking showed the highest value. Jackfruit-based snacks with lemon juice after cooking had higher β-carotene content.
Mineral content
The mineral contents of the fruit-nut-bars are shown in Tables 7 and 8. There are nine minerals in fruit-nut-bars, namely K, P, S, Mg, Ca, Na, Cu, Fe, Zn and Mn. In general, jackfruit-based samples had a little higher mineral content than guava-based samples. Except for K, Mg and Mn in specific guava-based samples, the mineral content among other samples were not significantly different (P<0.05).
Textural profile analysis
Textural properties of guava- and jackfruit-based products after drying are shown in Table 9. For jackfruit-based samples, there were significant differences (P < 0.05) in the peak distance reached by the probe due to the thickness of the samples. The force to break (max Peak Force) each of the four products was not significantly different (P < 0.05). Appendix Figures 1 and
2 show the curves of hardness and crispness parameters of the investigated samples.
The availability of fruits during off-season in order to increase fruit consumption is crucial for balanced and healthy diets. One solution to bridge seasonal gaps can be the processing of fruits into dried fruit-nut-bars which was tested in this study. Through physical and chemical analyses of different fruit-nut-bars combinations based on either jackfruit or guava, we have found partly very different and also promising results in terms of nutrient content.
Sensory test
The aim of this preliminary test at the beginning of the product development was to find out which fruit combinations might most closely match the sensory expectations of the East African consumers and which might not. The sensory test used in this study can be defined as affective testing, attempting to quantify the degree of liking or disliking of a product (Stone et al., 2012). On average, for both guava- and jackfruit-based bars, recipes with mango were relatively more popular than recipes with banana. As a result of this sensory test, banana was no longer used in the final recipes, whereas fruit bars with desiccated coconut were also characterized by a higher acceptability, which is the reason that all final recipes contained desiccated coconut. Higher concentration of lemon juice in jackfruit-based bars gained more acceptability than guava-based bars. However, for guava-based bars, recipes without lemon juice were more popular than those with lemon juice. So in final recipes, lemon juice was added as an alternative choice.
Jafari et al. (2016) scored sensory properties of dried kiwifruit samples, which were dried by two different drying methods, oven drying and refractance window (RW) drying, respectively. Results showed that oven dried samples gained less acceptibility than refractance window dried ones, and the main reason was the changed organoleptic properties. RW drying was also found to preserve more nutrients in fruits and vegetables and to be favourable in terms of color and texture as compared to other drying techniques (Shende and Datta, 2019). When taken into practice, it should be considered to dry products from this study by RW drying when produced in East Africa as it is already tested by other projects in Uganda and Kenya (icipe, n.d.).
Water content, total soluble solids, titratable acidity and color
Samples after cooking were put into the oven until the products became totally dry, so the water content of the dried products was around 10%, which is highly important for the microbiological safety (Samotyja, 2015). Drying time of 19 h was quite long and would need a considerable amount of energy. In order to reduce the drying time, less water than the 50% of fruit-nut-mass which was added during the cooking process could be admixed.
Total soluble solids are determined by a refractometric index of the proportion (%) of dissolved solids in a solution, which not only indicates the content of sugars (sucrose and hexoses; 65%), but also acids (citrate and malate; 13%) and other compounds (polyphenolics, amino acids, soluble pectins, ascorbic acid and minerals) in the product (Balibrea et al., 2006; Kader, 2008). Results showed that jackfruit-based samples had more total soluble solids than guava, which according to USDA (2019, 2020), was a reliable result. In general, dried samples had low concentrations of total soluble solids (ranged from 4.0 ºBrix to 4.6 ºBrix for different recipes), when compared with the results of dried guava-orange bars (73-81ºBrix) (Srivastava et al., 2019) and guava-papaya fruit bars (30-38 ºBrix) in the research of Bisen and Ruchi (2020). These low-sugar snacks would not aggravate the diabetes situation in East Africa; however, it could possibly influence the acceptability of consumers (Kader, 2008).
Acids influence the taste of the product (Stevens, 1972) in the way that an acid addition can minimize sweetness (Stampanoni, 1993). Acid addition and the resulting pH reduction can extend the shelf-life of the product (Ramachandran et al., 2017). Furthermore, increased ascorbic acid content may have a protective effect against discoloration (Martí et al., 2002), which is important for product development. In the present study samples with lemon juice had higher acidity content than without, as expected. Titratable acidity content in dried Guava-based samples with lemon juice was similar to the result of pure fresh guava pulp (0.704±0.09%) in the study of Srivastava et al. (2019). In dried Guava-based samples without lemon juice they also reported a similar result of a 50%:50% guava-orange bar (0.38±0.006%) (Srivastava et al., 2019). Interestingly, dried Jackfruit-Cashew samples had a higher titratable acidity content (0.705±0.02%) than dried Jackfruit-Cashew-Lemon samples (0.63±0.03%), and both results were higher than in the other two dried Jackfruit-based bars. The reason may be due to the difference of ripeness of mangos that were used in different recipes. Results measured in this study were quite similar to the results of the top portion of the fresh jackfruit in day five after harvesting (0.61±0.14%) (Ong et al., 2006). All in all, contents of titratable acidity in our fruit-nut-bars showed quite similar results when comparing with other studies, not only for mixed pulp and fresh fruit, but also for dried guava-orange bars. Titratable acidity of all dried Guava-based samples and two Jackfruit-based samples without lemon juice increased, comparing to samples before cooking, which showed the same tendency recorded by Toor and Savage (2006) and Khazaei et al. (2008). Increased titratable acidity content could have positive effects on prolonging shelf life (Zomo et al., 2015)
As for color testing, according to Rahman et al. (2020), the darker browning on the top side of the fruit-nut-bars can result from non-enzymatic ascorbic acid oxidation and enzymatic oxidation of polyphenols. In this study, products before drying were much brighter than dried final products, which lost the yellow-orange color. The reason for this is the degradation of β-carotene, when the temperature is higher than 22°C (Kläui and Bauernfeind, 1981; Krokida and Maroulis, 1998). In the final products, the bottom side was brighter than the top side, because the bottom side was protected by the baking mat from direct heating. This uneven discoloration could possibly affect consumer acceptance, the dark brown color at the top side of products may not be appealing to them. In further studies, more attention should be paid to the protection of the product surface.
Ascorbic acid, total phenolic content and β-carotene
In this study, the ascorbic acid content of the Guava- Cashew-Lemon bars before cooking was 81.19±0.37 mg/100 g FM, which was similar to the value of 60:40% guava-orange bar reported by Srivastava et al. (2019). However, for Guava-Cashew-Lemon and Guava-Cashew samples after cooking, the results were less than 50 mg/100 g FM, similar to the result of 40:60% guava-orange fruit bar of Srivastava et al. (2019). This implies that the effect of cooking on the ascorbic acid content is similar to the effect of reducing the share of guava. Vice versa, an increased ratio of guava may lead to an increased content of ascorbic acid. The jackfruit-based bars showed much lower contents than guava-based bars. According to Shwetha and Ranganna (2016), different genotypes of jackfruit could also result in different levels of ascorbic acid, which could range from 3.57 to 5.00 mg/100 g FM. Comparing results of products in this study, lemon juice led to relatively higher ascorbic acid content.
Cooking led to nearly 30% loss of ascorbic acid in guava-based bars, compared with the value before cooking. Afterwards samples were dried under 70ºC to 90ºC for 23 to 19 h, respectively. According to Siow and Hui (2013), ascorbic acid content in guava slices dried by convection for 9 hours at 40ºC decreased 27% compared to fresh fruits. Assuming that in this study ascorbic acid content in guava-based bars would decrease by 54% during the drying process; nearly 20 mg/100 g FM ascorbic acid would still be contained in the final samples. This could be beneficial to children as well as other population groups in East Africa when comparing with the recommended daily allowance. In order to avoid some diseases such as cardiovascular risks and cataract, 110 mg ascorbic acid per day for an adult is recommended (Fain, 2004). For teenagers at the age of 9-13 years old, the recommended dietary allowances (RDAs) of ascorbic acid are 45 mg per day (Institute of Medicine, 1998). Thus, a guava-nut-bar of 100 g could provide about half of the daily requirement of vitamin C for 9-13 years old children.
Phenolic contents in guava samples with and without lemon juice before cooking showed no significant differences. The lowest result of guava-based-bars in this study (550.4±9.9 mg GAE/100 g FM) was higher than the results in the experiment of Patel et al. (2016), although in the current study only white fleshed guava was used. As Chiveu et al. (2019) has tested, phenolic content for red fleshed guava samples were higher than white fleshed ones. Consequently, products produced by red- fleshed guava in East Africa may contain more phenolics than products in this study, which is desirable as phenolic secondary metabolites are, attributed protective effects against different non-communicable diseases (Crozier et al., 2008). Jackfruit-based samples in this study showed higher values than the freeze-dried jackfruit chips measured by Yi et al. (2016) and jackfruit pulp measured by Shafiq et al. (2017). Overall, dried products of this study showed higher phenolic content than dried single fresh fruit (guava and jackfruit) in other studies. One reason may be the use of different cultivars and the addition of mango with high phenolic content. In the experiment of Ongphimai et al. (2013), mango showed a result of 6646 mg/100 g DM of insoluble phenolic acids and 37 mg/100 g DM of soluble phenolic acids. In general, high phenolic content of products can have protective effects against diabetes, hypertension and cardiovascular disease (Liu et al., 2008; Sun et al., 2002; Visioli and Davalos, 2011; Yi et al., 2005).
According to the results of β-carotene, it can be seen that in samples of guava, almost all samples before cooking had higher content than samples after cooking, which can be attributed to the heat sensitivity of β-carotene. The contents of β-carotene in guava-based samples before drying were less than in the studies of Nwaichi et al. (2015) (0.38 mg/100 g), Leiton-Ramírez et al. (2020) (0.85 mg/100 g dry basis in fresh pink fruits), and Nora et al. (2014) (0.51 mg/100 g dry fruit in red guava). Since β-carotene is lipid-soluble (Palan et al., 1994), peanuts and cashew nuts increased the fat content, which may lead to loss of β-carotene during measurement. After the 5-h-evaporation, there was still lipid inside the reaction tube that was not completely dissolved in MeOH/THF (1:1, v/v). As with the jackfruit samples, the samples with lemon juice in the same processing step showed a higher content of β-carotene than without, which can be explained by the protection effect caused by ascorbic acid, since β-carotene is sensitive to oxygen (Goldman et al., 1983). Result from Ahiduzzaman (2016) was lower than the most values from this study (except Jackfruit-Peanut samples), which might be due to the high content of β-carotene in mango (Godoy and Rodriguez-Amaya, 1989; Mercadante et al., 1997). An average content of 1105 µg of β-carotene per 100 g of ripe mango is given in the food composition table of Kenya (FAO/GOK, 2018). As mango is also highly perishable and seasonal, processing in combination with our key fruits would make a further contribution to prevent food losses and at the same time enhance the nutritional content of the new products.
According to the Institute of Medicine in the USA (2001), β-carotene is one kind of provitamin A, which must be transferred into retinol after absorption in the small intestine. The Recommended Dietary Allowance (RDA) of vitamin A and preformed vitamin A depicted as the form of retinol activity equivalents (RAE) per day, is 445 and 420 µg RAE per day for boys and girls, respectively, 9-13 years old (Institute of Medicine, 2011). Besides, the retinol activity equivalency (μg RAE) ratio for β-carotene from plant sourced food is estimated to be 12:1 (Debelo et al., 2017), which means that every 100 g of the fruit-nut-bars could provide between 3.3 and 20 µg RAE. Overall, products in this study can provide more β-carotene than single fresh guava or jackfruit but less than fresh mango, which can still be regarded as a reasonably good source of β-carotene for 9-13 years old children.
Mineral content
The fruit-nut-bars contained significantly higher amounts of minerals than single fruits. Taking K as an example, guava-based bars ranged from 934-1034 mg/100 g FW, which was higher than values measured by Chiveu et al. (2019) in freeze dried guava samples and values from USDA (2018) in fresh guava. This is due to the high K content of peanuts and cashew nuts (Settaluri et al., 2012; Rico et al., 2016). Comparing results of jackfruit pulp gained from Ojwang et al. (2018), K was double as high as results of jackfruit-based bars in this study; Zn showed almost the same result. On the contrary, Mg was two times less than results of jackfruit-based bars in this study. The difference between results of other literature and fruit-nut-bars in this study may be due to different fruit species in different countries that were used for the experiment (Abedin et al., 2012).
According to the Institute of Medicine in the USA (2011), for teenagers at the age of 9-13 years, the recommended dietary allowances (RDAs) of Fe and Zn are 8 mg each per day. Products in this study contained Fe and Zn from 4 to 7 mg / 100 g DM. Overall, both guava- and jackfruit-based bars of this study combining fruits and nuts can provide considerable amounts of minerals, which could contribute to a balanced diet of local people in East Africa.
Textural profile analysis
From these results, it is quite obvious that each sample had different texture behaviour, which is connected with the drying condition. Texture tests were performed to examine the maximum chewing force for customers to break these final products (expressed as max Peak Force). When the moisture content was less, the bars were quite easy (less force was needed) to be broken. Conversely, samples with higher moisture content were not as easy to break, which may be due to the less crispness (Yi et al., 2016). However, Vijayanand et al. (2000) showed an opposite result that guava leathers with higher moisture content had lower hardness.
On average, the force for biting through the jackfruit-based bars (max Peak Force) was smaller than that of guava-based bars. According to Yi et al. (2016) using a ball probe (p/0.25S), pear chips showed a result of 32.5 N maximum peak force which was similar to values in the pre-test of guava-based bars in this study (33.86 N). After hot air-drying method, pear chips showed the largest value of 59.9 N maximum peak force (Yi et al., 2016), which was considered as a hard structure. In our investigation, most values of hardness (max Peak Force) were similar or even higher than the study of Vijayanand et al. (2000), namely more than 160 N with 7% equilibrium moisture content (EMC). They reported for guava-bars higher EMC (15.4%) and lower hardness (94.2 N) (Vijayanand et al., 2000). Overall, almost all final samples in this study could be defined as having a very hard structure (except Jackfruit-Peanut-Lemon samples). Moreover, although mouth feel properties are not necessarily related to the force of breakdown, attributes like stickiness and viscosity are still related to it (Stone et al., 2012). So, the value of max Peak Force also showed a relative sticky texture of the final products, which was also seen in the texture test; some products even stuck to the cylinder probe after testing.
Crispness of the fruit-nut-bars was characterized by the number of peaks (Appendix Figures 1 and 2). In the study of Yi et al. (2016), pear chips with more compression peaks had crisper textures. Our results showed the same tendency, namely Jackfruit-Peanut-Lemon samples exhibited more than hundred peaks and had the highest crispness. At the same time, the maximum Peak Force of Jackfruit-Peanut-Lemon samples were the lowest among the investigated samples. Each sample showed a different texture, with almost all guava-based fruit-nut-bars showing smooth curves with few compression peaks, indicating less crispness and a harder texture.
As the acceptance between hard and softer products can vary to a great extent, it is important to understand the preferences of consumers. Bower and Whitten (2000) found a negative correlation between the force of chewing and the acceptance of cereal bars, meaning that increased chewing effort can lead to a decrease of liking. However, different customers have different preferences, and cultural background and age will play an additional role in the acceptance of a product (Köster, 2009). According to the sensory test in this study as tested with eleven East African participants, results showed a relatively moderate point (neither like nor dislike) of the overall acceptability. However, this small number of adult participants cannot be representative of local residents in East Africa, yet, show an acceptability trend. Further analysis of texture acceptability should be done in order to know more about the preference of the local population. The drying time can also be reduced to achieve a relatively higher moisture content (15%), so that the fruit-nut-bars are less hard to the bite, which can lead to better acceptability (Vijayanand et al., 2000).
In this study, eight different fruit-nut-bars based on two main fruits, guava and jackfruit were produced and tested.
The bottom side of products had brighter color than the upper side and bars based on jackfruit showed brighter color than guava, which might have an influence on consumer acceptance. Texture showed quite different results for each piece of bars, which was also influenced by moisture content of the oven during drying procedure and, thus, will vary depending on the production facilities.
Ascorbic acid content increased with the addition of lemon juice and can possibly contribute to a longer shelf-life.
Total soluble solids showed an inverse result, which also indicates that fruit-nut-bars are “low-sugar-snacks”. Total phenolic content showed higher values than single fruits in other literatures, which can provide protective effects against several non-communicable diseases. Mineral contents in 100 g DM of fruit-nut-bars such as Fe and Zn could provide sufficient amounts for teenagers aged 9 to 13 years and to combine fruits and nuts seems to be advantageous. The content of β-carotene in samples before drying was higher than in fruit pulp of single fresh guava or jackfruit, however, data of dried product were not obtained and will be much lower because of the heat sensitivity of β-carotene.
Overall, this study provides a simple method of processing local fruits. It is shown that guava- and jackfruit-based fruit-nut-bars can prolong the shelf life of fruits, so that they can be also consumed during off-season when fresh fruits are either not available or very expensive. The bars provide essential nutrients such as ascorbic acid and mineral elements. When producing guava-based bars with different flesh color, the investigated ingredients may also show different values, for example, higher β-carotene values when pink fleshed guavas are used. In conclusion, these products can provide a nutrient rich and low-sugar snack especially during off-season and contribute to a diverse and balanced diet of local communities in East Africa.
The authors have not declared any conflict of interests.
This study was financially supported by the German Federal Ministry of Food and Agriculture and the German Federal Office of Agriculture and Food (BLE). The technical support by technical staff and PhD students at the Division of Quality of Plant Products, University of Goettingen, is highly acknowledged. They also thank all colleagues from Tanzania, Uganda and Kenya who provided valuable advice on improving the recipes of products in this study.
REFERENCES
|
Abedin MS, Nuruddin MM, Ahmed KU, Hossain A (2012). Nutritive compositions of locally available jackfruit seeds (Artocarpus Heterophyllus) in Bangladesh. International Journal of Biosciences 2(8):1-7.
|
|
|
|
Ahiduzzaman M (2016). Development of nutritious dried powder from jackfruit bulb and development of nutritious dried powder from jackfruit. International Journal of Business, Social and Scientific Research 4(3):189-195.
|
|
|
|
|
Balibrea ME, Cristina MA, Jesús C, María CB, Francisco PA (2006). The high fruit soluble sugar content in wild lycopersicon species and their hybrids with cultivars depends on sucrose import during ripening rather than on sucrose metabolism. Functional Plant Biology 33(3):279-88.
Crossref
|
|
|
|
|
Bieber N, Ker JH, Wang X, Triantafyllidis C, van Dam KH, Koppelaar R, Shah N (2018). Sustainable planning of the energy-water-food nexus using decision making tools. Energy Policy 113 (July 2017):584-607.
Crossref
|
|
|
|
|
Bisen BP, Ruchi V (2020). Standardization of recipes on chemical characteristics and storability of guava and papaya mixed fruit bar. International Journal of Chemical Studies 8(4):824-29.
Crossref
|
|
|
|
|
Bower JA, Whitten R (2000). Sensory Characteristics and consumer liking for cereal bar snack foods. Journal of Sensory Studies 15(3):327-345.
Crossref
|
|
|
|
|
Chiveu J, Naumann M, Kehlenbeck K, Pawelzik E (2019). Variation in fruit chemical and mineral composition of kenyan guava (Psidium guajava L.): Inferences from climatic conditions, and fruit morphological traits. Journal of Applied Botany and Food Quality 92:151-59.
|
|
|
|
|
Crozier A, Jaganath IB, Clifford MN (2009). Dietary phenolics: Chemistry, bioavailability and effects on health. Natural Product Reports 26(8):1001-1043.
Crossref
|
|
|
|
|
Debelo H, Novotny JA, Ferruzzi MG (2017). Vitamin A. Advances in Nutrition 8(6):992-994.
Crossref
|
|
|
|
|
Fain O (2004). Carences en vitamine C [Vitamin C deficiency]. La Revue de Médecine Interne 25(12):872?880.
Crossref
|
|
|
|
|
FAO/GOK (2018). Government of Kenya Food Composition.
|
|
|
|
|
FAO, IFAD, UNICEF, WFP, WHO (2020). The State of Food Security and Nutrition in the World 2020: Transforming Food Systems for Affordable Healthy Diets. IEEE Journal of Selected Topics in Applied Earth Observations and Remote Sensing.
|
|
|
|
|
Flores G, Wu SB, Negrin A, Kennelly EJ (2015). Chemical composition and antioxidant activity of seven cultivars of guava (Psidium guajava) fruits. Food Chemistry 170:327-335
Crossref
|
|
|
|
|
Godoy HT, Rodriguez-Amaya DB (1989). Carotenoid Composition of Commercial Mangoes from Brazil. LWT - Food Science and Technology.
|
|
|
|
|
Goldman M, Horev B, Saguy I (1983). Decolorization of β?carotene in model systems simulating dehydrated foods. Mechanism and kinetic principles. Journal of Food Science 48(3):751-754.
Crossref
|
|
|
|
|
icipe (n.d). Adapting Refractance Window Drying Technology to Produce High Quality Fruit and Vegetable Bioproducts.
View
|
|
|
|
|
Institute of Medicine (1998). Dietary Reference Intakes (DRIs): Vitamins. Washington DC: National Academy Press. Available at:
View
|
|
|
|
|
Institute of Medicine (2011). Dietary Reference Intakes (DRIs): Elements Food and Nutrition Board. Washington DC: National Academy Press. Available at:
View
|
|
|
|
|
Institute of Medicine (2001). Vitamin A. Dietary Reference Intakes for Vitamin A, Vitamin K, Arsenic, Boron, Chromium, Copper, Iodine, Iron, Manganese, Molybdenum, Nickel, Silicon, Vanadium, and Zinc. Washington DC: National Academy Press, 65-126.
|
|
|
|
|
International Diabetes Federation (IDF, 2019). IDF Diabetes Atlas, 9th Edn. Brussels, Belgium. Atlas de La Diabetes de La FID.
|
|
|
|
|
Itten J (1972). The Elements of Color. Leonardo.
|
|
|
|
|
Jafari SM, Azizi D, Mirzaei H, Dehnad D (2016). Comparing Quality Characteristics of Oven-Dried and Refractance Window-Dried Kiwifruits. Journal of Food Processing and Preservation 40(3):362-372.
Crossref
|
|
|
|
|
Kader AA (2008). Flavor quality of fruits and vegetables. Journal of the Science of Food and Agriculture.
Crossref
|
|
|
|
|
Kanski L, Naumann M, Pawelzik E (2020). Flavor-related quality attributes of ripe tomatoes are not significantly affected under two common household conditions. Frontiers in Plant Science 11(May):1-14.
Crossref
|
|
|
|
|
Khazaei J, Chegini G, Bakhshiani M (2008). A novel alternative method for modelling the effect of air dry temperature and slice thickness on quality and drying kinetics of tomato slices: Superposition technique. Drying Technolology 26:759-775.
Crossref
|
|
|
|
|
Kläui H, Bauernfeind JC (1981). Carotenoids as Food Colors. In Carotenoids as Colorants and Vitamin A Precursors.
Crossref
|
|
|
|
|
Koch M, Busse M, Naumann M, Jákli B, Smit I, Cakmak I, Herrmans C, Pawelzik E (2019). Differential effects of varied potassium and magnesium nutrition on production and partitioning of photoassimilates in potato plants. Physiologia Plantarum 166:921-935.
Crossref
|
|
|
|
|
Köster EP (2009). Diversity in the determinants of food choice: A Psychological perspective. Food Quality and Preference 20(2):70-82.
Crossref
|
|
|
|
|
Krokida MK, Maroulis ZB (1998). Kinetics on Color Changes during Drying of Some Fruits and Vegetables. Drying Technology. 16 (3-5): 667-685.
Crossref
|
|
|
|
|
Leite KMSC, Tadiotti AC, Baldochi D, Oliveira OMMF (2006). Partial purification, heat stability and kinetic characterization of the pectin methylesterase from Brazilian guava Plauma cultivars. Food Chemistry 94:565-572.
Crossref
|
|
|
|
|
Leiton-Ramírez YM, Ayala-Aponte A, Ochoa-Martínez CI (2020). Physicochemical properties of guava snacks as affected by drying technology. Processes 8(1):106.
Crossref
|
|
|
|
|
Liu L, Wang Y, Lam KS, Xu A (2008). Moderate wine consumption in the prevention of metabolic syndrome and its related medical complications. Endocrine Metabolic and Immune Disorders Drug Targets 8:89-98.
Crossref
|
|
|
|
|
Lukmanji Z, Hertzmark E, Mlingi N, Assey V (2008). Tanzania food composition tables. Muhimbili University of Allied Science,Tanzania food and nutrition centre and Havard school of public health. Available at:
View
|
|
|
|
|
Martí N, Pérez-Vicente A, García-Viguera C (2002). Influence of Storage Temperature and Ascorbic Acid Addition on Pomegranate Juice. Journal of the Science of Food and Agriculture 82(2):217-221.
Crossref
|
|
|
|
|
McLaughlin J, Crane JH, Balerdi CF (2018). Cashew Apple Fruit Growing the Florida Home Landscape. University of Florida. Hs1127, no. IFAS Extention:1-11.
|
|
|
|
|
Mercadante AZ, Rodriguez-Amaya DB, Britton G (1997). HPLC and Mass Spectrometric Analysis of Carotenoids from Mango. Journal of Agricultural and Food Chemistry 45(1): 120-123.
Crossref
|
|
|
|
|
Mondal C (2013). Product development from jackfruit (Artocarpus heterophyllus) and analysis of nutritional quality of the processed products. IOSR Journal of Agriculture and Veterinary Science 4(1):76-84.
Crossref
|
|
|
|
|
Njoroge EW, Matofari JW, Mulwa RMS, Anyango JO (2015). Effects of blanching time/temperature combination coupled with solar-drying on the nutritional and microbial quality of indigenous leafy vegetables in Kenya. African Journal of Food Science and Technology 06(07).
Crossref
|
|
|
|
|
Nora CD, Müller CDR, de Bona GS, de Oliveira Rios A, Hertz PF, Jablonski A, de Jong EV, Flôres SH (2014). Effect of Processing on the Stability of Bioactive Compounds from Red Guava (Psidium Cattleyanum Sabine) and Guabiju (Myrcianthes Pungens). Journal of Food Composition and Analysis 34(1):18-25.
Crossref
|
|
|
|
|
Nwaichi EO, Chuku LC, Oyibo NJ (2015). Profile of ascorbic acid, beta-carotene and lycopene in guava, tomatoes, honey and red wine. International Journal of Current Microbiology and Applied Sciences 4(2):39-43.
|
|
|
|
|
Ojwang R, Muge E, Mbatia B, Mwanza B, Ogoyi D (2018). Compositional, elemental, phytochemical and antioxidant characterization of jackfruit (Artocarpus heterophyllus) pulps and seeds from selected regions in Kenya and Uganda. European Journal of Medicinal Plants 23(3):1-12.
Crossref
|
|
|
|
|
Okafor GI, Ugwu FC (2014). Production and evaluation of cold extruded and baked ready-to- eat snacks from blends of breadfruit (Treculia africana), cashewnut (Anacardium occidentale) and coconut (Cocos nucifera). Food Science and Quality Management 23:65-77.
|
|
|
|
|
Omayio DG, Abong GO, Okoth MW, Gachuiri CK, Mwang' ombe AW (2019). Current status of guava (Psidium guajava L) production, utilization, processing and preservation in Kenya: A review. Current Agriculture Research Journal.
Crossref
|
|
|
|
|
Ong BT, Nazimah SAH, Osman A, Quek SY, Voon YY, Hashim DM, Chew PM, Kong YW (2006). Chemical and flavour changes in jackfruit (Artocarpus heterophyllus Lam.) cultivar J3 during ripening. Postharvest Biology and Technology 40(3):279-86.
Crossref
|
|
|
|
|
Ongphimai N, Lilitchan S, Aryusuk K, Bumrungpert A, Krisnangkura K (2013). Phenolic acids content and antioxidant capacity of fruit extracts from Thailand. Chiang Mai Journal of Science 40(4):636-42.
|
|
|
|
|
Palan PR, Goldberg GL, Basu J, Runowicz CD, Romney SL (1994). Lipid-soluble antioxidants: β-Carotene and α-tocopherol levels in breast and gynecologic cancers. Gynecologic Oncology 55(1):72-77.
Crossref
|
|
|
|
|
Patel P, Sunkara R, Walker LT, Verghese M (2016). Effect of drying techniques on antioxidant capacity of guava fruit. Food and Nutrition Sciences 07(07):544-54.
Crossref
|
|
|
|
|
Ponte JG, Payne JD, Ingelin ME (1993). The shelf life of bakery foods. Shelf Life of Foods and Beverages (ed G. Charalambous). Elsevier Science Publishers pp.1143-1197.
|
|
|
|
|
Rahman MS, Ahmed J, Ramaswamy HS (2020). Applications of Magnetic Field in Food Preservation. Handbook of Food Preservation.
Crossref
|
|
|
|
|
Ramachandran C, Sudha RR, Lavanya K, Nivetha S, Usha A (2017). Optimization of Shelf Stability of Sugarcane Juice with Natural Preservatives. Journal of Food Processing and Preservation 41(1):1-11.
Crossref
|
|
|
|
|
Ranganna S (1976). Manual of analysis of fruit and vegetable products. McGraw-Hill Publishing Company Ltd., New Delhi, India.
|
|
|
|
|
Rico R, Bulló M, Salas-Salvadó J (2016). Nutritional composition of raw fresh cashew (Anacardium occidentale L.) kernels from different origin. Food Science and Nutrition 4(2):329-338.
Crossref
|
|
|
|
|
Rutter AJ, Slatyer RO (1968). Plant-water relationships. The Journal of Applied Ecology 106(6):478.
Crossref
|
|
|
|
|
Samotyja U (2015). Influence of shelf life labelling on the sensory acceptability of potato snacks. British Food Journal 117(1):222-233.
Crossref
|
|
|
|
|
Schex R, Lieb VM, Jiménez VM, Esquivel P, Schweiggert RM, Carle R, Steingass CB (2018). HPLC-DAD-APCI/ESI-MSn Analysis of Carotenoids and α-Tocopherol in Costa Rican Acrocomia Aculeata Fruits of Varying Maturity Stages. Food Research International 105:645-653.
Crossref
|
|
|
|
|
Settaluri VS, Kandala CKV, Puppala N, Sundaram J (2012). Peanuts and their nutritional aspects - A review. Food and Nutrition Sciences 03(12):1644-1650.
Crossref
|
|
|
|
|
Shafiq M, Mehmood S, Yasmin A, Khan SJ, Khan NH, Ali S (2017). Evaluation of phytochemical, nutritional and antioxidant activity of indigenously grown jackfruit (Artocarpus heterophyllus Lam). Journal of Scientific Research.
|
|
|
|
|
Shende D, Datta AK (2019). Refractance window drying of fruits and vegetables: A review. Journal of the Science of Food and Agriculture 99(4):1449-1456.
Crossref
|
|
|
|
|
Shwetha MS, Ranganna B (2016). Development of squash from Jackfruit (Artocarpus heterophyllus. L). Indian Journal of Science 23(77):26-33.
|
|
|
|
|
Singleton VL, Rossi JA (1965). Colorimetry to total phenolics with phosphomolybdic acid reagents. American Journal of Enology and Viniculture 16:144-158.
|
|
|
|
|
Siow LF, Hui YW (2013). Comparison on the Antioxidant Properties of Fresh and Convection Oven-Dried Guava (Psidium Guajava L.). International Food Research Journal 20(2):639-644.
|
|
|
|
|
Sonntag F, Bunzel D, Kulling SE, Porath I, Pach F, Pawelzik E, Smit I, Naumann M (2020). Effect of Potassium fertilization on the concentration of antioxidants in two cocktail tomato cultivars Journal of Applied Botany and Food Quality 43:34-43.
|
|
|
|
|
Stampanoni CR (1993). Influence of Acid and Sugar Content on Sweetness, Sourness and the Flavour Profile of Beverages and Sherbets. Food Quality and Preference 4(3):169-176.
Crossref
|
|
|
|
|
Stevens MA (1972). Relationships between components contributing to quality variation among tomato lines. Journal of American Society for Horticultural Science 97:70-73.
|
|
|
|
|
Sucheta S, Gehlot R, Siddiqui S (2017). Standardization of Mixed Fruit Cheese from Guava (Psidiumguajava Cv. Hisar Safeda) and Mango (Mangiferaindica Var. Safeda) and Its Quality Evaluation during Storage. Journal of Applied and Natural Science.
Crossref
|
|
|
|
|
Sun AY, Simonyi A, Sun GY (2002). The 'French Paradox' and beyond: neuroprotective effects of polyphenols. Free Radical Biology and Medicine 32:314-318.
Crossref
|
|
|
|
|
Srivastava A, Kohli D, Vishnoi S, Kumar S, Badola R (2019). Quality evaluation of prepared guava-orange fruit bar. International Journal of Chemical Studies 7(4):1574-81.
|
|
|
|
|
Stone H, Bleibaum R, Thomas HA (2012). Chapter 5 - Discrimination Testing. In Sensory Evaluation Practices.
Crossref
|
|
|
|
|
Toor R, Savage G (2006). Effect of semi-drying on the antioxidant components of tomatoes. Food Chemistry 94(1):90-97.
Crossref
|
|
|
|
|
Tulchinsky TH, Varavikova EA (2009). ELSEVIER Science and Technology Books Academic Press Tools All Teaching • PowerPoint Lecture Slides by the Authors on Topics Related to the Chapters • Case Studies • Instructor's Guide.
|
|
|
|
|
USDA (2018). National Nutrient Database for Standard Reference Release Legacy April, 2018 full report (All nutrients) 09139, Guavas, common, raw. National Nutrient Database.
|
|
|
|
|
USDA (2019). FoodData Central for SR Legacy April, 2019, 174687, Jackfruit, raw. Available at:
View
|
|
|
|
|
USDA (2020). FoodData Central for SR Legacy October, 2020, 1102666, Guava, raw. Available at:
View
|
|
|
|
|
Vijayanand P, Yadav AR, Balasubramanyam N, Narasimham P (2000). Storage stability of guava fruit bar prepared using a new process. LWT - Food Science and Technology 33(2):132-37.
Crossref
|
|
|
|
|
Visioli F, Davalos A (2011). Polyphenols and cardiovascular disease: a critical summary of the evidence. Mini Reviews in Medical Chemistry 11:1186-1190.
Crossref
|
|
|
|
|
Wanyama J, Ssegane H, Kisekka I, Komakech AJ, Banadda N, Zziwa A, Ebong TO (2017). Irrigation Development in Uganda: Constraints, Lessons Learned, and Future Perspectives. Journal of Irrigation and Drainage Engineering 143(5):04017003.
Crossref
|
|
|
|
|
Yadav L, Bhatnagar V (2017). Effect of legume supplementation on physical and textural characteristics of ready to eat cereal bars. Asian Journal of Dairy and Food Research (36):246-250.
Crossref
|
|
|
|
|
Yi W, Fischer J, Akoh CC (2005). Study of anticancer activities of muscadine grape phenolics in vitro. Journal of Agricultural and Food Chemistry 53:8804-8812.
Crossref
|
|
|
|
|
Yi J, Zhou L, Bi J, Chen Q, Liu X, Wu X (2016). Influence of pre-drying treatments on physicochemical and organoleptic properties of explosion puff dried jackfruit chips. Journal of Food Science and Technology 53:1120-1129.
|
|
|
|
|
Zomo SA, Ismail SM, Jahan MS, Kabir K, Kabir MH (2015). Chemical properties and shelf life of banana (Musa sapientum L.) as influenced by different postharvest treatments. The Agriculturists 12(2):06-17.
Crossref
|
|