ABSTRACT
The effectiveness of multiple cleaning in place (CIP) procedures was observed from different local breweries in the North East of England. Experiments were also carried out to investigate possible reductions in chemical, water and energy use with regards to CIP, without compromising the effectiveness of the CIP performed. The effectiveness of CIP cycles was quantified using Hygiena’s UltraSnap adenosine triphosphate (ATP) swabs, with a relative light unit (RLU) tolerance of <10-30 indicating a clean vessel. It is recommended that microbreweries use at least a 2% v/v dilution for caustic CIP cleaning cycles (based on a ~32% wt caustic liquor) for 35 min to ensure a thorough clean. High temperatures (40-60°C) did not indicate an improvement in cleanliness levels over ambient temperature water (10-20°C) over the 35 min cycle time. A single pass of 100 L of rinse water is adequate for vessels up to 1200 L to ensure removal of caustic residue and should be followed by a sterilisation stage. These recommendations are based on a final acid sterilisation cycle with 1% v/v dilution of a 5% wt Peracetic Acid (PAA) for at least 10 min. Reductions in the usage of caustic liquor, water and energy (heating) for caustic CIP cycles could yield microbreweries savings of over £1000 annually.
Key words: Cleaning, optimization, microbrewery.
Brewing and beer production have been human activities since the beginning of urbanisation and civilization in the Neolithic period (Meussdoerffer, 2009). Over many years the brewing industry in the United Kingdom (UK) developed until it was dominated by a few large players with a relatively small product range. However, the UK no longer plays host to only a few ‘powerhouse’ brewing companies; as of 2018, there are now almost 2000 breweries in the UK (statista.com, 2020), and according to the Beer and Pub Association these are opening up at the rate of one every other day (Jones, 2015). Many of these are classed as microbreweries; a UK microbrewery is typically considered as a brewery that produces less than 15000 barrels of beer annually (Barron, 1995)*[1UK barrel (bbl) is approximately 163L]*.
It is imperative that all product is made in compliance with the regulations of the Food Standards Agency (FSA), in the UK, ensuring that beer produced is not contaminated and is fit for human consumption. Cleaning Techniques in the brewing industry vary widely, and there is no set procedure that is considered optimal, especially within the microbrewing community. Breweries tend to develop their own in house standard operating procedures with a lot of this handed down by word of mouth. Local microbreweries in the North East of England tend to perform cleaning based on an ‘it has always worked’ principle, without necessarily considering the potential reduction in water or chemical usage, unlike some of the larger breweries. Cleaning as a topic brewers are expected to know about, but too often ‘will not admit to having a small amount of potentially prejudiced knowledge’ (Boulton and Quain, 2006).
In addition to FSA regulations, brewers would be disappointed to find their product contaminated with unwanted microorganisms and compromised in flavour due to lack of brewery hygiene (Davies et al., 2015). Whilst the alcohol produced causes an inhospitable environment for most microorganisms, it is widely known that there are select few that can survive in the environment (Davies et al., 2015). The presence of contaminating microorganisms can affect product yield and beer flavour which, if passed to customers, can impact profitability (Davies et al., 2015; Hill, 2009). For microbreweries, local reputation could also be damaged, a potentially catastrophic outcome. Once the boiling of the wort and the fermentation stages of the brewing process have ended and the vessels drained, there remains on the interior of the vessels and in the heat exchanger microbiological and organic residue. This tends to be a combination of yeast, hops, some bacteria and any other ingredients/adjuncts used in the brewing process (such as fruit). There may also be inorganic residues (scales) from the hard water chemicals and beer stone (calcium oxalate and proteins) which is largely due to reactions between the alkaline cleaners used, hard water minerals and proteins in the beer. To prevent cross contamination from batch to batch, the vessel should thus be cleaned thoroughly.
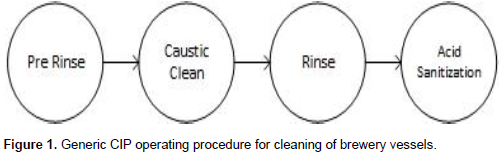
The Standard Operating Procedure (SOP) for most breweries is to use Cleaning in Place (CIP) to clean vessels, a process thought to have been established by the dairy industry to provide adequate cleaning without the need to dismantle equipment (Meyers, 1959). CIP involves the pumping of various liquids through a spray ball to remove debris and microorganisms, and sterilise the equipment for future use. It is advisable that high shear is provided from the spray ball to encourage the displacement of biofilms and debris; a 1.5-3.5 m3/h flow rate per metre of tank circumference is recommended (Boulton and Quain, 2006). It is generally accepted that there are 4 main phases to the cleaning of brewing equipment to ensure a thorough clean: pre-wash rinse, detergent (typically Caustic based) wash, rinse and sterilisation (typically acid) wash (Figure 1). An additional acid cycle and rinse is occasionally performed after the alkali rinse as a de-scaling measure. On some larger scale breweries the acid sterilisation stage is replaced with a sterilisation in place (SIP) procedure, using (sterile) steam to create a sterile environment (Davies et al., 2015). The pre-wash is used to remove loosely bound soil, alkali chemicals to remove organic soils and acids are used to remove inorganic soils, mineral scales (Goode, 2012)and sterilise the vessel, and the final rinse is to remove any alkali or acid from the vessel.
Typical guidance on the concentration of an alkali wash is to use Sodium Hydroxide (NaOH) at 2-5% w/v (Boulton and Quain, 2006), however, there have been drives towards a change to procedures in recent years due to price changes in chemicals. Traditionally, a hot caustic solution of 2-4% w/v was used for cleaning, but with increasing prices, the approach was changed to save costs and 1-2% w/v concentrations of caustic are now advised for the alkali wash for stainless steel vessels (Miller et al., 1960). It is generally accepted that no one material has all the desired qualities of a good detergent, but the detergent of choice is usually a mixture of different chemicals, with the primary chemical being Caustic Soda (NaOH) and possible sequestrant additions ‘to improve emulsification and rinsability’ (Miller et al., 1960).
Heineken NV, a large scale brewery, performs an ambient rinse, a hot caustic wash at 65-70°C, and then an intermediate water disinfection, using a 2% w/v concentration of caustic (Goode, 2012). However, Heineken suggest that lower CIP temperatures and chemical concentrations could be used in the UK to achieve the same level of cleanliness. The potential optimisation of detergent (caustic) use at Heineken was investigated, with results showing that concentrations of NaOH >1% w/v do not improve cleaning results, and therefore it is not cost effective to use higher concentrations than this. It was therefore advised that 1% w/v concentrations should be employed (Atwell et al., 2017), which is equivalent to 3% v/v dilution (Appendix A for conversion). Currently some microbreweries use less than 3% v/v NaOH, therefore there is scope to investigate further potential consequences.
In contrast, a pharmaceutical company will require a higher sterile level of cleanliness, but the CIP methods used may still be useful for the brewing sector. A pre-rinse of approximately 5-6 min is usually sufficient, and then a 1% (w/v) solution of sodium hydroxide at 75-80°C should be circulated for 15-20 min (Chisti and Moo-Young, 1994), supporting the premise that the reduced concentration NaOH solutions can still provide adequate cleaning.
A similar study investigated the CIP regimes of yeast removal post fermentation for large scale breweries. The results found that a visibly clean surface could be achieved using 0.2 and 2% wt caustic based solution at temperatures ranging between 20-70°C (Goode et al., 2010); however, the largest removal of yeast film in the shortest time was recorded at 50°C, which suggested that the current operating procedure of the brewery (2% wt at 70°C) could need reductions of both temperature and chemical concentration (Goode et al., 2010).
A study on two American breweries, with the same SOP, identifies that they perform a soft rinse, a caustic cycle at 130°F (54°C) in half hour bursts, followed by a water rinse and then a sanitizing cycle of peracetic acid for 10 min (Deraney et al., 2015), where the tank is then considered clean. The suggested temperature for this procedure seems lower compared to that of other publications which typically suggest high temperatures of 60+°C. Above 60°C a significant steam and caustic vapour is produced which is unpleasant to work in (a problem with open top non-sealed vessels), which is another incentive to reduce the temperature of caustic cleaning solutions if possible.
However, it may be possible to compensate for the low cleaning temperature by increasing the cleaning agent concentration, and/or the flow speed (Praeckel, 2009), or on a microbrewing scale the temperature may be less important than previously considered. ‘The Handbook of Brewing’ advises the cleaning of fermentation and storage tanks from ‘cold’ to 40°C, but temperatures of 70-90°C for the cleaning of lauter tuns, mash tuns, wort coolers and all pipelines (Praeckel, 2009). This suggests that colder caustic CIP procedures could provide adequate cleaning potential.
It is apparent that there is no single identified best procedure for CIP in the brewing industry: the types of rinse and wash, chemicals used (and concentrations thereof), volumes of liquid, duration and temperature of the washes differ from brewery to brewery. To optimise the effectiveness of the CIP procedures, an optimum (or minimum) combination of temperature, volume of liquid and concentration of chemical needs to be sought. In addition, it will be necessary to define a method for measuring the cleanliness of vessels after the cleaning is done (and therefore compare the effectiveness of the CIP). This is the purpose of this study.
Previously mentioned studies tend to measure cleanliness of fermenters through visual inspections only. This study aims to use a more quantitative measure of cleanliness levels post cleaning to determine the effectiveness of different CIP regimes. To compare the different cleaning procedures in the breweries, a measurement technique for cleanliness was required. According to the FSA, all equipment that touches food must be ‘cleaned effectively and… disinfected frequently’, be kept in ‘good order, repair and condition’, and all chemical additives should be used in ‘accordance with good practice’ (FSA, 2013). Barron (1995)discusses the concept of Hazard Analysis and Critical Control Points (HACCP). The HACCP system was developed by the World Health Organisation and is the standard used throughout the EU Food Industry and is recognised by several legislative bodies (McCrimmon, 2004). The document discusses how beer is generally thought to be a safe beverage, but possible contamination should still be considered to ensure it does not occur. Whilst the document provides information on why beer can be considered safe (e.g., alcohol content and low pH inhibit bacterial growth) there is no information contained within it as to the procedures for CIP to ensure equipment is cleaned. The document advises that brewers should know the influence of temperature on microorganisms, but no further detail is presented. There is also no indication as to the level of cleanliness that should be achieved.
Currently, there is no defined legal quantitative ‘standard’ to which all breweries should comply, and this in turn results in different methods of cleaning practice from brewery to brewery. In addition, there are no guidelines for selection of a cleaning process and ensuring it is operated correctly. It also appears that very little action is taken in the way of regular testing for cleanliness by breweries (Moretti, 2013); this is likely due to the expense of the equipment and testing procedures such that small-scale brewing companies are reluctant to repeatedly test their equipment to see how clean it is. There are multiple ways in which cleanliness can be measured and quantified: for microbiological substances ATP Bioluminescence, the Direct Epifluorescence Filter Technique (DEFT) and Antibody DEFT are just some of the ‘rapid’ methods for testing (Priest and Stewart, 2006), and for the chemical residue of the cleaning products themselves, conductivity and pH tests on the vessel walls and of effluent rinse water can be used (Chisti and Moo-Young, 1994).
The ATP technique measures living organic residual matter that may remain after a surface, device or piece of equipment is cleaned. A simple swab is taken of a surface area, and a handheld scanner provides a reading of Relative Light Units (RLUs). Whilst this method cannot specifically identify what live organic matter is on the tested surface, it provides a fast and easy method of estimating the overall condition of the vessel and plant and can be used as a rapid ‘go/no go hygiene check (Thomas, 2010). The ATP bioluminescence method is in use in many breweries for hygiene monitoring, product quality control and validating CIP performance in real time (Storgårds, 2000; Boulton and Quain, 2006).
Regarding ATP tolerance values for this study, the Hygiena’s ‘EnSURE’ bioluminescence reader was available, using their UltraSnap swabs that can be used on a wet or dry surface. For this study, Hygiena’s recommended tolerance of less than 10-30 RLU was implemented; a reading of <10 RLU indicates an ideally clean surface, and <30 RLU indicates a surface is clean enough for use but further improvements to cleaning could be made. It is also advised that, like the rest of the food and beverage manufacturing industry, brewers use industry accepted critical limits of 10 and 30 RLU (Hygiena, 2014).
CIP procedures used in the brewing industry vary from brewery to brewery, with no stipulated method. Varying temperature, duration and chemical concentration can yield different cleaning results (Goode, 2012). There is a gap in the knowledge of the microbrewing sector with regards to what is considered the ‘optimal’ CIP procedure. Further research will be undertaken into the effectiveness of the different cleaning techniques used in industry, posing the question of whether there is an optimal combination of temperature and concentration of detergent that offers optimal cleanliness.
In this study we compare the different cleaning procedures implemented by ten microbreweries situated on the North East of England and perform further experimental cleaning procedures on industrial sized brew kits in an attempt to determine if they can possibly reduce water, chemical and energy use. These breweries were chosen due to their various sizes within the microbrewing industry, their willingness to take part in the study and they were within reasonable travelling distance. The study focuses more on the cleaning of fermentation vessels, as this stage of the process presents the highest risk of contamination of the wort due to the operating temperature and sugar rich solution providing ideal conditions for bacterial growth. The exact method implemented by each brewery is presented in Table 1; the data has been anonymised and the ten breweries are known by the letters A-J and they range in size from 100 to 3200 L brew kit capacities.
Taking ATP swabs
ATP swabs were used as the primary monitoring tool to determine how clean a vessel was before and after a clean. The Hygiena ‘EnSURE’ monitoring system was used to measure ATP samples, using the Hygiena UltraSnap™ surface swabs (which can be used to test a wet or dry surface). The following procedure, outlined in the EnSURE operational instructions, was used to obtain ATP sample swabs:
(i) For a flat surface, rub the swab over an approximate 4x4 inch area, rotating the swab repeatedly to ensure good coverage.
(ii) For irregular surfaces, such as outlet taps, valves or probes, ensure the area is swabbed thoroughly, rotating and using the same technique each time.
(iii) Return swab to holder, ensuring no contact with any surface.
(iv) Once swab has been taken, take reading within 4 h.
For each of the breweries visited, ATP swabs were taken before and after the full cleaning procedure were completed. Swabs taken prior to cleaning were only to ensure that a vessel was sufficiently ‘dirty’ before cleaning took place (typically providing RLU readings greater than 1000), but these tests are not reported here. After the cleaning SOP was completed, swabs were taken in various locations inside the vessel. The main focuses for swab locations were:
(i) The Krausen Line*;
(ii) Interior side wall of vessel (lower than the Krausen line); and
(iii) Any ‘Other’ difficult to clean areas, such as Welded joints, Valves or Probe Inlets.
*The interface between liquid and vessel where foaming occurs. Fermentation usually leaves a crusty residue in this area due to the typical ‘top fermenting yeasts’ used. Figure 2 gives detail of .the areas inside of the fermenter.
A vessel was considered “clean” if the ATP swab reading was below 10 RLU and considered clean enough to use if less than 30 RLU, but improvements to cleaning techniques could be made. If the RLU was above 30, the vessel was not considered clean. Hence, the tolerance for determining whether a vessel is clean or not, for this study, was <30 RLU, as advised in the Hygiena ‘EnSURE’ operating instructions, however, <10 RLU is more desirable.
Cleaning procedures at external breweries
As part of this research, ten microbreweries in the North East of England were visited to observe their cleaning procedures in the hopes of optimising their cleaning regimes. The breweries that were visited as part of this study each had their own SOP for cleaning their vessels. ATP swabs were taken just after a pre-rinse, prior to cleaning, to ensure areas were sufficiently dirty and after the full CIP cycle to determine cleanliness. Each brewery used the generic SOP for cleaning shown in Figure 1 (with the exception of one that does not practise CIP but hand scrubs each vessel) with variations in Caustic concentration, temperature, duration and rinse water volumes. The actual procedures for each brewery are summarised in Table 1.
In this study, the concentrations of cleaning solutions are reported in % v/v; for example, a 2% v/v caustic solution refers to 2 L of raw caustic liquor per 100 L cleaning solution. Although the branding of caustic liquor varied between some of the breweries, the % w/v concentration recorded on the Material Safety Data Sheets (MSDS) of each liquor used was the same (~32% w/v). Some liquors contained anti-foaming agents, which is not believed to impact the overall cleaning performance of the caustic liquor. Peracetic acid (PAA) is exclusively used for the Acid Sterilization stage, and each brewery purchases PAA at the same raw concentration (5% w/v), regardless of branding. Typical manufacturer advice is to use 5-10 ml PAA per litre of water (0.5-1% v/v dilution) to form the acid sanitizing solution.
Optimising the caustic cycle experiments
To optimise the cleaning procedures, further CIP tests were carried out on two industrial microbrewery kits: one with 450 L fermenters and the other with 900 L fermenters. These tests focused on the caustic cleaning cycle of the CIP regime outlined in Figure 1, to determine the effects of reducing temperature, duration and concentration of solution on the cleaning power of this stage.
The following steps were taken for the experiments performed:
(i) For each experimental run, the volume of rinse water and acid sterilization stage remained constant (a 100 L rinse following the caustic cycle, followed by a 10 min PAA cycle at ambient temperature and 1% v/v dilution).
(ii) The temperature was varied from ambient temperature water (typically 18-21°C depending the time of year) up to 60°C, the recommended maximum temperature for caustic solutions.
(iii) The chemical concentration of the brewery regimes observed were either 2.0% v/v or higher. Hence to determine a lower optimum, concentrations tested ranged from 0.5% v/v to 2.0 % v/v, increasing in steps of 0.5% v/v. The 0.5% increments were used as, typically, the breweries studied used half litre increments for measurements.
(iv) ATP swabs were taken throughout the duration of the caustic cycle (flow was paused momentarily), and the cycle was stopped if the RLU was within tolerance during this phase or until 60 min had passed, whichever came first. According to the breweries visited, as time is a precious commodity for brewers, leaving a tank on CIP for over an hour would be undesirable. Final RLU values stated in this study were taken after the PAA cycle.
Comparative study
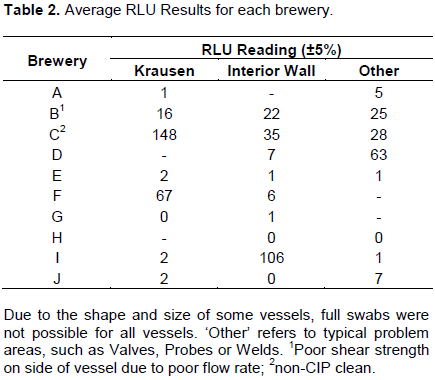
Table 2 shows the results of the average RLU swab tests taken at each site, and for some breweries this will be across several fermenters. Although cleaning regimes vary widely in duration, temperature, water and chemical usage, most breweries were able to meet the 10-30 RLU tolerance.
In general, swabs taken around the Krausen Line and Other areas (e.g. welds, scratches, thermowells, sample points) tended to have a higher RLU post clean, as shown in Table 2; these areas are harder to clean, with larger deposits of biomass in the Krausen and the rough uneven surfaces allowing for increased accumulation of organisms compared to the smooth surface of the interior wall. Particular care and attention to these areas should be taken when cleaning fermentation vessels, to prevent possible accumulation of unwanted organisms that could lead to product contamination.
A notable observation from all breweries observed is the regime employed by Brewery C, who do not have a CIP cycle on their vessels. On two separate cleans of two separate vessels, the RLU swabs did not meet the cleanliness tolerance.
Thus, it is arguably better and more consistent to perform CIP in fermentation vessels than it is to manually scrub them. However, it should be noted that Brewery C has not yet encountered any contamination issues in their products. It was noticed that on some occasions, flow of caustic solution through the spray ball was not very high, and this resulted in poor cleanliness levels (that is, Brewery B’s results). Whilst high flow rates can cause leakage or drips from the top of ‘open top fermenters,’ it is recommended that breweries ensure the spray ball pressure for their CIP regimes is sufficiently high to reach all areas of the fermenter with reasonable shear force. It is believed that the higher the shear force through the spray ball, the better removal of contaminants a CIP regime can provide.
Some of the breweries heat their CIP solution to high temperatures as it is believed to provide better cleaning results by the brewery staff. In contrast, Brewery A and Brewery E’s results show that low temperature and lower caustic concentrations can achieve just as high levels of cleanliness, in comparison to those of Brewery H and Brewery J’s regime and results. Although the brewery cleaning procedures of the study vary, the comparable high levels of cleanliness lead to the hypothesis that some breweries could reduce their chemical, water and/or energy usage, and have little to no impact on their cleanliness procedures. This would lead to long term potential savings for the brewery, but also a reduced environmental impact through use of less resource and reduced chemical loading on wastewater discharge. It was on this basis that further cleaning regime tests were carried out to investigate the impact of lowering chemical dosage, duration and temperature of the caustic cycle.
Further caustic cycle experiments
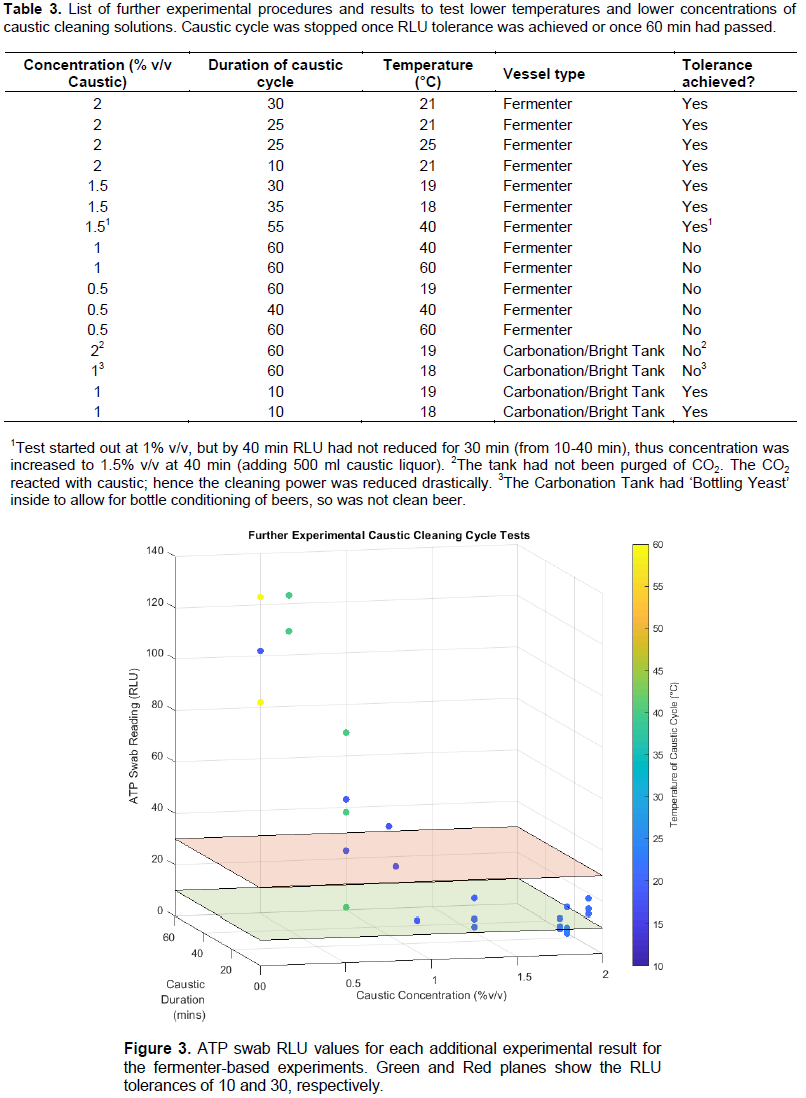
The main focus of the optimisation experiments was on fermenters that had a high amount of yeast, however, a few low concentration tests were performed on Bright Tanks (tanks that typically hold ‘clean beer’ free from yeast and hops, and on occasion can also be Carbonation tanks). The experiments performed are summarised in Table 3, and show whether the experimental setup achieved the cleanliness tolerance or not. Swabs were still taken from multiple locations (Krausen, Interior Wall and ‘Other’ areas) inside the fermenter, and cleanliness was achieved if all three areas swabbed were within tolerance. The measured RLU values from all the fermenter-based experiments are shown graphically on Figure 3, with the RLU tolerances of 10 and 30 shown as the Green and Red planes, respectively.
The results from Table 3 indicate that low concentrations of caustic (1.0% v/v) would be adequate for the cleaning of Bright beer tanks at low temperatures in as little as 10 min, provided that the tank has not had any micro-bacterial additives (such as bottling yeasts) and is purged of CO2 prior to the commencement of cleaning. However, further investigations into the cleaning of Bright tanks should be performed before a formal recommendation to use such a low concentration. As a note of warning for using NaOH to clean carbonation vessels, CO2 and Caustic react together with the resulting reaction causing a vacuum which could lead to vessel collapse (Boulton and Quain, 2006; Manzano et al., 2011) if left fully sealed.
Table 3 and Figure 3 show clearly that, for fermentation vessels of at least 450 L in size, a caustic solution of concentration 1% v/v is inadequate to provide consistent acceptable levels of cleanliness, and a solution of 0.5% v/v cannot reach the tolerance values, regardless of the temperatures or duration of the cycle. It would suffice to argue that the concentrations of 1% v/v and lower of caustic is too weak to ensure the removal of all microbial growth within the vessel. A concentration of 1.5% v/v, however, consistently reaches the tolerance requirements after around 30-40 min of CIP, with 2% v/v reaching tolerance within 20-30 min. At lower concentrations of caustic solutions, increasing the temperature of the solution did not provide an increase in cleaning power, contrary to the expectations of the local brewers.
Optimal caustic cleaning cycle
When examining all the cleaning regimes used as part of this study (industry observed or experimental, Figure 4), it is evident that the most important variable when deciding on a caustic cleaning SOP is the concentration of the caustic solution. Caustic solutions of 0.5% v/v were unable to meet the required tolerances and should not be employed for cleaning of fermentation vessels. Similarly, whilst 1.0% v/v concentrations can achieve the cleanliness tolerance, solutions of this strength are unable to do so consistently, even at warmer temperatures.
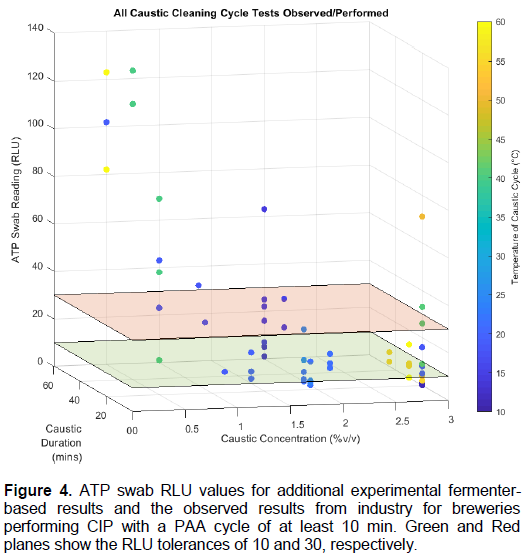
Once a concentration of at least 1.5% v/v has been reached, the results indicate that the overall cleanliness of vessels does not improve with increased temperature; non-heated (ambient) temperature water is perfectly acceptable for cleaning at this concentration. Increasing the concentration beyond 1.5% v/v still provides cleaning results within tolerance but does show more RLU values below the 10 RLU threshold.
Increasing the concentration from 2% to 3% v/v does not appear to provide any significant benefit regarding the cleanliness of vessels, even with the increased temperatures observed in industry. The duration required to ensure that cleaning reaches tolerance does reduce slightly when the concentration is increased from 1.5-3% v/v regardless of temperature as follows: 30-40 min for 1.5% v/v, 25-35 min for 2.0% v/v and 20-30 min for 3.0% v/v.
Based on the results shown in Figure 4, it is sufficient for a brewery to employ a caustic CIP cycle of 1.5% v/v concentration for at least 40 min at ambient temperatures to ensure a thorough clean, providing there is adequate sheer force on the inside of the vessel and the raw liquor is at least 32% w/v. However, to provide a better clean (under the lower RLU tolerance) a caustic solution of 2% w/w for at least 30 min is recommended for fermentation vessels. Although there are a few instances where higher concentrations do not meet tolerance, these could be explained through non-optimal cleaning conditions, such as reduced spray ball shear force or a lack of attention to the manual cleaning required for difficult places to clean, such as sample taps and valves.
Potential caustic savings
Most breweries observed used a 2% v/v dilution of caustic for their cleaning procedures, but some used higher amounts. For a microbrewery, the difference between using 2 and 3% v/v in caustic can result in reasonable savings in chemical usage (e.g., for 100 L, it is 1 L of caustic saved per cleaning cycle). Lower chemical doses also help with compliance of chemical discharge into sewers. At the time of writing, a 25 L Caustic Liquor drum could be purchased for £26.45 ( Murphy and Son LTD, 2018). Assuming a brewery does CIP six times a week (312 cleans annually) and uses 100 L of water per cycle, a 2% v/v dilution would require 26 drums of caustic a year, whereas a 3% v/v would require 39 drums, a difference of £344. However, other caustic brands can cost as much as £37.50 a drum (Niche Solutions, 2020), which would increase savings to £488 annually.
Similarly, providing there is no compromise on the cleanliness of the vessel, reducing the volume of caustic
solution from 200 to 100 L per cycle would half the caustic costs annually; although some of the larger breweries observed used 100 L of caustic solution, it is important that breweries ensure the volume of caustic solution used does not cause pumps to run dry on occasion if reducing the volume.
Potential rinse water savings
It was witnessed that large volumes of rinse water are used in some breweries compared to others, such as the comparison between Brewery D and I, where brewery D uses over 500 L more to rinse vessels of a similar size. Regardless of the volume of caustic used for a rinse, the amount of caustic left in a vessel after draining it is reflective on the size of the vessel; the film left on the vessel walls should be all that remains. Thus, there seems to be little justification for extra water usage.
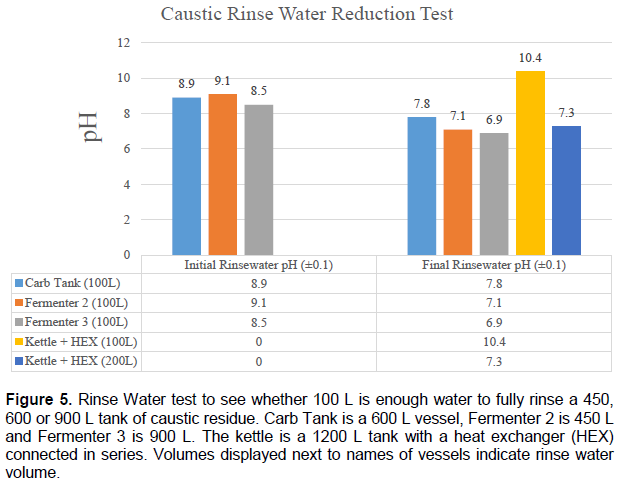
Figure 5 shows the results of a pH test on initial and final samples of rinse water used when rinsing a fermenter, carbonation tank or brew Kettle with a connected heat exchanger. These results indicate that, for at least up to 1200 L vessel, 100 L of rinse water should be adequate to ensure no caustic residue is left inside the vessel, with an additional 100 L required for a heat exchanger.
According to Northumbrian Water, the local water company to the area, the cost of a cubic meter of water (1000 L) is 106.46p (Northumbrian Water, 2020). Again, assuming a brewery performs 312 cleans annually, a 500 L saving of rinse water per clean would result in a cost saving of £166. Breweries must also pay per cubic meter of sewerage, at 77.39p (Northumbrian Water, 2020), resulting in an additional saving of £121 in sewerage charges. Hence a 500L saving in rinse water per clean, based on six cleans a week, could result in a saving of almost £300 per year (£287 per year), as well as a reduced load on local sewerage systems and the environment.
Potential energy savings
Based on the findings of this study, adequate cleaning is provided by ambient temperature caustic solutions just as well as higher temperature solutions. This suggests that there could be no need to heat the cleaning solution, which could present significant energy savings for small business breweries.
As a rough estimate, assuming a 100 L cleaning solution requires heating prior to the addition of caustic, it will require approximately 18.9MJ of energy to heat 100 L from 15-60°C, which is equivalent to 5.25 kWh. Most breweries heat their water using electric immersion heaters. Based on the UK Governments published small business energy rates, electricity cost small businesses 14 p/kWh per quarter in 2019 (UK Government, 2020). As before, assuming 312 annual cleans of a brew kit, eliminating the heating of caustic cleaning solutions could save small breweries up to £230 in electricity costs annually. For breweries using 200 L of heated solution, the savings would double to £460.
Microbreweries must maintain high levels of cleanliness to ensure products are not contaminated, thereby avoiding reputational damage to their brands. It is reassuring that, in general, the microbrewery cleaning practices currently employed across North East England are considered acceptable and vessel cleanliness adheres to the RLU tolerance applied as part of this study. There is no single CIP technique employed by microbreweries in the North East that could be deemed ‘the best.’ Although some will feel peace of mind with SOP above the minimum requirements, based on the observations of this study, some breweries could be over using chemical, water and/or energy resources during cleaning. Thus, there is the clear potential for financial savings and mitigation of environmental impacts.
It is recommended that microbreweries use at a bare minimum 1.5% v/v dilution of caustic solutions during CIP of fermentation vessels for 40 min, though at least 2.0% v/v for 35 min is preferable to ensure a high level of cleanliness. Should a brewery prefer to use 3.0% v/v, then a cleaning duration of 30 min should suffice. This is assuming there is adequate shear force provided by the spray ball on the vessel surfaces. Reducing chemical dosage amounts for some breweries could provide annual savings of up to £300.
Heating the caustic solution does not appear to provide an additional benefit; breweries could use cooler or ambient temperature water without hindrance to cleaning results and achieve savings of over £200 annually in energy costs. At least 100 L of rinse water should be used for vessels up to 1200 L in size, including heat exchangers. Larger vessels were not tested as part of this study, but it is likely that 150 L would suffice for vessels up to 1800 and 200 L for vessels up to 3200 L. Although the cleanliness of vessels is of highest importance, work done as part of this study shows that microbreweries should still consider the potential cost savings of optimising cleaning regimes and that cleanliness levels do not necessarily have to suffer as a result of such optimisation practices. It should also be noted that this is only one measure of cleanliness; non-living organic residues are also important to consider, particularly for craft beers with non-standard additions.
The authors have not declared any conflict of interests.
This work has been funded by Newcastle University. The authors express thanks to all local microbreweries that took part in the study, with special thanks to Tom Meads, Martyn Stockley, Andrew Aitchison and Carl Kennedy for their insight and extended support with additional testing.
REFERENCES
|
Atwell C, Martin E, Montague G, Swuste J, Picksley M (2017). Optimization of cleaning detergent use in brewery fermenter cleaning. Journal of the Institute of Brewing 123(1):70-76.
Crossref
|
|
|
|
Barron FH (1995). HACCP and Microbreweries. Practical Guidelines of Food Safety for Microbreweries, Brewpubs and the Beer Industry.
View
|
|
|
|
|
Boulton C, Quain D (eds.) (2006). "Microbiology" in Brewing Yeast and Fermentation. Oxford, UK, Blackwell Science Ltd, 510-585.
Crossref
|
|
|
|
|
Chisti Y, Moo-Young M (1994). Clean-in-place systems for industrial bioreactors: Design, validation and operation. Journal of Industrial Microbiology 13(4):201-207.
Crossref
|
|
|
|
|
Davies S, Sykes T, Philips M, Hancock J (2015). Hygienic design and Cleaning-In-Place (CIP) systems in breweries" in Brewing Microbiology. Elsevier, pp. 221-239.
Crossref
|
|
|
|
|
Deraney N, Manning J, Morse L, Palmer J (2015). Efficient Brewery Sanitation. Wachusett Brewing Company and Worcester Polytechnic Institute.
View
|
|
|
|
|
Food Standards Agency (FSA) (2013). Food Hygiene - A Guide from Businesses, (FSA/1690/0613).
|
|
|
|
|
Goode KR (2012). Characterising the cleaning behaviour of Brewery Foulants: to minimise the cost of cleaning in place operations. e-theses repository, University of Birmingham Research Archive.
View
|
|
|
|
|
Goode KR, Asteriadou K, Fryer PJ, Picksley M, Robbins PT (2010). Characterising the cleaning mechanisms of yeast and the implications for Cleaning in Place (CIP). Food and Bioproducts Processing 88(4):365-374.
Crossref
|
|
|
|
|
Hill AE (2009). "Microbiological stability of beer" in Beer. Elsevier 163-183.
Crossref
|
|
|
|
|
Hygiena (2014). The Quality Monitoring Tool for Every Brewer's Toolkit. 1-5.
View.
|
|
|
|
|
Jones M (2015). Press release: Britain becomes "brewing powerhouse."
View.
|
|
|
|
|
Murphy and Son LTD (2018). Brewery Hygiene.
View (Accessed February 5, 2018).
|
|
|
|
|
Manzano M, Iacumin L, Vendrames M, Cecchini F, Comi G, Buiatti S (2011). Craft Beer Microflora Identification Before and After a Cleaning Process. Journal of the Institute of Brewing 117(3):343-351.
Crossref
|
|
|
|
|
McCrimmon E (2004). Managing Food Safety in the European Brewing Industry through the Application of HACCP Principles.
View
|
|
|
|
|
Meussdoerffer FG (2009). "A Comprehensive History of Beer Brewing" in HM. (Editor) Eßlinger (ed.), Handbook of Brewing. Weinheim, Germany, Wiley-VCH Verlag GmbH & Co. KGaA, pp. 1-42.
Crossref
|
|
|
|
|
Meyers VE (1959). Recent Developments in Automatic Cleaning of Storage Tanks. Journal of Dairy Science 42(10):1730-1733.
Crossref
|
|
|
|
|
Miller JLHR, Miller JLHR, Comrie AAD (1960). Detergents and Sterilization in Breweries. Journal of the Institute of Brewing 66(2):134-141.
Crossref
|
|
|
|
|
Moretti E (2013). Development of guidelines for microbiological control in microbrewery. Alma Master Studiorum - University of Bologna.
|
|
|
|
|
Niche Solutions (2020). Food & Beverage Brewery Hygiene Products. [online]
View (Accessed May 27, 2020).
|
|
|
|
|
Northumbrian Water (2020). Tariffs and charges. [online]
View (Accessed May 27, 2020).
|
|
|
|
|
Praeckel U (2009). "Cleaning and Disinfecting" in HM (Editor) Eßlinger (ed.), Handbook of Brewing. Weinheim, Germany, Wiley-VCH Verlag GmbH & Co. KGaA, pp. 595-620.
Crossref
|
|
|
|
|
Priest FG, Stewart GG (2006). Handbook of Brewing (Graham G. Stewart and Fergus G. Priest, eds.), CRC Press.
Crossref
|
|
|
|
|
Statista.com (2020). Number of active beer microbreweries in the United Kingdom (UK) from 2012 to 2018. [online]
View (Accessed June 1, 2020).
|
|
|
|
|
Storgårds E (2000). Process hygiene control in beer production and dispensing (T. editing L. Ukskoski, ed.) (ISBN 951-38-5559-7). [online]
View
|
|
|
|
|
Thomas D (2010). How clean is clean? - Good practices and good relationships to keep the brewery clean.
View
|
|
|
|
|
UK Government (2020). Gas and electricity prices in the non-domestic sector. [online]
View (Accessed May 27, 2020).
|
|