Full Length Research Paper
ABSTRACT
Powder fractions of Dichrostachys glomerata were produced by drying the fruits to 10% moisture, grinding followed by sieving to differentiate powders of sizes <180, 180-212, 212-315 and ≥315 µm, respectively. The obtained powders were analysed for their phenolics profile and in vivo antioxidant activities in rats were evaluated. Lyophilized ethanolic extract and crude powder were used for comparison. The phenolic compounds (mg/100 g): epicatechin (19-30), caffeic acid (313-468), protocatechuic acid (338-799) and quercetin (369-639) were significantly correlated (r>0.65; p<0.05). For in vivo antioxidant properties, the rats fed with fine powders reduced their malondialdehyde in all organs from 19 to 64% (180-212 µm) and 29 to 38% (<180 µm), while increase in catalase (250-1310% and 249-1121%) and superoxide dismutase (72-251 and 5-404%) were observed, respectively in the 180- 212 µm fraction and ethanolic extract powder. The 180 to 212 µm fraction with high phenolic contents protected rats from oxidation by modulating malondialdehyde, superoxide dismutase and catalase levels similar to ethanolic extract powder, although still lower than vitamin C. Thus, sieving fractionation has a huge potential as substitute to ethanol extraction of phenolic compounds from D. glomerata to obtain powder fractions usable as natural bio-functional ingredients.
Key words: Dichrostachys glomerata fruits, powder, sieve fractionation, phenolic compounds, antioxidant bioactivity.
INTRODUCTION
The manufacture of nutraceuticals (food products that offered health and fitness benefits beyond their nutritional value) is, nowadays, a research challenge in the area of food and nutrition sciences. Nutraceuticals are gaining prominence in the daily diet of many people due to the increasing prevalence of lifestyle diseases and people consciously taking preventive healthcare measures. Global nutraceuticals market accounted for $379.06 billion in 2017 and is expected to grow at a Compound Annual Growth Rate (CAGR) of 7.6% to reach $734.60 billion by 2026 (Globe Newswire, 2019). The nutraceutical sector included functional foods and dietary supplements among others. Dietary supplements are commercialised in a variety of forms including tablets, capsules, drinks and powders. Popular supplements are vitamins E and D, minerals like Ca and Fe, herbs and polyphenols (Hamid et al., 2014).
The ADG/CDSp (alternative drying and grinding followed by controlled differential sieving process) is a technique aiming at concentrating active plant molecules in a specific powder fraction. The technique consisted of drying at computed assisted drying rate, finely grinding of the plant material followed by sieving on column sieves of decreasing sizes. Using this technique, many authors demonstrated that powder fractionation resulted in concentrating polyphenols in finer particle size (Becker et al., 2016; Zaiter et al., 2016; Becker et al., 2017; Deli et al., 2019). Phenolic and polyphenolic compounds represented the most widely distributed plant secondary metabolites exerting their beneficial effects as free radical scavengers and chelators of pro-oxidant metals, and thus prevent oxidation. Recent reports on Rosa canina and Salix alba showed that ADG/CDSp technology has an advantage over hydroethanol solvent in extracting antioxidant compounds (Soualeh et al., 2018, 2019). Powder formulation of nutraceuticals using the ADG/CDSp technology offered an opportunity for economic growth for developing countries endowed with rich biodiversity and a good knowledge of the health benefits of certain indigenous plant species such as Dichrostachys glomerata fruits (Deli et al., 2020).
D. glomerata fruit is a spice widely used in Cameroon for soup seasoning. The fruits are either harvested dry or fresh then sun-dried. Studies have reported high polyphenol content, anti-inflammatory and antioxidant activities of D. glomerata (Deli et al., 2019). More so, CDSp fine fractions of D. glomerata concentrated more micronutrients and exhibited antioxidant activities as ethanolic extract (Deli et al., 2020). However, there is no report on the polyphenolic profile in relation to antioxidant activity of CDSp fractions. Hence the objective of this study was to evaluate the polyphenol profile and in vivo antioxidant activities of the CDSp powder fractions of D. glomerata fruits.
MATERIALS AND METHODS
Chemical reagent and sampling
The chemicals and solvents used in this study were purchased from Sigma-Aldrich (Saint Louis, USA) and were of analytical grade. Dried fruits of D. glomerata were purchased in a local market in Yaounde, Cameroon. They were cleaned to eliminate foreign materials, then sorted to degraded fruits before ground for powder production.
Milling of dried D. glomerata fruits and controlled differential sieving process
To produce the crude D. glomerata powder (CDG), the dried fruits were milled using an electric Ultra-Centrifugal Mill ZM 200 (Haan, Germany) operating at 12,000 rpm (8 049.6 g) with mesh sieve of 1 mm. The powder fractions were produced on an Analysette 3 Spartan apparatus (Fritsch, Idar-Oberstein, Germany). In this respect, 100 g of the crude powder was poured on a sieve column of decreasing sizes 315, 212, and 180 µm, ending with the collecting pan. The system was allowed to vibrate at an amplitude of 0.5 mm for 10 min generating powders of respective sizes >315, 212-315, 180-212 µm, and <180 µm (Figure 1). Thereafter, the powders on the respective sieves and collecting pan (CDSp powder fractions) were collected, packed in polyethylene bags and stored at 10°C until analyses.

Production of ethanolic extract powder fraction
About 100 g of D. glomerata crude powder was mixed with ethanol (ratio 1/10, w/v) and stirred (Variomag Poly) for 24 h at 18°C. The mixture was filtered using a Whatman paper No.1 of pore size 12- 15 µm and ethanol evaporated (evaporator BUCHI - R210/215) at 40°C at a pressure of 175 mbar. The resulting solution was frozen at -18°C for 24 h and lyophilised at -60°C for 48 h. The resulting powder, called ethanolic extract powder (EE), was conditioned as the powder fractions in polyethylene bags and stored at 10°C until analyses.
Phenolic characterization of D. glomerata powder samples
Extraction of phenolic compounds
All the powder samples including ethanolic extract powder, <180, 180-212, 212-315 and >315 µm, respectively along with the crude powder were each used for extraction. About 2 g of powder was mixed with 20 ml methanol/water (70/30, v/v) solution. The mixture was stirred at 300 rpm for 24 h at room temperature (18±2°C) and then filtered with a Whatman No.1 (GE Healthcare companies, China) of 2-3 µm pore size. Thereafter, the supernatant was brought to 15 ml by addition of extraction solvent and stored at 4°C for phenolic profile analysis.
LC-MS analysis of phenolics
The bioactive components were determined on a LC-MS 2020 equipment (Shimadzu, Tokyo, Japan), which contained an HPLC unit coupled to an electro-spray ionization source permitting the MS detection. The injection volume was 20 μl, the quaternary pump delivered a flow rate of 0.6 ml/min and the oven temperature was set at 30°C. Separation was performed on C18 reverse phase Gemini column, having the following parameters: internal diameter 4.6 mm, length 150 mm, particle size 3 μm and pore size 130 Å. The two eluent solvents were water (A), acidified to 0.5% with formic acid, and acetonitrile (B). The 30 min used gradient was as follows: 0-10 min from 90:10 to 85:15 (A:B) (linear gradient); 10-15 min from 85:15 to 80:20 (A:B) (linear gradient); 15-18 min from 80:20 to 75:25 (A:B) (linear gradient); 18-22 min from 75:25 to 60:40 (A:B) (linear gradient); 22-25 min held at 50:50 (A:B) (isocratic); 25-28 min from 50:50 to 90:10 (A:B) (linear gradient); 28-30 min held at 90:10 (A:B) (re-equilibration step). In the mass detector, the nebulization gas flow rate was 1.5 l/ min, drying gas flow rate was 10 l/min, the desolvation line temperature was 250°C and the source temperature was 350°C. The probe voltage was −4 kV, and the electrospray ionization mode was in the negative. The identification of phenolic compounds in plant extracts was based on standard compounds analyses opposing m/z ratio and LC retention time (t). Quantification of the phenolics was done on a calibration curve made with each standard (protocatechuic acid, catechin, chlorogenic acid, caffeic acid, epicatechin, p-coumaric acid, ferulic acid, rutin, quercetin) (0.1-1 mg/ml) prepared in methanol/water (70:30, v/v). The linearity and sensitivity were evaluated by determining the limits of detection (LOD) and quantification (LOQ), defined as the concentration leading to a signal-to-noise ratio (S/N) of 3 and 10, respectively.
Evaluation of the antioxidant activities of D. glomerata powder samples
The experiment was operated on adult male Wistar rats (Rattus norvegicus), 3 months old of weight range 200-230 g according to the procedure reported earlier by Ngatchic et al. (2013) with some modifications. The animals randomly organised in groups of 5 rats per cage were housed at the animal house (25±2°C) of Biophysics, Food Biochemistry and Nutrition Laboratory, ENSAI, University of Ngaoundere in Cameroon. The rats were organised into nine (9) groups composing of 1 normal control group, 1 negative control group, 1 positive control group, and 6 experimental groups (receiving ethanolic extract powder, <180 µm, 180-212 µm, 212- 315 µm, >315 µm and the crude powder). The normal control group was administered normal food while the others received high-fat diet. Before food was given to the rats, they were administered per os 1 h before, 10 ml of aqueous suspension of D. glomerata powder (for the experimental groups), vitamin C 1 mg/ml (for the positive control group), and distilled water (for the negative controlled group). D. glomerata powders were administered at a dose of 250 mg/kg while vitamin C was administered at a dose of 20 mg/kg body weight. The animals had free access to water. The experiment lasted 28 days after which the rats were fasted overnight and anesthetized with chloroform to obtain 1 to 2 ml of blood sample withdrawn by cardiac puncture. The blood was centrifuged at 3500 rpm to obtain serum, which was kept frozen at - 4°C until used for analyses. Liver, kidney and heart were equally dissected and the homogenates of these organs were prepared for antioxidant markers analyses. Based on the protein content determined according to the method of Lowry et al. (1951), malondialdehyde content (MDA, μmol/mg protein), catalase (CAT, unit/mg protein) and superoxide dismutase (SOD, Units/mg protein) activities were determined in the organ homogenates and blood plasma as reported by Ngatchic et al. (2013). This study was carried out with approval from the Cameroonian National Ethics Committee Ref. No. FWIRD00001954.
Statistical analysis
Obtained data were recorded in Ms Excel and analyses were carried out in triplicates. Results were expressed as mean ± standard error mean deviation. One-way analysis of variance (ANOVA), followed by Duncan’s multiple range test were used to determine significant differences (p ≤ 0.05) among the samples using Statgraphics. Principal components analysis (PCA) was performed for structuring correlation between studied samples, phenolic content and antioxidant activities (XLSTAT, version 2016, Addinsoft, New York, US).
RESULTS
Phenolics profile of D. glomerata powders
The phenolic contents of D. glomerata crude powder and ethanolic extract are shown in Figure 2A. The principal phenolic compounds analysed were phenolic acids (protocatechuic acid, p-coumaric acid, ferulic acid, caffeic acid and chlorogenic acid), flavonoids (quercetin and rutin) and tannins (catechin and epicatechin).
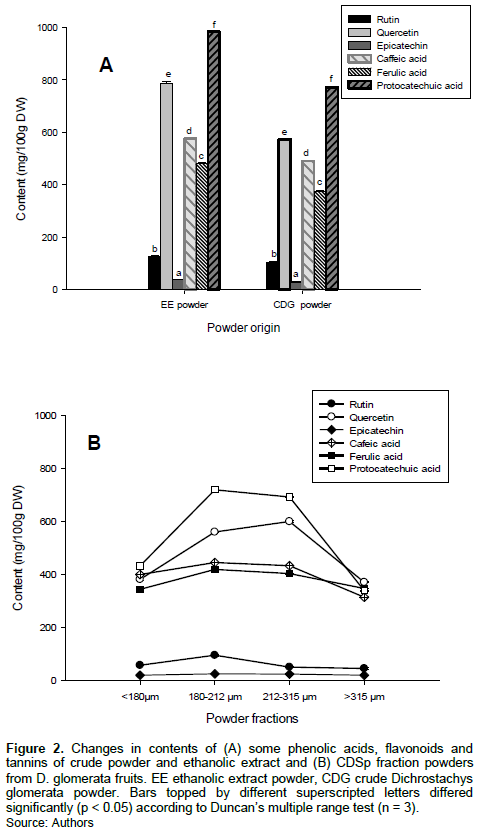
It can be seen from Figure 2A that of the nine phenolic compounds screened, p-coumaric acid, catechin and chlorogenic acid was non-detectable. When compared to commonly consumed dried fruits and vegetables, D. glomerata powders have higher contents of protocatechuic acid, quercetin, caffeic acid and ferulic acid with values varied from 338-799, 369-639, 313-468 and 298-418 mg/100 g DW, respectively (M'hiri et al., 2017; Sir Elkhatim et al., 2018; Zhang et al., 2018). Rutin and epicatechin were observed at lower quantities with values ranged between 45-101 and 19-30 mg/100 g DW, respectively.
Among the examined flavonoids, quercetin was the most prominent while phenolic acids were the most represented polyphenols in the D. glomerata powders, with the quercetin content comparable to that of onion (Allium cepa), one of the most important sources of quercetin (341-647 mg/100 g DW) (Domanska et al., 2018). These values are higher than those of green beans (1.1 mg/100 g DW), white radishes (0.9 mg/100 g DW), carrots (3.75 mg/100 g DW) and celery (8.05 mg/100 g DW), as well as those reported by Cao et al. (2010) on 100 edible vegetables and fruits. The contents in each of the phenolic compounds analysed were higher in the ethanolic extract powder, while significant variation (p < 0.05) were found among the CDSp fractions (Figure 2B). The CDSp fraction having the highest quercetin contents was 212-315 µm, while the fraction exhibiting the highest ferulic acid, protocatechuic acid, rutin and epicatechin was 180-212 µm.
MDA content, CAT and SOD activities of D. glomerata powders
One important consequence of lipid accumulation in the body is the increase in lipid peroxidation and reduction in antioxidative enzymes. Figure 3 showed the change in MDA content, CAT and SOD activities in the plasma and some organs of rats in the control groups. It is evident from Figure 3A that there was a significant (p < 0.05) MDA increase in the plasma and organs of rats in the negative control, while CAT (Figure 3B) and SOD (Figure 3C) activities decreased significantly. The percentage increase (compared to the normal control group) in MDA ranged from 52% in the heart to 176% in the liver, while the percentage reduction in CAT and SOD ranged from 10-59% and 69-80%, respectively. The importance of administering vitamin C was the reduction in oxidation processes in the body (Deli et al., 2020). This is seen in Figure 3 where the MDA decreased significantly by 2- 87% in rats administered with vitamin C (positive control group), while the CAT and SOD increased significantly from 0-148 and 11-124%, respectively.
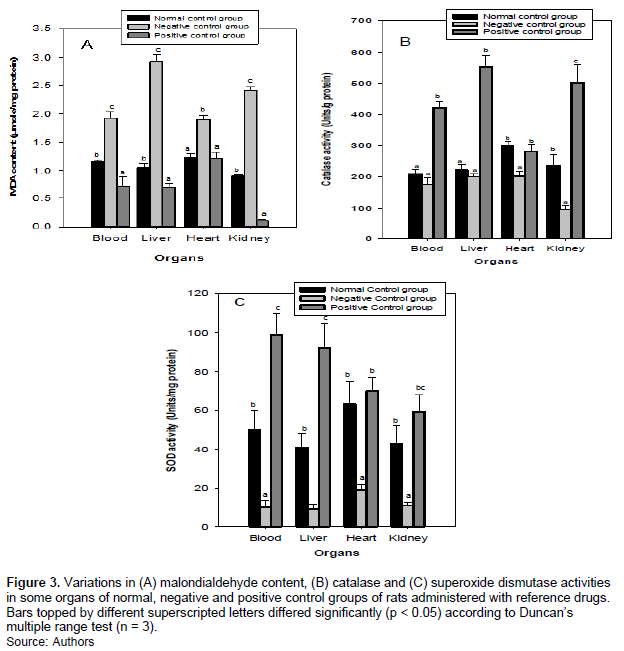
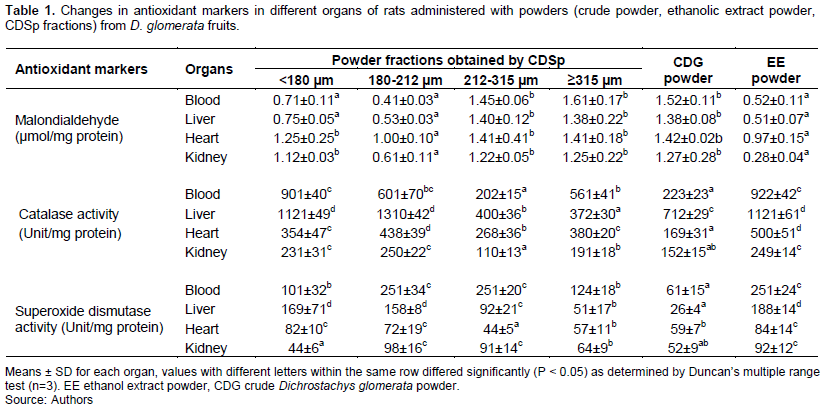
Table 1 presented a summary of the MDA content, CAT and SOD levels in the plasma and some organs of rat administered with D. glomerata powders. Generally, the group administered with CDG powder showed a decrease in MDA from 14-41%, while the group administered with EE powder showed a decrease in MDA from 21-69% in all organs. Otherwise, a decrease in MDA was observed upon administration of 180-212 µm powder fraction (19-64%, in all organs), while a decrease in MDA was observed only in blood (38%) and liver (29%) for the powder fraction <180 µm. An increase was observed for other fractions (0-39%), but this was still lower as compared to the negative control group, which was 176% in the liver. However, the decrease in MDA observed for the 180-212 µm fraction and ethanolic extracts of D. glomerata were still significantly lower (p < 0.05) than the positive control group administered with vitamin C. Inversely, much more increase in CAT (250-1310% and 249-1121%) and SOD (72-251 and 5-404%) levels were observed respectively in the 180-212 µm fraction and ethanolic extract powder as compared to the vitamin C control group (11-124% increase for SOD and 0-148% increase for CAT). These results suggested an interaction between molecules in D. glomerata powders, which may boost the oxidative status of rats in different aspects (Yang et al., 2016; Gao et al., 2017). For instance, D. glomerata was reported by Deli et al. (2020) to be a source of oligo elements such as Cu (0.8 g/100 g), Zn (0.2 mg/100 g), Mn (2.2 mg/100 g) and Se (0.11 mg/100 g), which are components of enzymes involved in the antioxidative and detoxification processes.
DISCUSSION
Figure 4 showed the correlation between the different variables studied as well as the distribution of D. glomerata powders on the axis system F1 × F2. The two axes F1 and F2, based on the correlation between the variables, showed 84.98% variation among the powder properties. F1, which showed 61.86% of inertia, was negatively correlated to most phenolic compounds (r varying from -0.65 for ferulic acid to -0.79 for epicatechin) and the antioxidant enzymes (r varying from -0.70 for SOD in the blood to -0.89 for SOD in the liver). On the other hand, there was a positive correlation between F1 and MDA (r ranged from 0.93 to 0.98). F2 showed only 23.12% variation and was mostly correlated with quercetin (r = 0.74), SOD in the kidney (r = 0.68) and CAT in the blood (r = -0.68). The MDA content in all the organs, as shown in Figure 4, were opposite to phenolic compounds and the CAT and SOD activities, reflecting a perfect negative correlation. Thus, phenolic compounds may be implicated in reducing MDA levels, but this might be mostly associated with rutin, caffeic acid and epicatechin which are more negatively correlated (ranged from -0.75 to -0.89) to MDA group. As seen in the axis system, F1 × F2, SOD activities were not very close, suggesting different behaviour vis-a-vis the phenolic compounds. The SOD in the liver and heart were closer to each other and varied in same manner as CAT, but far from the SOD in kidney and blood, which were more positively influenced by the phenolic compounds. Thus, phenolic compounds contained in the D. glomerata powders might have protected the kidney and plasma cells membranes against damage caused by free radicals. The structure and lipophilic property of polyphenols have been shown to be important factors from which their antioxidant activity is derived, since they affected the depth of incorporation of these compounds into the lipid phase of the cell membranes (Djeridane et al., 2010).
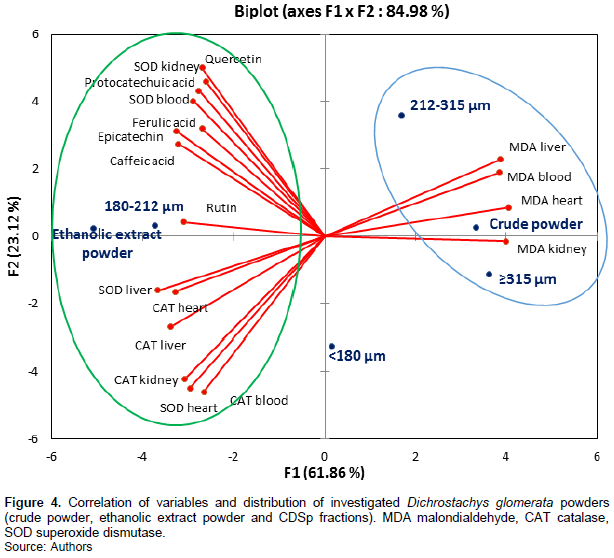
This justified that the powder fractions richest in phenolic compounds are classified among the powder fractions having the highest SOD and CAT activities. Similar biological effects of the powder fractions from R. canina and S. alba have been demonstrated by Soualeh et al. (2018). These authors also noted an inhibition of the MDA production in rat organs placed under oxidative stress conditions and receiving different powder fractions from Hedera nodosa and Helix scrophularia (Soualeh et al., 2019). This has been attributed to the presence of various bioactive compounds such as polyphenols, flavonoids and tannins.
Figure 4 also showed the distribution of D. glomerata powder samples in the same axis system F1 x F2. It appeared that the 180-212 µm fraction was very close to the ethanolic extract commonly used as a standard for administration to patients. These powders exhibited characteristics between high levels of antioxidant enzymes and high levels of phenolic compounds. They were opposite to D. glomerata crude powder, which exhibited high levels of MDA than all the powders tested. The powder fractions, which were revealed to be poor in phenolic compounds, failed in reducing the MDA levels as compared to the 180-212 µm fraction.
CONCLUSION
The sieve fractionation, otherwise called CDSp technology, showed significant effect on the phenolic composition and antioxidant bioactivity of D. glomerata fruit powders. Epicatechin, caffeic acid, protocatechuic acid, rutin, ferulic acid and quercetin were all affected by CDSp to different extents. One granulometric class of interest 180-212 µm was highlighted for D. glomerata powders, which had phenolic content not far from that of ethanolic extract powder. There was a strong correlation between phenolic contents and antioxidant activities, implying that identified polyphenolic compounds were mainly responsible for the antioxidant activities of D. glomerata powders. More specifically, the 180-212 µm powder fraction recorded the highest antioxidant activities compared to the crude powder and other fractions. It was postulated that grinding enhanced the extraction yield of bioactive compounds and sieving induced the discrimination of these compounds according to particle size. The CDSp approach would be of interest for nutraceutical purposes by providing plant powder fractions with enhanced biological activity and contents in active compounds.
CONFLICT OF INTERESTS
The authors have not declared any conflict of interests.
ACKNOWLEDGEMENTS
The research was supported by the Extrapole project funded by the former Lorraine Region (France). The authors are grateful to the LABBAN (Laboratoire de Biophysique, Biochimie Alimentaire et Nutrition) of The University of Ngaoundere, Cameroon) as well as the URAFPA (Unite de Recherche Animal et Fonctionnalités des Produits Animaux) of Lorraine University, France for their technical support.
REFERENCES
|
Becker L, Zaiter A, Petit J, Karam M-C, Sudol M, Baudelaire E, Scher J, Dicko A (2017). How do grinding and sieving impact on physicochemical properties, polyphenol content, and antioxidant activity of Hieracium pilosella L. powders. Journal of Functional Foods 35:666-672. |
|
|
Becker L, Zaiter A, Petit J, Zimmer D, Karam M-C, Baudelaire DE, Scher J, Dicko A (2016). Improvement of antioxidant activity and polyphenol content of Hypericum perforatum and Achillea millefolium powders using successive grinding and sieving. Industrial Crops and Products 87:116-123. |
|
|
Cao J, Chen W, Zhang Y, Zhao X (2010). Content of Selected Flavonoids in 100 Edible Vegetables and Fruits. Food Science and Technology Research 16(5):395-402. |
|
|
Deli M, Baudelaire E, Nguimbou RM, Njintang YN, Scher J (2020). Micronutrients and in vivo antioxidant properties of powder fractions and ethanolic extract of Dichrostachys glomerata Forssk. Food Science and Nutrition 8:3287-3297. |
|
|
Deli M, Djantou EB, Ngatchic MJT, Petit J, Njintang YN, Scher J (2019). Successive grinding and sieving as a new tool to fractionate polyphenols and antioxidants of plants powders: Application to Boscia senegalensis seeds, Dichrostachys glomerata fruits and Hibiscus sabdariffa calyx powders. Food Science and Nutrition 7:1795-1806. |
|
|
Djeridane A, Yousfi M, Brunel JM, Stocker P (2010). Isolation and characterization of a new steroid derivative as a powerful antioxidant from Cleome arabica in screening the in vitro antioxidant capacity of 18 Algerian medicinal plants. Food and Chemical Toxicology 48:2599-2606. |
|
|
Domanska U, Wisniewska A, Dabrowski Z (2018). Extraction of Quercetin from red onion (Allium cepa L.) with ionic liquids. Journal of Chromatography and Separation Techniques 1(2):116. |
|
|
Gao Y, Chu S, Shao Q, Zhang M, Xia C, Wang Y (2017). Antioxidant activities of ginsenoside Rg1 against cisplatin-induced hepatic injury through Nrf2 signaling pathway in mice. Free Radical Research 51:1-13. |
|
|
Globe Newswire (2019). The Nutraceuticals - Global Market Outlook (2017-2026). Research And Markets, Dublin. Available at: |
|
|
Hamid N, Azar B, Hedayatollah S, Mahmoud R, (2014). New concepts in nutraceuticals as alternative for pharmaceuticals. International Journal of Preventive Medicine 5(12):1487-1499. |
|
|
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951). Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry 193:265-275. |
|
|
M'hiri N, Ioannou I, Ghoul M, Boudhrioua NM (2017). Phytochemical characteristics of citrus peel and effect of conventional and nonconventional processing on phenolic compounds: A review. Food Reviews International 33(6):587-619. |
|
|
Ngatchic MTJ, Njintang NY, Oben JE, Mbofung CMF (2013). Protein quality and antigrowth effect of protein isolate of Mucuna (Mucuna pruriens) and Canavalia (Canavalia ensiformis) seeds. Scholars Academic Journal of Biosciences 1(5):183-191. |
|
|
Sir Elkhatim KA, Elagib RAA, Hassan AB (2018). Content of phenolic compounds and vitamin C and antioxidant activity in wasted parts of Sudanese citrus fruits. Food Science and Nutrition 6(5):1214-1219. |
|
|
Soualeh N, Stiévenard A, Baudelaire DE, Bouayed J, Soulimani R (2019). Powders with small microparticle size from Hedera helix (L.) and Scrophularia nodosa (L.) exhibited high preventive antioxidant activity against H2O2-induced oxidative stress in mouse primary spleen cells. International Journal for Vitamin and Nutrition Research 88(3-4):208-218. |
|
|
Soualeh N, Stiévenard A, Baudelaire DE, Soulimani R, Bouayed J (2018). Improvement of cytoprotective and antioxidant activity of Rosa canina (L.) and Salix alba (L.) by controlled differential sieving process against H2O2-induced oxidative stress in mouse primary splenocytes. International Journal for Vitamin and Nutrition Research 87(3-4):191-200. |
|
|
Yang Q, Wang F, Rao J (2016). Effect of putrescine treatment on chilling injury, fatty acid composition and antioxidant system in kiwifruit. PLoS One 11(9) e0162159. |
|
|
Zaiter A, Becker L, Petit J, Baudelaire E, Zimmer D, Karam M-C, Scher J, Dicko A (2016). Antioxidant and antiacetylcholinesterase of different granulometric classes of Salix alba (L.) bark powders. Powder Technology 301:649-656. |
|
|
Zhang H, Yang YF, Zhou ZQ (2018). Phenolic and flavonoid contents of mandarin (Citrus reticulata Blanco) fruit tissues and their antioxidant capacity as evaluated by DPPH and ABTS methods. Journal of Integrative Agriculture 17(1):256-263. |
|
Copyright © 2024 Author(s) retain the copyright of this article.
This article is published under the terms of the Creative Commons Attribution License 4.0